Advanced Catalytic Synthesis and Resolution of Chiral 6-Fluoro-Benzopyran Intermediates for Hypertension Therapy
The pharmaceutical industry continuously seeks robust and scalable pathways for producing high-value chiral intermediates, particularly for cardiovascular therapies. Patent CN1629154A introduces a significant technological advancement in the synthesis of optical enantiomers of 6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid, a critical precursor for beta-adrenergic antagonist antihypertensive drugs. This innovation addresses long-standing challenges in stereoselective synthesis by replacing hazardous reagents with catalytic systems and introducing a cost-effective resolution strategy. By leveraging palladium-catalyzed hydrogenation and alpha-phenylethylamine resolution, the disclosed method achieves high optical purity while mitigating the environmental burdens associated with traditional chlorinating agents. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these benzopyran derivatives relied heavily on European Patent 0334429, which utilized thionyl chloride (SOCl2) to convert carboxylic acids into acid chlorides prior to resolution. This approach presents severe drawbacks for modern manufacturing, primarily due to the generation of corrosive sulfur dioxide and hydrochloric acid gases, necessitating expensive scrubbing systems and specialized corrosion-resistant equipment. Furthermore, the traditional resolution employed dehydroabietylamine, a bulky and costly chiral auxiliary that is difficult to recover economically. The subsequent hydrolysis of the resulting amide intermediates often required harsh acidic or basic conditions, which inadvertently promoted racemization, thereby compromising the optical purity of the final active pharmaceutical ingredient. These factors collectively inflated production costs and complicated regulatory compliance regarding waste disposal and worker safety.
The Novel Approach
In stark contrast, the methodology outlined in CN1629154A establishes a streamlined, environmentally benign route that bypasses the need for acid chloride formation entirely. The process initiates with the acylation of p-fluoroanisole, followed by a efficient cyclization and a pivotal catalytic hydrogenation step using 10wt% Pd/C. This hydrogenation proceeds smoothly at ambient temperature and pressure, drastically reducing energy consumption compared to high-pressure alternatives. The subsequent resolution utilizes alpha-phenylethylamine, a commercially abundant and inexpensive chiral base, which forms diastereomeric salts that are easily separated by crystallization. 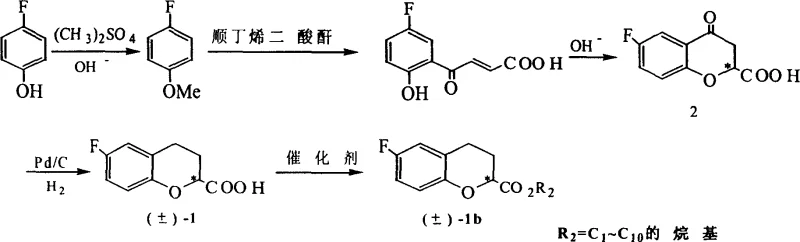 This novel approach not only eliminates the use of SOCl2 but also ensures that the resolving agent can be recycled after acid decomposition, creating a closed-loop system that significantly lowers the cost reduction in API manufacturing while maintaining exceptional stereochemical integrity.
This novel approach not only eliminates the use of SOCl2 but also ensures that the resolving agent can be recycled after acid decomposition, creating a closed-loop system that significantly lowers the cost reduction in API manufacturing while maintaining exceptional stereochemical integrity.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Chiral Resolution
The core of this synthetic strategy lies in the selective reduction of the unsaturated benzopyran ring system without affecting other sensitive functional groups. The mechanism involves the adsorption of the substrate onto the surface of the palladium catalyst, where molecular hydrogen is dissociated into atomic hydrogen. This species then adds across the carbon-carbon double bond in a syn-addition manner, saturating the 3,4-position of the pyran ring to yield the racemic dihydro-acid. The choice of 10wt% Pd/C is critical, as it offers a balance between activity and selectivity, preventing over-reduction of the aromatic ring or the carbonyl group. Following hydrogenation, the resolution mechanism relies on the formation of diastereomeric salts between the racemic acid and the optically pure alpha-phenylethylamine. Due to the differences in crystal lattice energy and solubility between the (R)-acid/(S)-amine and (S)-acid/(S)-amine salts, one isomer preferentially crystallizes out of the solution, allowing for physical separation.
