Revolutionizing Vitamin H Production: A Deep Dive into Efficient Dibenzyl Biotin Debenzylation
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to optimize the production of critical vitamins and intermediates. Patent CN103788112A introduces a transformative approach to the synthesis of Biotin (Vitamin H), specifically addressing the bottlenecks associated with the debenzylation of dibenzyl biotin. This technology shifts the paradigm from static, high-temperature batch processing to a dynamic, continuous removal system utilizing hydrobromic acid assisted by solid dispersants. By integrating azeotropic distillation principles directly into the reaction vessel, the process effectively drives the equilibrium forward while simultaneously mitigating the formation of detrimental ring-opened byproducts. For stakeholders in the pharmaceutical intermediates sector, this represents a significant leap forward in process intensification, offering a pathway to higher purity profiles and drastically reduced operational expenditures without compromising on safety or environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of biotin from its dibenzyl precursor has been plagued by inefficiencies inherent to traditional acidolysis methods. Conventional techniques typically involve heating dibenzyl biotin with concentrated hydrobromic acid or sulfuric acid at extreme temperatures exceeding 145°C for prolonged periods ranging from 24 to 36 hours. These harsh thermal conditions inevitably lead to severe degradation of the sensitive ureido ring structure, resulting in ring-opening rates that can surpass 60%. Consequently, manufacturers are forced to implement complex and costly downstream remediation steps, such as phosgenation, to reclose the rings, which generates substantial quantities of hazardous wastewater and complicates the recovery of valuable solvents. Furthermore, the accumulation of oily byproducts like benzyl bromide within the reaction matrix often creates a barrier that prevents the reactant from fully contacting the acid catalyst, thereby stalling reaction kinetics and necessitating even longer reaction times to achieve completion.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages a sophisticated understanding of phase behavior and reaction kinetics to overcome these historical hurdles. By introducing a solid dispersant, such as silica gel, into the hydrobromic acid medium, the process ensures that the dibenzyl biotin remains finely dispersed rather than aggregating into an impermeable oil layer. Coupled with rapid stirring and controlled heating to a moderate range of 110°C to 130°C, the system facilitates the immediate vaporization and removal of the benzyl bromide byproduct via a condenser and phase separator. This continuous extraction of the byproduct not only adheres to Le Chatelier's principle to drive the reaction to completion but also prevents the byproduct from interfering with the reaction mixture. The result is a dramatic reduction in reaction time to merely 2 to 5 hours, alongside a significant suppression of ring-opening side reactions, ultimately delivering a crude product with superior purity and a theoretical yield approaching 87%.
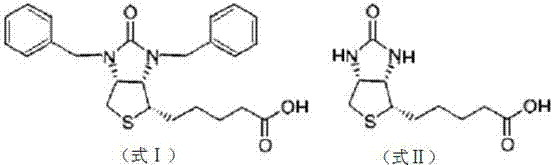
Mechanistic Insights into Hydrobromic Acid Debenzylation with Dispersant Assistance
The core innovation of this synthetic route lies in the synergistic interaction between the dispersant agent and the azeotropic distillation setup. In standard liquid-liquid acidolysis, the organic substrate often separates from the aqueous acid phase, creating a diffusion-limited environment where the reaction only occurs at the interface. The addition of a porous solid dispersant fundamentally alters this dynamic by adsorbing the organic reactant and distributing it throughout the acidic medium, thereby maximizing the surface area available for protonation and nucleophilic attack. This physical modification ensures that the cleavage of the benzyl groups proceeds uniformly and rapidly across the entire bulk of the reaction mixture, eliminating hot spots that typically lead to localized overheating and structural degradation of the biotin skeleton.
Furthermore, the management of impurities is handled with exceptional precision through the physical separation of phases during the distillation step. As the reaction progresses, the generated benzyl bromide co-distills with hydrobromic acid vapors. Upon condensation in the receiving trap, the immiscible benzyl bromide settles as a distinct lower layer, while the hydrobromic acid remains as the upper layer and is automatically returned to the reaction flask. This elegant engineering solution ensures that the concentration of the acid catalyst remains constant throughout the process, while the concentration of the inhibitory byproduct in the reactor remains near zero. This mechanism effectively shuts down the reverse reaction and minimizes secondary interactions between the byproduct and the sensitive biotin intermediate, ensuring that the final crystalline product requires minimal purification beyond simple decolorization and recrystallization.
