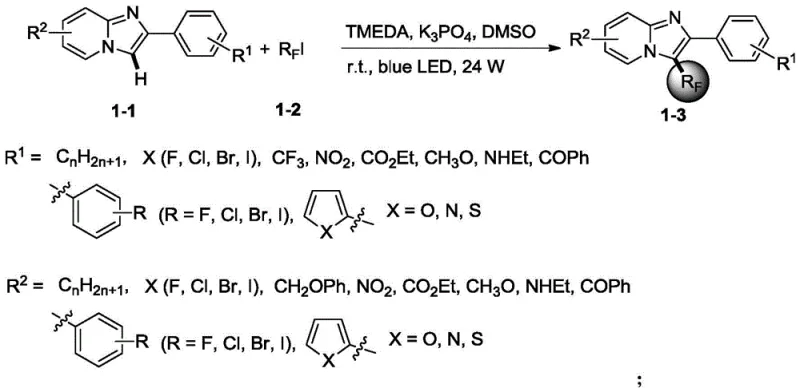
Advanced Photocatalytic Synthesis of Fluorinated Imidazo[1,2-a]pyridines for Commercial Scale-up
The pharmaceutical industry continuously seeks robust methodologies for incorporating fluorine atoms into heterocyclic scaffolds, as these modifications often drastically enhance metabolic stability and bioavailability. Patent CN112851670B discloses a groundbreaking simple synthetic method for 3-polyfluoroalkylated substituted imidazo[1,2-a]pyridine, addressing a significant gap in current medicinal chemistry toolkits. This technology leverages a photocatalytic strategy to induce free radical generation from polyfluoroalkyl iodides, enabling direct functionalization under remarkably mild conditions. Unlike traditional approaches that rely on harsh thermal energy or expensive transition metal catalysts, this invention utilizes visible light irradiation to drive the reaction efficiently. The process demonstrates exceptional versatility across a broad range of substrates, maintaining high yields while adhering to green chemistry principles. For R&D teams focusing on novel drug candidates, this patent represents a pivotal advancement in constructing fluorinated nitrogen-containing heterocycles. The ability to access these structures through a sustainable pathway aligns perfectly with modern regulatory demands for environmentally responsible manufacturing processes.
![General formula of imidazo[1,2-a]pyridine derivatives showing variable R groups for diverse chemical space exploration](/insights/img/fluoroalkylated-imidazopyridine-photocatalysis-pharma-supplier-20260313130221-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of polyfluoroalkyl groups into imidazo[1,2-a]pyridine systems has been fraught with significant technical challenges and economic inefficiencies. Conventional synthetic routes often necessitate the use of stoichiometric amounts of expensive transition metal catalysts, which not only inflate raw material costs but also introduce complex downstream purification burdens. These metal residues must be rigorously removed to meet stringent pharmaceutical standards, requiring additional processing steps that extend production timelines and reduce overall throughput. Furthermore, traditional thermal methods frequently demand high temperatures and pressures, creating safety hazards and limiting the compatibility with sensitive functional groups present in complex drug molecules. The lack of regioselectivity in many older protocols often leads to mixtures of isomers, complicating the isolation of the desired 3-substituted product and resulting in substantial material loss. These cumulative factors create a bottleneck for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing, as the overall cost of goods sold remains prohibitively high for many potential applications.
The Novel Approach
The methodology outlined in CN112851670B offers a transformative solution by employing a metal-free photocatalytic system that operates at ambient temperatures. By utilizing common organic amines as inducers and simple inorganic salts as additives, this route eliminates the dependency on precious metals entirely. The reaction proceeds smoothly in polar aprotic solvents like DMSO or DMF under blue LED irradiation, providing a safe and energy-efficient alternative to thermal heating. This shift in paradigm allows for the direct activation of polyfluoroalkyl iodides to generate radicals that selectively attack the heterocyclic core. The mildness of the conditions preserves sensitive substituents, thereby expanding the scope of accessible chemical space for medicinal chemists. From a supply chain perspective, the simplicity of the reagent profile enhances supply continuity, as the required materials are commodity chemicals rather than specialized catalysts. This novel approach not only streamlines the synthesis but also significantly reduces the environmental footprint, making it an attractive option for companies aiming to improve their sustainability metrics while maintaining high production standards.
Mechanistic Insights into Photocatalytic Polyfluoroalkylation
At the heart of this innovation lies a sophisticated radical mechanism initiated by the interaction between the inducer and the polyfluoroalkyl iodide under light irradiation. Upon exposure to LED light, the inducer facilitates the formation of an electron donor-acceptor complex or directly transfers energy to cleave the carbon-iodine bond in the polyfluoroalkyl iodide. This homolytic cleavage generates a highly reactive polyfluoroalkyl radical species, which is the key active intermediate in the transformation. The radical then selectively attacks the electron-rich 3-position of the imidazo[1,2-a]pyridine ring, forming a new carbon-carbon bond with high precision. The presence of the additive, such as potassium phosphate, plays a crucial role in neutralizing the hydrogen iodide byproduct, driving the equilibrium towards product formation and preventing acid-mediated decomposition of the substrate. This mechanistic pathway ensures that the reaction proceeds with minimal side reactions, resulting in a clean impurity profile that is highly desirable for regulatory filings. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as light intensity and reagent stoichiometry, to optimize yields for specific derivatives.
