Advanced Polymer Reverse-Phase Purification for High-Purity Carbetocin Manufacturing
Introduction to Advanced Carbetocin Purification Technologies
The pharmaceutical landscape for oxytocin analogues demands exceptionally high purity standards, particularly for potent uterotonic agents like carbetocin. Patent CN103435687A introduces a groundbreaking methodology specifically designed to address the persistent challenges in purifying synthetic long-acting oxytocin nonapeptide analogues. This technology leverages a sophisticated polymer reverse-phase chromatography system to achieve separation efficiencies that were previously unattainable with conventional silica-based media. The core innovation lies in the precise manipulation of mobile phase gradients combined with a specialized polymeric stationary phase, which offers superior selectivity for separating structurally similar peptide isomers. For R&D directors and process chemists, this represents a significant leap forward in ensuring the safety profile of the final API by effectively eliminating genotoxic or unstable impurities.
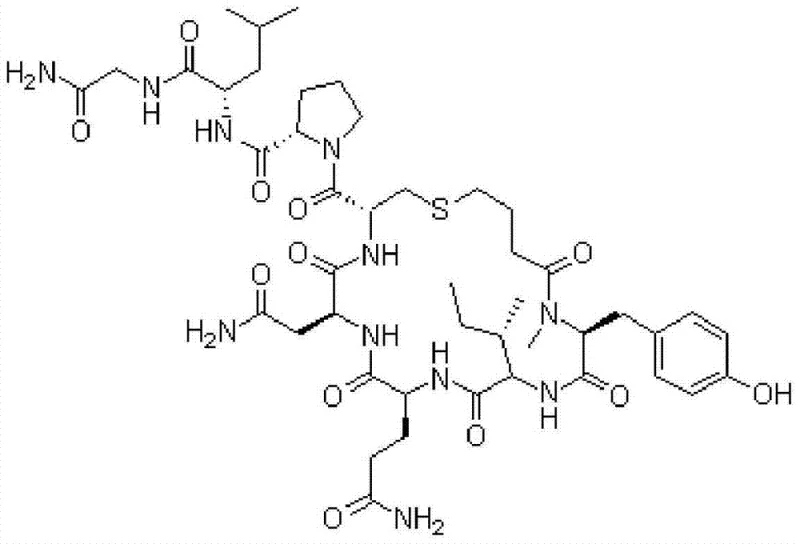
As illustrated in the molecular structure above, carbetocin possesses a complex cyclic architecture that is highly susceptible to epimerization during synthesis. The presence of multiple chiral centers necessitates a purification strategy capable of distinguishing between L-amino acid configurations and their D-isomer counterparts. The patent details a robust process that not only enhances the overall yield but also guarantees the removal of critical impurities that could compromise patient safety. By adopting this advanced purification protocol, manufacturers can align their production capabilities with the stringent regulatory requirements of global health authorities, ensuring a reliable supply of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of carbetocin has relied heavily on traditional reversed-phase chromatography often coupled with anion exchange techniques, as disclosed in earlier patents such as CN101531705. These conventional approaches frequently suffer from inadequate resolution power when attempting to separate the target peptide from its closely related stereoisomers, specifically the D-Cys 5-carbetocin impurity. The structural similarity between the L-cysteine and D-cysteine variants results in overlapping retention times on standard C18 columns, leading to co-elution and compromised purity profiles. Furthermore, the use of harsh modifiers like trifluoroacetic acid (TFA) in older methods introduces additional downstream processing burdens, requiring extensive salt conversion steps to meet pharmacopeial limits for residual solvents and counter-ions. This inefficiency not only drives up manufacturing costs but also results in significant product loss, with yields often falling short of commercial viability targets.
The Novel Approach
The novel approach detailed in CN103435687A circumvents these limitations by utilizing a specialized polymer reverse-phase packed column, specifically the UniPS TM10 series, which exhibits distinct selectivity characteristics compared to silica-based supports. This method employs a meticulously optimized gradient elution program using a benign mobile phase system comprising 0.3% acetic acid and acetonitrile. The gradient begins at a low organic concentration of 5% and ramps precisely to 25% and subsequently to 40% over a defined timeframe, creating a separation window where the hydrophobic interactions of the impurity differ sufficiently from the main product. This strategic adjustment allows for the baseline resolution of D-Cys 5-carbetocin, reducing its content to below the critical threshold of 0.1%. The elimination of complex ion-exchange steps and the use of volatile, non-toxic acetic acid streamline the entire workflow, offering a direct path to high-purity isolation without the need for cumbersome salt swapping procedures.
