Advanced Catalytic Hydrogenation Technology for High-Content Trimethylhydroquinone Production
Introduction to Advanced TMHQ Manufacturing Technology
The global demand for high-purity Vitamin E intermediates continues to surge, driving the need for robust and scalable synthetic routes for Trimethylhydroquinone (TMHQ). Patent CN102295536B introduces a significant technological advancement in this domain by optimizing the critical catalytic hydrogenation step of 2,3,5-trimethylbenzoquinone. Unlike traditional methods that rely on alcoholic solvents which often compromise product quality through discoloration, this innovative process utilizes ethyl acetate as the reaction medium. This strategic solvent substitution, combined with a refined post-reaction workup involving hot filtration and azeotropic distillation, addresses long-standing industry pain points regarding product color, stability, and solvent recovery efficiency. For R&D directors and procurement specialists alike, this methodology represents a pivotal shift towards more sustainable and economically viable manufacturing of high-content pharmaceutical intermediates.
The technical breakthrough lies not merely in the choice of solvent but in the holistic integration of reaction conditions and separation techniques. By maintaining specific temperature ranges between 75-85°C and hydrogen pressures of 0.2-1.0 MPa, the process ensures complete conversion while minimizing side reactions. Furthermore, the inclusion of a stabilization step using sodium hydrosulfite effectively mitigates the risk of re-oxidation, a common issue that plagues hydroquinone derivatives during isolation. This comprehensive approach guarantees a final product with exceptional purity levels, consistently exceeding 98%, making it an ideal reliable pharmaceutical intermediates supplier solution for downstream Vitamin E synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Trimethylhydroquinone has been fraught with challenges related to solvent selection and process complexity. Traditional catalytic hydrogenation protocols frequently employ lower alcohols such as methanol or ethanol as solvents. While these solvents offer good solubility for the quinone precursor, they possess a detrimental tendency to induce discoloration in the final TMHQ product. This phenomenon is widely attributed to the formation of quinhydrone complexes, where the hydroquinone product interacts with trace amounts of unreacted quinone or oxidation byproducts, resulting in a darkened, inferior quality material that fails to meet stringent pharmaceutical specifications. Additionally, conventional separation techniques often involve cooling the reaction mixture prior to filtration, which leads to premature crystallization of the product on the catalyst bed, causing significant yield losses and complicating catalyst recovery.
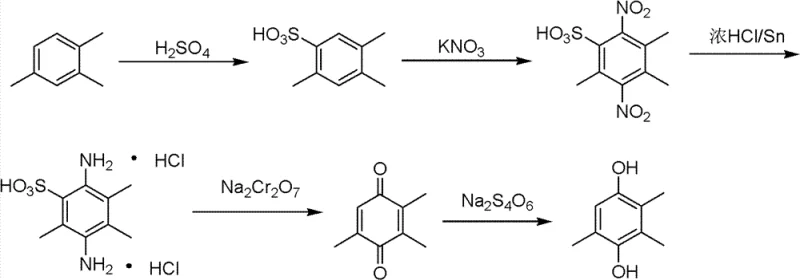
Beyond the reduction step, the precursor synthesis routes themselves have historically been cumbersome. As illustrated in the traditional pathways, methods such as the 1,2,4-trimethylbenzene route involve multiple hazardous steps including concentrated acid sulfonation and nitration. These processes generate substantial quantities of acidic wastewater and require complex purification sequences to isolate the 2,3,5-trimethylbenzoquinone intermediate. The cumulative effect of these inefficient upstream processes, combined with the limitations of the final reduction step, results in a manufacturing footprint that is both environmentally burdensome and economically suboptimal. The reliance on solvents that are difficult to recover completely without affecting product quality further exacerbates the cost structure, creating a compelling case for the adoption of novel, streamlined technologies.