Controlling the impurity profile is paramount for any pharmaceutical intermediate, and this photocatalytic route offers inherent advantages in this regard. The radical nature of the reaction, combined with the specific electronic properties of the imidazo[1,2-a]pyridine scaffold, ensures high regioselectivity, effectively suppressing the formation of unwanted isomers at other positions on the ring. Additionally, the absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as homocoupling or over-alkylation, which are common pitfalls in traditional cross-coupling methodologies. The mild reaction temperature further prevents thermal degradation of the product or starting materials, preserving the integrity of complex molecular architectures. For quality control teams, this translates to a more straightforward purification process, often requiring only standard silica gel chromatography to achieve high-purity pharmaceutical intermediates. The consistency of the reaction across different substrates, as evidenced by the broad scope of examples in the patent, suggests a robust process that can be reliably transferred from the laboratory to pilot plant scales without significant deviation in quality attributes.
How to Synthesize 3-Polyfluoroalkylated Imidazo[1,2-a]pyridine Efficiently
Implementing this synthesis requires careful attention to the reaction environment to maximize the efficiency of the photocatalytic cycle. The process begins by dissolving the imidazo[1,2-a]pyridine derivative and the polyfluoroalkyl iodide in a suitable solvent under an inert nitrogen atmosphere to prevent oxygen quenching of the radical species. The addition of the inducer and additive must be precisely controlled to maintain the optimal molar ratios specified in the patent, ensuring complete conversion of the starting materials. Once the mixture is prepared, it is subjected to continuous irradiation using a 24-70W LED lamp, with the reaction temperature maintained between 20-45°C to balance reaction rate and selectivity. Detailed standardized synthesis steps see the guide below.
- Dissolve imidazo[1,2-a]pyridine derivative and polyfluoroalkyl iodide in DMSO or DMF under nitrogen.
- Add inducer such as TMEDA and additive like potassium phosphate to the mixture.
- Irradiate with 24-70W LED or UV lamp at 20-45°C for 24-96 hours, then purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology presents a compelling value proposition centered around cost efficiency and operational reliability. The elimination of expensive transition metal catalysts directly translates to substantial cost savings in raw material procurement, removing the volatility associated with precious metal markets. Furthermore, the simplified workup procedure reduces the consumption of purification materials and solvents, lowering the overall waste disposal costs and environmental compliance burdens. The use of commodity reagents and standard lighting equipment enhances supply chain reliability, as these items are readily available from multiple vendors, mitigating the risk of single-source bottlenecks. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands. The scalability of the process is another critical advantage, as the reaction conditions are easily adaptable to larger photoreactors without requiring specialized high-pressure equipment.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of transition metal catalysts, which are often the most expensive component in traditional cross-coupling reactions. By substituting these with inexpensive organic amines and inorganic salts, the direct material cost is drastically simplified, leading to significant margin improvements. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature thermal processes, further contributing to lower utility costs per kilogram of product. The high yields reported in the patent examples indicate efficient atom economy, minimizing the loss of valuable starting materials and maximizing the output from each batch. These factors combine to create a highly competitive cost structure that supports long-term commercial viability for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available commodity chemicals rather than specialized or proprietary reagents. Solvents like DMSO and DMF, along with additives like potassium phosphate, are standard inventory items for most chemical manufacturers, ensuring consistent availability even during market fluctuations. The equipment requirements are also minimal, utilizing standard LED lighting systems that are durable and easy to replace, reducing downtime risks associated with equipment failure. This accessibility allows for flexible sourcing strategies, enabling procurement teams to negotiate better terms with multiple suppliers and avoid dependency on a single vendor. Consequently, the continuity of supply for critical pharmaceutical intermediates is greatly enhanced, supporting uninterrupted production schedules for downstream drug manufacturing.
- Scalability and Environmental Compliance: Scaling this process to commercial levels is facilitated by the inherent safety of the mild reaction conditions, which do not require high-pressure vessels or extreme temperatures. The environmental profile is markedly improved due to the absence of heavy metal waste, simplifying effluent treatment and reducing the regulatory burden associated with hazardous waste disposal. The use of light as a traceless reagent aligns with green chemistry principles, appealing to stakeholders focused on sustainability and corporate social responsibility goals. As production volumes increase, the efficiency of the photocatalytic system can be maintained through optimized reactor design, ensuring that the benefits observed at the laboratory scale are preserved in ton-scale manufacturing. This seamless scalability supports the commercial scale-up of complex fluorinated heterocycles, meeting the growing demand for these motifs in modern therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology. Understanding these aspects is crucial for making informed decisions about process adoption and supply chain integration. The information below highlights the key differentiators of this method compared to existing alternatives.
Q: What are the advantages of this photocatalytic method over traditional fluorination?
A: This method operates under mild conditions (20-45°C) without expensive transition metal catalysts, significantly reducing purification costs and environmental impact compared to harsh thermal methods.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the use of common solvents like DMSO and scalable LED light sources facilitates commercial scale-up of complex fluorinated heterocycles with high yield consistency.
Q: How does this route impact the impurity profile of the final product?
A: The radical mechanism ensures high regioselectivity at the 3-position, minimizing side reactions and simplifying the removal of impurities for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Polyfluoroalkylated Imidazo[1,2-a]pyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into industrial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of photocatalytic processes, ensuring that the transition from patent to plant is executed with precision and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-polyfluoroalkylated imidazo[1,2-a]pyridine meets the exacting standards required by global pharmaceutical clients. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical fluorinated intermediates. By leveraging our infrastructure, clients can accelerate their development timelines and bring life-saving medications to market faster.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this metal-free methodology for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Partnering with us ensures access to cutting-edge technology combined with reliable manufacturing capabilities, positioning your projects for success in a competitive market landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
