Scalable Synthesis of Gemcitabine Intermediates via Novel Asymmetric Reformatsky Reaction
Scalable Synthesis of Gemcitabine Intermediates via Novel Asymmetric Reformatsky Reaction
The pharmaceutical industry continuously seeks robust and scalable pathways for producing high-value anticancer agents, and patent CN1229372C presents a significant breakthrough in the synthesis of Gemcitabine intermediates. This intellectual property details a novel synthetic process that leverages polymerized (R)-2,3-O-acetonide-glyceraldehyde to achieve superior diastereoselectivity in the critical Reformatsky reaction step. Unlike previous methodologies that struggled with low selectivity and complex purification requirements, this invention utilizes a controlled feeding strategy to favor the formation of the desired trans-product. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient pharmaceutical intermediate manufacturing, promising enhanced purity profiles and streamlined operations. The technology addresses the inherent instability of the starting aldehyde by embracing its polymeric form, turning a traditional liability into a strategic advantage for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of D-erythro-2'-deoxy-2',2'-difluoro-1-carbonyl ribose derivatives has been plagued by poor stereocontrol and cumbersome purification steps. As illustrated in early literature by L.W. Hertel, the substrate-controlled asymmetric Reformatsky reaction using monomeric aldehyde typically yielded a cis-to-trans product ratio of merely 1:3, necessitating labor-intensive column chromatography to isolate the chiral pure trans-product. 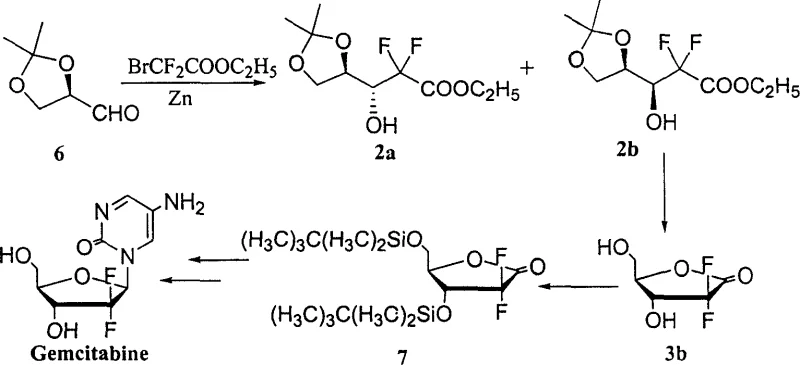 Furthermore, subsequent improvements by T.S. Chou, while eliminating chromatography, resulted in an overall yield of only 25% due to the lengthy synthetic sequence and persistent selectivity issues.
Furthermore, subsequent improvements by T.S. Chou, while eliminating chromatography, resulted in an overall yield of only 25% due to the lengthy synthetic sequence and persistent selectivity issues. 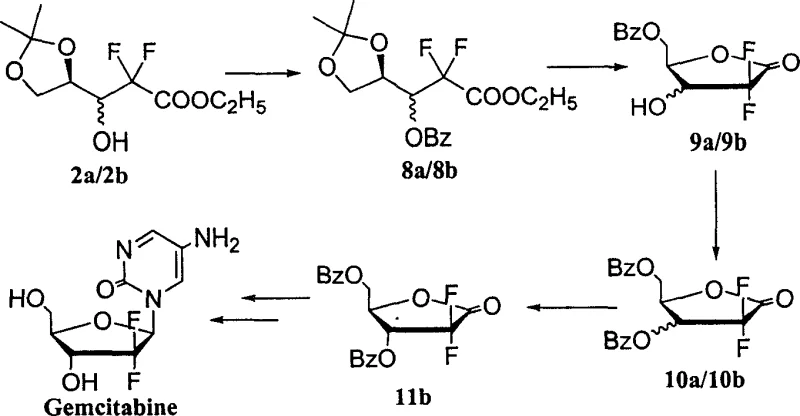 Other approaches attempted to boost selectivity using expensive reagents like iododifluoroacetate or Titanocene catalysts at cryogenic temperatures, which rendered them economically unviable for commercial API manufacturing. These conventional bottlenecks severely impacted supply chain reliability and inflated the cost of goods for this critical oncology building block.
Other approaches attempted to boost selectivity using expensive reagents like iododifluoroacetate or Titanocene catalysts at cryogenic temperatures, which rendered them economically unviable for commercial API manufacturing. These conventional bottlenecks severely impacted supply chain reliability and inflated the cost of goods for this critical oncology building block.