Controlling impurities during this process is paramount for meeting stringent pharmacopeial standards. The mild conditions of the hydrogenation step minimize the formation of polymeric byproducts or ring-opened degradation products that are common in harsher chemical reductions. Furthermore, the recrystallization of the diastereomeric salts serves as a powerful purification step, effectively excluding structurally related impurities that do not fit into the chiral crystal lattice. 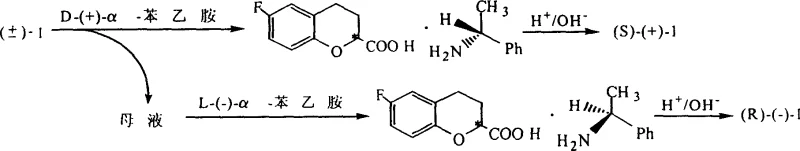 The final acidification step to liberate the free acid is conducted under controlled pH conditions to prevent epimerization at the chiral center. This meticulous control over reaction parameters ensures that the final high-purity OLED material or pharmaceutical intermediate retains its specific rotation values, such as [α]D = +14.2° for the (S)-enantiomer, confirming the efficacy of the resolution protocol.
The final acidification step to liberate the free acid is conducted under controlled pH conditions to prevent epimerization at the chiral center. This meticulous control over reaction parameters ensures that the final high-purity OLED material or pharmaceutical intermediate retains its specific rotation values, such as [α]D = +14.2° for the (S)-enantiomer, confirming the efficacy of the resolution protocol.
How to Synthesize 6-Fluoro-Benzopyran Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the methylation of p-fluorophenol to protect the phenolic hydroxyl group, followed by a Lewis acid-catalyzed acylation to introduce the side chain necessary for ring closure. The cyclization step is carefully managed using alkaline conditions to form the chromone skeleton, which is then subjected to the aforementioned catalytic hydrogenation. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results with high fidelity.
- Perform Friedel-Crafts acylation on p-fluoroanisole using maleic anhydride or acetylating agents with Lewis acid catalysts to form the ketone intermediate.
- Execute cyclization under alkaline or acidic conditions to construct the benzopyran ring system, followed by Pd/C catalytic hydrogenation to reduce the double bond.
- Conduct chemical resolution of the racemic acid using optically active alpha-phenylethylamine to separate the (R) and (S) enantiomers via diastereomeric salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of thionyl chloride removes a major bottleneck related to hazardous material handling and storage, simplifying logistics and reducing insurance premiums associated with toxic reagents. Moreover, the use of alpha-phenylethylamine as a resolving agent represents a substantial cost saving, as it is significantly cheaper than dehydroabietylamine and, crucially, can be recovered and reused multiple times without loss of optical activity. This recyclability transforms a consumable cost into a capital-efficient loop, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by removing the need for expensive chiral auxiliaries and corrosive chlorinating agents. By utilizing catalytic hydrogenation with recoverable palladium on carbon and a low-cost amine resolver, the overall material cost per kilogram of the active intermediate is drastically lowered. Additionally, the mild reaction conditions reduce energy expenditures for heating and cooling, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as p-fluorophenol and maleic anhydride, are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to raw material shortages. The robustness of the catalytic steps ensures consistent batch-to-batch quality, minimizing the need for rework or rejection of off-spec material, which stabilizes delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The absence of sulfur-based waste streams simplifies effluent treatment, making it easier to meet increasingly strict environmental regulations in various jurisdictions. The process is inherently scalable from gram to multi-ton quantities because the unit operations involved, such as filtration, crystallization, and hydrogenation, are well-understood and easily engineered for commercial scale-up of complex pharmaceutical intermediates without requiring exotic reactor designs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational requirements.
Q: What are the primary limitations of conventional synthesis methods for 6-fluoro-benzopyran derivatives?
A: Conventional methods often rely on hazardous reagents like thionyl chloride (SOCl2) which generate significant environmental pollution and require complex waste treatment. Additionally, traditional resolution agents like dehydroabietylamine are expensive and difficult to recover, leading to higher production costs and potential racemization during harsh hydrolysis steps.
Q: How does the novel catalytic hydrogenation process improve product purity?
A: The novel process utilizes 10wt% Pd/C catalytic hydrogenation under mild temperature and pressure conditions. This approach minimizes side reactions and degradation compared to harsher chemical reduction methods, ensuring high chemical purity before the resolution step even begins.
Q: Is the chiral resolving agent used in this process recyclable?
A: Yes, the process employs alpha-phenylethylamine as a resolving agent. After the diastereomeric salts are decomposed with acid to release the optical pure acid, the resolving agent can be recovered and recycled, significantly reducing material costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Fluoro-Benzopyran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antihypertensive medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