The Novel Approach
The methodology disclosed in CN102295536B fundamentally reengineers the reduction phase by substituting problematic alcoholic solvents with ethyl acetate. This solvent switch is transformative; ethyl acetate provides an inert environment that prevents the discoloration reactions associated with alcohols, thereby preserving the pristine white appearance of the crystalline TMHQ. Moreover, the process incorporates a sophisticated hot filtration technique performed at 35-55°C. This operational parameter is critical as it keeps the product in solution during catalyst removal, ensuring that the valuable palladium catalyst is recovered cleanly without entrapping the product, thus maximizing yield. Following filtration, the solvent recovery system employs a unique water-addition distillation strategy. By adding water and continuing distillation, the process effectively removes residual ethyl acetate through azeotropic behavior, ensuring the final product is free from solvent contaminants that could impact stability.
This novel approach also integrates a chemical stabilization step that is often overlooked in standard protocols. After solvent removal, sodium hydrosulfite is introduced to the aqueous slurry. This reducing agent scavenges any dissolved oxygen or trace quinone species that may have formed during the workup, locking the molecule in its reduced hydroquinone state. The result is a product with superior storage stability and consistent high content, typically ranging from 98.9% to 99.4% as demonstrated in the patent examples. For supply chain managers, this translates to a more reliable inventory with extended shelf life, reducing the risk of waste due to degradation. The simplification of the workflow, eliminating the need for complex recrystallization to fix color issues, significantly shortens the production cycle time, enhancing overall plant throughput.
Mechanistic Insights into Pd/C Catalyzed Hydrogenation in Ethyl Acetate
The core of this technology rests on the heterogeneous catalytic hydrogenation of the quinone carbonyl groups using a 5% palladium on carbon (Pd/C) catalyst. In this mechanism, molecular hydrogen adsorbs onto the surface of the palladium particles, dissociating into reactive atomic hydrogen species. Simultaneously, the 2,3,5-trimethylbenzoquinone substrate adsorbs onto the catalyst surface. The atomic hydrogen then transfers to the oxygen atoms of the quinone, sequentially reducing the carbonyl groups to hydroxyl groups to form the hydroquinone structure. The choice of ethyl acetate as the solvent is mechanistically significant because, unlike alcohols, it lacks alpha-hydrogens that are susceptible to abstraction or oxidation under these conditions. This chemical inertness prevents the formation of aldehyde or ketone byproducts that could otherwise condense with the hydroquinone to form colored impurities, ensuring the electronic and steric environment around the catalyst favors clean reduction.
Impurity control is further managed through the precise manipulation of thermodynamic parameters during the workup. The hot filtration step is designed to exploit the solubility curve of TMHQ in ethyl acetate; by filtering above the saturation temperature, the system avoids nucleation on the catalyst pores. Subsequently, the addition of water serves a dual purpose: it facilitates the removal of ethyl acetate via low-boiling azeotropes, and it creates a biphasic environment where the TMHQ, being less soluble in hot water, can be induced to crystallize in a controlled manner upon cooling. The final addition of sodium hydrosulfite acts as a redox buffer. In the presence of trace air, hydroquinones are prone to auto-oxidation back to quinones. The hydrosulfite ion ($HSO_3^-$) has a lower oxidation potential than the hydroquinone, meaning it will oxidize preferentially, thereby sacrificially protecting the TMHQ product. This multi-layered approach to impurity management ensures that the final material meets the rigorous purity standards required for high-content vitamin E manufacturing.
How to Synthesize Trimethylhydroquinone Efficiently
The synthesis of high-content Trimethylhydroquinone via this patented route requires precise adherence to reaction parameters and workup sequences to maximize yield and purity. The process begins with the loading of 2,3,5-trimethylbenzoquinone and ethyl acetate into a pressurized hydrogenation vessel, followed by the addition of the palladium catalyst. The detailed standardized synthesis steps, including specific molar ratios, agitation speeds, and safety protocols for handling hydrogen gas, are outlined below to ensure reproducibility and safety in a commercial setting.
- Load 2,3,5-trimethylbenzoquinone, ethyl acetate solvent, and 5% palladium/carbon catalyst into a hydrogenation reactor and exchange atmosphere with hydrogen.