The Novel Approach
The patented process introduces a paradigm shift by directly utilizing polymerized (R)-2,3-O-acetonide-glyceraldehyde (where n=2~20) as the starting material, effectively bypassing the difficulties associated with monomer stabilization. By reacting this polymeric precursor with ethyl difluorohaloacetate in the presence of zinc and meticulously controlling the addition rate, the method achieves a remarkable diastereoselectivity with a cis-to-trans ratio ranging from 1:4 to 1:14. 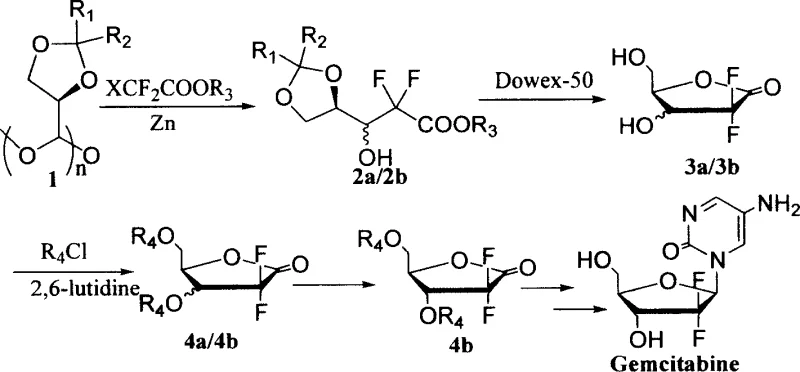 This high selectivity allows for the isolation of the desired trans-isomer through simple vacuum distillation and recrystallization, completely eliminating the need for column chromatography. The subsequent steps involve mild deprotection using Dowex-50 acidic resin followed by protection and recrystallization to yield the pure intermediate 4b with a total yield of 30-50% from compound 2. This streamlined approach not only simplifies the operational workflow but also significantly enhances the feasibility of commercial scale-up of complex pharmaceutical intermediates, making it an attractive option for reliable suppliers.
This high selectivity allows for the isolation of the desired trans-isomer through simple vacuum distillation and recrystallization, completely eliminating the need for column chromatography. The subsequent steps involve mild deprotection using Dowex-50 acidic resin followed by protection and recrystallization to yield the pure intermediate 4b with a total yield of 30-50% from compound 2. This streamlined approach not only simplifies the operational workflow but also significantly enhances the feasibility of commercial scale-up of complex pharmaceutical intermediates, making it an attractive option for reliable suppliers.
Mechanistic Insights into Zinc-Mediated Asymmetric Reformatsky Reaction
The core of this technological advancement lies in the nuanced manipulation of the Reformatsky reaction mechanism, specifically how the organozinc species interacts with the chiral aldehyde equivalent. In this system, zinc metal inserts into the carbon-halogen bond of ethyl difluorohaloacetate to generate a reactive organozinc enolate, which then attacks the carbonyl group of the glyceraldehyde derivative. The stereochemical outcome is heavily influenced by the transition state geometry, which is modulated by the concentration of the reactants and the physical state of the aldehyde source. By controlling the feeding speed of the reagents, the process maintains an optimal concentration of the organozinc intermediate, favoring the formation of the thermodynamically stable trans-transition state over the cis-configuration. This kinetic control is essential for minimizing the formation of the unwanted cis-isomer, thereby reducing the burden on downstream purification processes and ensuring a cleaner reaction profile.
Impurity control is further refined through the strategic use of acidic resin Dowex-50 for the deprotection step, which converts the acetonide protected mixture into the corresponding diol without inducing significant epimerization or degradation. The subsequent protection step with benzoyl chloride creates a crystalline derivative where the solubility differences between the cis and trans isomers are maximized. This allows for the effective removal of the minor cis-isomer impurity through a single recrystallization step from a dichloromethane and n-heptane solvent system. Such a robust impurity rejection mechanism is critical for meeting the stringent purity specifications required for high-purity pharmaceutical intermediates intended for final drug substance synthesis. The ability to achieve this level of purity without chromatographic intervention demonstrates a deep understanding of the physicochemical properties of the intermediates involved.
How to Synthesize Gemcitabine Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing this high-selectivity transformation in a laboratory or pilot plant setting. The procedure begins with the activation of zinc powder in anhydrous tetrahydrofuran, followed by the slow, controlled addition of the polymeric aldehyde and the difluoroester to maintain a gentle reflux. This careful management of reaction kinetics is the key determinant for achieving the superior 1:14 diastereomeric ratio described in the examples. Following the reaction, the workup involves standard aqueous quenching and extraction, followed by vacuum distillation to isolate the crude ester mixture. The subsequent chemical transformations are designed to be operationally simple, utilizing common reagents and avoiding exotic catalysts, which facilitates easy technology transfer to manufacturing sites focused on cost reduction in API manufacturing.