Mechanistic Insights into Polymer Reverse-Phase Separation
The efficacy of this purification method is rooted in the unique surface chemistry of the polymer stationary phase, which provides a more homogeneous distribution of hydrophobic binding sites compared to irregular silica particles. In the context of peptide separations, the polymer matrix minimizes secondary interactions such as silanol activity, which can often cause peak tailing and reduced resolution for basic amino acid residues. The separation mechanism relies on the subtle differences in the three-dimensional conformation of the cyclic peptide caused by the epimerization at the cysteine residue. The D-isomer impurity, shown below, adopts a slightly different spatial arrangement that alters its exposure of hydrophobic side chains to the stationary phase.

By fine-tuning the acetonitrile gradient, the method exploits these conformational differences to achieve differential elution. The initial hold at 5% mobile phase B ensures that highly polar components are washed through or retained strongly, while the subsequent ramp to 25% and 40% selectively desorbs the target carbetocin while leaving the more tightly bound or differently interacting D-Cys impurity behind or eluting it in a distinct fraction. This level of control is critical for managing the impurity profile, as D-Cys 5-carbetocin is known to be unstable and prone to discoloration, which can severely impact the aesthetic and chemical stability of the final drug product. The use of acetic acid as the ion-pairing agent further modulates the protonation state of the peptide, optimizing the interaction kinetics for maximum resolution without introducing persistent fluorinated contaminants.
How to Synthesize Carbetocin Efficiently
The implementation of this purification protocol requires strict adherence to the specified chromatographic parameters to ensure reproducibility and compliance with quality standards. The process begins with the dissolution of the crude peptide in purified water followed by pH adjustment to approximately 3.4, creating the optimal loading condition for the polymer column. Detailed operational steps regarding column equilibration, sample loading volumes, and fraction collection criteria are essential for scaling this technology from laboratory to commercial production. For a comprehensive guide on executing this synthesis and purification workflow, please refer to the standardized procedure outlined below.
- Dissolve crude carbetocin in water and adjust pH to acidic conditions (approx. pH 3.4) using acetic acid.
- Equilibrate the polymer reverse-phase column (UniPS TM10) with 0.3% acetic acid aqueous solution.
- Elute using a specific gradient: start at 5% acetonitrile, increase to 25% over 5 minutes, then to 40% over 30 minutes.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this polymer-based purification technology offers substantial advantages for procurement managers and supply chain leaders focused on cost efficiency and reliability. The shift away from complex multi-step purification sequences involving anion exchange resins significantly simplifies the manufacturing workflow, thereby reducing the consumption of consumables and the labor hours associated with process monitoring. The use of acetic acid as a mobile phase modifier presents a direct cost reduction in pharmaceutical intermediate manufacturing, as it is considerably more economical and easier to handle than specialized fluorinated acids, while also mitigating the environmental hazards associated with TFA disposal. This simplification translates into a more robust supply chain with fewer potential points of failure.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive ion-exchange materials and the associated buffer preparation and conversion steps, leading to significant operational expenditure savings. By achieving higher recovery rates through improved resolution, the overall cost per gram of pure API is drastically lowered, enhancing the economic viability of large-scale production runs. The reduction in solvent complexity also lowers waste treatment costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the polymer column material ensures consistent performance over extended usage cycles, reducing the frequency of column replacement and minimizing downtime for maintenance. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for global pharmaceutical clients. Furthermore, the use of common, readily available solvents like acetonitrile and acetic acid mitigates the risk of supply disruptions associated with specialty reagents.
- Scalability and Environmental Compliance: The method is inherently scalable, allowing for seamless transition from pilot scale to multi-ton annual production without the need for fundamental process re-engineering. The avoidance of halogenated solvents and heavy metal catalysts aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. This compliance is increasingly becoming a key differentiator in vendor selection processes for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this purification technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: Why is D-Cys 5-carbetocin difficult to remove?
A: D-Cys 5-carbetocin is a stereoisomer impurity with very similar physicochemical properties to the target molecule, making separation via standard silica-based C18 columns challenging without specialized polymer phases.
Q: What is the advantage of using acetic acid over TFA in this process?
A: Using 0.3% acetic acid instead of trifluoroacetic acid (TFA) simplifies downstream processing, reduces toxicity concerns, and lowers the cost of mobile phase preparation while maintaining effective separation.
Q: What purity levels can be achieved with this method?
A: This patented method consistently achieves purity levels exceeding 99.5%, with the specific toxic impurity D-Cys 5-carbetocin reduced to below 0.1%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of peptide therapeutics like carbetocin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chromatographic techniques described in recent patents are effectively translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the <0.1% impurity threshold for D-Cys 5-carbetocin. Our commitment to quality assurance guarantees that our clients receive materials that are ready for formulation without the need for additional reprocessing.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for high-value peptide intermediates. By leveraging our expertise in advanced purification technologies, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