How to Synthesize Biotin Efficiently
The implementation of this advanced debenzylation protocol requires precise control over material ratios and thermal gradients to maximize the efficiency of the azeotropic removal. Operators must carefully balance the mass ratio of dibenzyl biotin to hydrobromic acid, typically maintaining a range between 1:10 and 1:15, while ensuring the acid concentration stays within the optimal 38% to 42% window. The detailed standardized synthesis steps, including specific stirring speeds, vacuum levels for solvent recovery, and precise pH adjustments for crystallization, are outlined in the structured guide below to ensure reproducible results in a GMP-compliant environment.
- Disperse dibenzyl biotin in concentrated hydrobromic acid with a solid dispersant agent under rapid stirring to prevent agglomeration.
- Heat the mixture to 110-130°C to initiate azeotropic distillation, continuously removing benzyl bromide byproduct while recycling hydrobromic acid back to the reactor.
- Cool the reaction residue, dissolve in water, adjust pH, decolorize with activated carbon, and crystallize to obtain pure biotin, while recycling mother liquor for further recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into tangible economic and logistical benefits that strengthen the overall value proposition of biotin manufacturing. The shift from a batch-and-hold process to a continuous removal system drastically reduces the energy consumption per kilogram of product, as the reaction temperature is lowered and the duration is cut by nearly an order of magnitude. Moreover, the ability to recover and purify benzyl bromide transforms a traditional waste liability into a potential revenue stream, offsetting raw material costs. The elimination of massive volumes of saline wastewater, which is characteristic of sulfuric acid neutralization processes, significantly lowers the burden on effluent treatment plants and reduces the regulatory risks associated with environmental compliance.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the high recovery rate of hydrobromic acid, which circulates within the system rather than being consumed or neutralized. By avoiding the use of expensive organic solvents for extraction and minimizing the need for extensive downstream ring-closing repairs, the overall cost of goods sold is substantially decreased. The qualitative reduction in energy usage due to shorter reaction cycles and lower operating temperatures further contributes to a leaner manufacturing cost structure, allowing for more competitive pricing in the global market for vitamin intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reduction in cycle time from days to hours dramatically increases the throughput capacity of existing reactor infrastructure. This agility allows manufacturers to respond more rapidly to fluctuations in market demand without the need for capital-intensive expansion of plant footprint. Additionally, the robustness of the process against variations in raw material quality, ensured by the dispersant mechanism, leads to more consistent batch-to-batch performance, reducing the risk of production delays caused by off-specification batches and ensuring a steady flow of high-purity product to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard distillation and crystallization unit operations that are well-understood in the fine chemical industry. The absence of complex solvent swaps and the minimization of hazardous waste generation align perfectly with modern green chemistry principles and stringent environmental regulations. This facilitates smoother permitting processes for new facilities and ensures long-term operational continuity, as the technology is future-proofed against tightening restrictions on industrial wastewater discharge and volatile organic compound emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this debenzylation technology, derived directly from the patent specifications and comparative data. These insights are intended to clarify the operational advantages and help technical teams evaluate the feasibility of integrating this route into their existing production lines for vitamin H and related derivatives.
Q: How does this new debenzylation method improve yield compared to traditional sulfuric acid methods?
A: Traditional methods often suffer from high ring-opening rates (up to 60%) due to harsh conditions. This patented process utilizes continuous removal of benzyl bromide via distillation and a dispersant to maintain homogeneity, significantly reducing side reactions and improving overall yield by approximately 5% through mother liquor recycling.
Q: What are the environmental benefits of using hydrobromic acid with azeotropic distillation?
A: Unlike sulfuric acid methods that generate massive amounts of saline wastewater requiring neutralization, this process allows for the recovery and reuse of hydrobromic acid. Additionally, the byproduct benzyl bromide is separated cleanly and can be purified for sale, transforming a waste stream into a value-added product.
Q: Is this process suitable for large-scale commercial production of biotin intermediates?
A: Yes, the process is explicitly designed for industrial suitability. It reduces reaction time from 24-36 hours to just 2-5 hours, operates at lower temperatures (110-130°C vs >145°C), and eliminates the need for complex organic solvent extractions, making it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature, pressure, and stoichiometry required for this debenzylation process is maintained with absolute precision. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of biotin intermediate meets the exacting standards required by the global pharmaceutical and nutraceutical industries, providing you with a secure and high-quality supply chain foundation.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can optimize your bottom line. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
