Advanced C13+C7 Wittig Strategy for High-Purity Vitamin A Acetate Manufacturing
Advanced C13+C7 Wittig Strategy for High-Purity Vitamin A Acetate Manufacturing
The global demand for high-purity vitamins and nutritional intermediates continues to surge, driven by expanding applications in pharmaceuticals, food additives, and cosmetics. In response to the inefficiencies of legacy manufacturing protocols, patent CN112457229B introduces a transformative C13+C7 synthetic route for Vitamin A Acetate. This innovative methodology leverages a strategic Wittig coupling to bypass the extensive step-count and safety hazards associated with traditional C14+C6 and C15+C5 pathways. By utilizing readily available starting materials such as bromoethanol and acetone, this process achieves a remarkable yield improvement to approximately 90%, calculated based on beta-ionone consumption. For R&D directors and procurement specialists seeking a reliable vitamin intermediate supplier, this technology represents a critical advancement in process intensification and cost optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
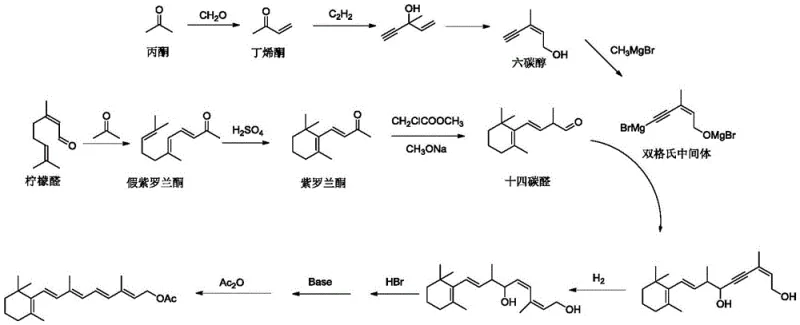
Historically, the industrial production of Vitamin A Acetate has been dominated by two primary technical routes, both of which suffer from significant structural and economic drawbacks. The C14+C6 route, while established, is notoriously complex, requiring more than 50 distinct raw materials and involving long reaction sequences that inflate fixed capital investment. Furthermore, this pathway relies on intermittent series reactions that are difficult to control at scale and necessitates the production of double Grignard reagents, posing severe safety risks during industrial amplification. Alternatively, the C15+C5 route utilizes expensive triphenylphosphine and generates substantial quantities of triphenylphosphine oxide solid waste, creating difficult disposal challenges. Additionally, the Vitamin A produced via the C15+C5 method often contains high levels of cis-isomers, which diminishes the utilization value and necessitates costly purification steps to meet pharmacopeial standards.
The Novel Approach
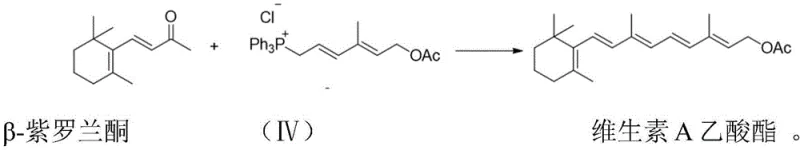
The novel C13+C7 strategy described in the patent data fundamentally restructures the synthesis logic to overcome these entrenched limitations. By constructing the carbon skeleton through the coupling of a C13 beta-ionone fragment with a newly synthesized C7 phosphonium salt, the process drastically shortens the reaction sequence. This approach eliminates the need for the hazardous double Grignard reagents found in the C14+C6 route and optimizes the use of phosphine reagents to minimize waste generation relative to output. The result is a streamlined workflow that not only enhances operational safety but also delivers superior stereochemical control, ensuring that the all-trans isomer content exceeds 85%. This high selectivity provides a distinct advantage for downstream processing, making it an ideal solution for cost reduction in vitamin manufacturing.
Mechanistic Insights into C13+C7 Wittig Coupling
The core of this technological breakthrough lies in the precise construction of the C7 side chain and its subsequent coupling via a Wittig reaction. The process initiates with the protection of bromoethanol using acetone under acid catalysis to form a stable ketal intermediate, which serves as the foundation for the Grignard reagent. This ketal compound then undergoes an in-situ Grignard reaction with a C5 aldehyde (4-acetoxy-2-methyl-2-butenal) in the presence of magnesium powder. Following acidolysis, this sequence yields a high-purity C7 alcohol. The conversion of this alcohol into a phosphonium salt using triphenylphosphine and hydrochloric acid sets the stage for the final, critical bond-forming event. This modular assembly allows for rigorous quality control at each stage, ensuring that impurities are minimized before the final coupling.