- Heat the mixture to 75-85°C under 0.2-1.0 MPa hydrogen pressure to facilitate catalytic reduction until pressure stabilizes.
- Perform hot filtration to remove catalyst, recover ethyl acetate via distillation, add water for azeotropic removal of residual solvent, and stabilize with sodium hydrosulfite before drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ethyl acetate-based hydrogenation process offers tangible strategic advantages beyond mere technical performance. The primary benefit lies in the drastic simplification of the downstream processing units. By eliminating the discoloration issues inherent to alcoholic solvents, the need for energy-intensive recrystallization steps or charcoal treatments to restore product color is removed. This reduction in unit operations directly correlates to lower utility consumption and reduced labor costs, contributing to significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the efficiency of the solvent recovery system, which utilizes water-assisted distillation, ensures that ethyl acetate losses are minimized. Since ethyl acetate is a commodity chemical with a stable price profile, the ability to recycle it efficiently without degradation adds a layer of predictability to the raw material budget.
- Cost Reduction in Manufacturing: The elimination of color-fixing steps and the high efficiency of catalyst recovery through hot filtration lead to substantial operational savings. By preventing product entrapment in the filter cake, the process maximizes the yield per batch, effectively lowering the cost of goods sold (COGS). Additionally, the avoidance of expensive solvent exchanges or complex purification trains reduces the capital expenditure required for equipment, as simpler distillation columns can be utilized compared to those needed for separating close-boiling alcoholic mixtures.
- Enhanced Supply Chain Reliability: The robustness of this process against variability in raw material quality enhances supply continuity. The use of sodium hydrosulfite as a stabilizer ensures that the product remains specification-compliant even during extended storage or transport, reducing the risk of customer rejections due to color drift. This stability allows for larger batch sizes and less frequent production runs, optimizing inventory management and ensuring a steady flow of high-purity pharmaceutical intermediates to downstream Vitamin E manufacturers without interruption.
- Scalability and Environmental Compliance: From an environmental perspective, the shift away from processes generating heavy metal waste or acidic effluents (common in precursor steps) to a cleaner hydrogenation step aligns with modern green chemistry principles. The closed-loop solvent recovery minimizes volatile organic compound (VOC) emissions, facilitating easier compliance with increasingly stringent environmental regulations. The process is inherently scalable, as the heat transfer and mass transfer characteristics of the hydrogenation in ethyl acetate are favorable for large-scale reactors, supporting the commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-content Trimethylhydroquinone synthesis method. These insights are derived directly from the patent data and practical experience in scaling similar hydrogenation processes, providing clarity on operational feasibility and quality assurance measures.
Q: Why is ethyl acetate preferred over alcoholic solvents for TMHQ hydrogenation?
A: Alcoholic solvents like methanol or ethanol can induce discoloration in Trimethylhydroquinone products, likely due to the formation of quinhydrone complexes. Ethyl acetate avoids this side reaction, ensuring superior product color and stability.
Q: How does the hot filtration step improve yield?
A: Filtering the reaction mixture while hot (35-55°C) prevents the premature crystallization of Trimethylhydroquinone on the filter cake, which would otherwise trap the catalyst and reduce overall recovery rates.
Q: What is the role of sodium hydrosulfite in the final stage?
A: Sodium hydrosulfite acts as a reducing agent to convert any trace amounts of re-oxidized trimethylbenzoquinone back to the hydroquinone form, ensuring high chemical purity and preventing color degradation during storage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethylhydroquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the production of high-purity Trimethylhydroquinone is a critical link in the global Vitamin E supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102295536B are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch. Our commitment to quality means that we do not just supply a chemical; we deliver a validated solution that integrates seamlessly into your downstream synthesis, guaranteeing the consistency required for pharmaceutical grade applications.
We invite you to collaborate with us to optimize your supply chain for Vitamin E intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced manufacturing capabilities can reduce your total landed cost. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, high-quality source of Trimethylhydroquinone that drives efficiency and reliability in your production operations.