- Suspend zinc powder in anhydrous THF with iodine activation, then slowly add polymerized (R)-2,3-O-acetonide-glyceraldehyde and ethyl difluorobromoacetate while controlling the feeding rate to maintain reflux.
- Treat the resulting cis-trans mixture (2a/2b) with acidic Dowex-50 resin in a methanol-water solvent system at room temperature to remove the acetonide protecting group.
- React the deprotected sugar with benzoyl chloride and 2,6-lutidine, followed by recrystallization from dichloromethane and n-heptane to isolate pure trans-isomer 4b.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical elegance, directly impacting the bottom line and supply security. The elimination of column chromatography is perhaps the most significant economic driver, as chromatographic purification is notoriously solvent-intensive, time-consuming, and difficult to scale efficiently in large reactors. By replacing this step with vacuum distillation and recrystallization, the process drastically reduces solvent consumption and waste generation, leading to substantial cost savings in raw materials and waste disposal. Furthermore, the use of inexpensive and readily available reagents such as zinc powder and ethyl difluorobromoacetate ensures that the raw material costs remain low and stable, mitigating the risk of price volatility often associated with specialized catalysts.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by removing the need for expensive transition metal catalysts like Titanocene and avoiding the high operational costs of chromatographic purification. The reliance on simple unit operations such as distillation and crystallization allows for higher throughput and lower energy consumption per kilogram of product. Additionally, the improved selectivity reduces the amount of starting material wasted on unwanted isomers, effectively increasing the atom economy of the overall synthesis. These factors combine to create a highly cost-competitive manufacturing process suitable for the price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The synthetic route relies on commodity chemicals that are widely available from multiple global suppliers, reducing the risk of supply disruptions caused by single-source dependencies. The robustness of the reaction conditions, which tolerate the use of polymerized starting materials, ensures consistent batch-to-batch quality even with variations in raw material grades. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream drug manufacturers. Consequently, partners adopting this technology can offer greater assurance of supply continuity for this critical oncology intermediate.
- Scalability and Environmental Compliance: The simplified workflow, characterized by fewer unit operations and the absence of complex purification techniques, makes this process inherently easier to scale from kilogram to multi-ton production levels. The reduction in solvent usage and the avoidance of heavy metal catalysts align well with modern green chemistry principles and environmental regulations, simplifying the permitting and compliance process for manufacturing facilities. This environmental friendliness not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturing organization, a key consideration for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway for Gemcitabine intermediates. These insights are derived directly from the experimental data and comparative analysis presented in patent CN1229372C, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or for sourcing strategies. Engaging with these technical specifics allows stakeholders to fully appreciate the operational advantages and quality improvements offered by this innovative approach.
Q: How does the new process improve stereoselectivity compared to conventional methods?
A: By utilizing polymerized (R)-2,3-O-acetonide-glyceraldehyde and strictly controlling the reagent feeding speed, the process achieves a diastereomeric ratio of up to 1:14 (cis:trans), significantly higher than the 1:3 ratio observed in traditional monomer-based routes.
Q: Is column chromatography required for purification in this synthetic route?
A: No, the process eliminates the need for column chromatography entirely. The crude product is purified through vacuum distillation and subsequent recrystallization, which drastically simplifies the workflow and reduces solvent consumption for industrial applications.
Q: What are the key cost-saving factors in this manufacturing method?
A: The method uses inexpensive reagents like zinc powder and ethyl difluorobromoacetate, avoids expensive catalysts like Titanocene, and removes costly chromatographic purification steps, leading to substantial operational cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value anticancer intermediates like those described in this patent. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries are translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the quality required for final drug substance manufacturing. Our capability to implement advanced selective reactions, such as the zinc-mediated Reformatsky reaction, positions us as a strategic partner for your long-term supply needs.
We invite you to contact our technical procurement team to discuss how we can support your project with a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in process optimization, we can help you secure a stable supply of high-quality intermediates while optimizing your overall manufacturing costs. Please reach out to request specific COA data and route feasibility assessments to see how our capabilities align with your development goals. Partnering with us ensures access to reliable supply chains and cutting-edge chemical manufacturing solutions.