The final step involves the Wittig reaction between the C7 phosphonium salt and beta-ionone under alkaline conditions. This coupling is highly sensitive to reaction parameters, yet the patented method optimizes temperature and base concentration to favor the formation of the thermodynamically stable all-trans isomer. The use of mild bases such as sodium carbonate or potassium carbonate facilitates the deprotonation of the phosphonium salt to generate the ylide, which then attacks the carbonyl group of the ionone. This mechanism is crucial for achieving the reported 90% yield and high isomeric purity. By controlling the reaction environment meticulously, the process avoids the formation of excessive cis-isomers, a common pitfall in older technologies.
How to Synthesize Vitamin A Acetate Efficiently
Implementing this C13+C7 synthesis route requires careful attention to reaction stoichiometry and thermal management to maximize yield and safety. The process is designed to be scalable, moving from laboratory benchtop conditions to commercial production with minimal modification to the core chemistry. Operators must ensure strict control over the dropwise addition of reagents during the Grignard and acidolysis phases to prevent exothermic runaways. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for technical teams aiming to adopt this superior manufacturing protocol.
- React bromoethanol with acetone under acid catalysis to form the protected ketal intermediate.
- Perform an in-situ Grignard reaction between the ketal compound and C5 aldehyde, followed by acidolysis to yield the C7 alcohol.
- Convert the C7 alcohol into the corresponding phosphonium salt using triphenylphosphine and hydrochloric acid.
- Execute the final Wittig reaction between the C7 phosphonium salt and beta-ionone to obtain Vitamin A Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this C13+C7 methodology offers profound strategic benefits beyond mere technical superiority. The reduction in raw material complexity directly translates to a more resilient supply chain, as the process relies on commodity chemicals like acetone and bromoethanol rather than exotic, hard-to-source intermediates. This shift mitigates the risk of supply disruptions and price volatility often associated with specialized fine chemical precursors. Furthermore, the simplified process flow reduces the number of unit operations required, leading to significant reductions in energy consumption and labor costs per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of over 50 raw materials required by the legacy C14+C6 route results in a drastic simplification of the procurement portfolio and inventory management. By avoiding the use of hazardous double Grignard reagents, the facility saves on specialized safety equipment and containment infrastructure, leading to substantial capital expenditure avoidance. Additionally, the higher yield of 90% means less raw material is wasted per unit of output, directly improving the gross margin profile of the manufacturing operation without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the key building blocks, such as the C5 aldehyde and beta-ionone, are produced via established, high-volume industrial processes. The robustness of the C13+C7 route against feedstock fluctuations ensures consistent production schedules, reducing lead time for high-purity vitamin intermediates. This reliability is critical for downstream customers in the pharmaceutical and nutraceutical sectors who require just-in-time delivery to maintain their own production lines without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex vitamin intermediates, with reaction conditions that are easily managed in large-scale reactors. The reduction in solid waste, particularly triphenylphosphine oxide compared to C15+C5 routes, simplifies effluent treatment and lowers environmental compliance costs. This eco-friendly profile aligns with modern sustainability goals, making the manufacturer a more attractive partner for global corporations with strict ESG mandates.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and performance of this novel synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield, purity, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing facilities.
Q: How does the C13+C7 route improve upon traditional C14+C6 synthesis methods?
A: The C13+C7 route significantly simplifies the synthetic pathway compared to the C14+C6 method, which requires over 50 raw materials and involves hazardous double Grignard reagents. The new process reduces fixed investment and operational complexity while achieving yields up to 90%.
Q: What is the stereochemical purity of the Vitamin A Acetate produced?
A: This novel methodology ensures high stereocontrol, resulting in an all-trans Vitamin A content exceeding 85%. This high purity minimizes downstream separation costs and enhances the biological efficacy of the final nutritional product.
Q: Does this process generate significant triphenylphosphine oxide waste?
A: While triphenylphosphine is used to form the phosphonium salt, the overall process efficiency and yield improvements offset waste management concerns compared to older C15+C5 routes. The streamlined steps reduce the total volume of solid waste generated per kilogram of product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin A Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the C13+C7 Wittig strategy is pivotal for maintaining competitiveness in the global vitamins market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this high-yield technology is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical all-trans isomer content, guaranteeing that every batch meets the highest international standards for pharmaceutical and nutritional applications.
We invite you to collaborate with our technical procurement team to explore how this innovative process can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits specific to your volume requirements. We encourage potential partners to contact us immediately to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the confidence of backed-by-data technical excellence.
