Advanced Chiral Copper Catalysis for High-Purity Optically Active Indole Compounds
Introduction to Breakthrough Catalytic Technology
The field of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical development, particularly in the construction of complex chiral scaffolds. Patent CN114160206B introduces a transformative approach to synthesizing optically active indole compounds, specifically targeting the challenging C3 position functionalization. This technology leverages a novel chiral copper-based catalyst system that enables the direct asymmetric alkenylation of 3-alkenyl indole compounds with isatin-like substrates. The significance of this innovation lies in its ability to produce 3-alkenyl-3-substituted oxindole compounds with exceptional stereochemical control, achieving enantioselectivity levels exceeding 91% ee and diastereoselectivity ratios greater than 20:1. For R&D directors and process chemists, this represents a critical advancement in accessing high-value structural motifs found in bioactive molecules such as spirotryprostatin B alkaloids and various receptor antagonists. The robustness of this method ensures that the resulting intermediates meet the stringent purity specifications required for downstream drug development, positioning it as a vital tool for reliable pharmaceutical intermediate supplier networks seeking to enhance their portfolio of chiral building blocks.
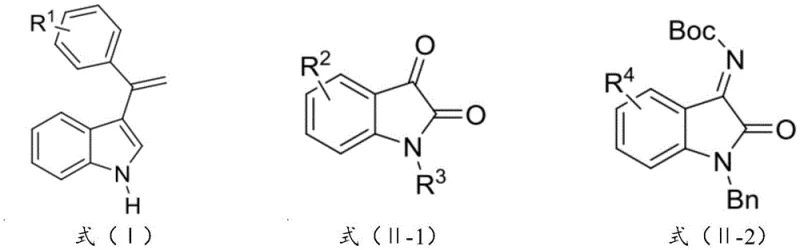
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric alkenylation of isatin and its analogues has presented significant synthetic hurdles due to the poor reactivity of bare alkenyl groups and the difficulty in controlling stereochemistry at the quaternary carbon center. Prior art, such as the work by Zhou's group in 2010 utilizing cinchona alkaloids or Zhao's group in 2016 employing chiral CoI2-bisphosphine complexes, often necessitated harsh reaction conditions, including elevated temperatures up to 70°C, which can compromise sensitive functional groups. Furthermore, these conventional methods frequently suffered from limited substrate scope and inconsistent enantioselectivity, making them less ideal for the cost reduction in API manufacturing where reproducibility is paramount. The reliance on expensive or difficult-to-handle catalysts in traditional Lewis acid-mediated pathways also introduces complexities in purification and waste management, creating bottlenecks in the supply chain for high-purity indole derivatives. These limitations underscore the industry's need for a more efficient, mild, and highly selective catalytic system that can operate effectively under ambient conditions without sacrificing yield or optical purity.
The Novel Approach
The methodology disclosed in CN114160206B overcomes these historical constraints through the deployment of a specifically designed chiral copper-based catalyst, designated as compounds of formula (C1) and/or (C2). This novel approach facilitates the direct asymmetric olefination reaction under remarkably mild conditions, typically between 0°C and 25°C, thereby preserving the integrity of thermally labile substituents. By utilizing 3-alkenyl indole compounds as nucleophiles against isatin-like electrophiles, the process achieves product yields exceeding 81% with purity levels reaching above 99.9%. This represents a substantial leap forward in process efficiency, eliminating the need for extreme thermal inputs and reducing the energy footprint of the synthesis. The versatility of this system allows for the accommodation of various substituents on both the indole and isatin frameworks, including alkyl, halogen, and alkoxy groups, ensuring broad applicability across different medicinal chemistry programs. For procurement managers, this translates to a more streamlined production process that minimizes raw material waste and maximizes the output of valuable chiral intermediates, directly supporting strategic goals for supply chain reliability and operational excellence.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Alkenylation
The core of this technological breakthrough resides in the unique architecture of the chiral copper catalyst, which creates a highly defined stereochemical environment around the reactive center. The catalyst is formed in situ or pre-prepared by coordinating a divalent copper salt with a specialized Schiff base ligand derived from L-phenylalanine and salicylaldehyde derivatives. This coordination generates a rigid chiral pocket that effectively discriminates between the prochiral faces of the incoming substrates during the bond-forming event. The nitrogen and oxygen atoms of the ligand coordinate with the copper center, stabilizing the transition state and directing the approach of the 3-alkenyl indole to the isatin carbonyl or imine group with high precision. This precise spatial arrangement is crucial for suppressing the formation of unwanted diastereomers and enantiomers, thereby driving the reaction towards the desired optical isomer with >91% ee. Understanding this mechanistic nuance is essential for R&D teams aiming to optimize reaction parameters or adapt the chemistry to novel substrate classes, as it highlights the importance of ligand sterics and electronics in governing catalytic performance.
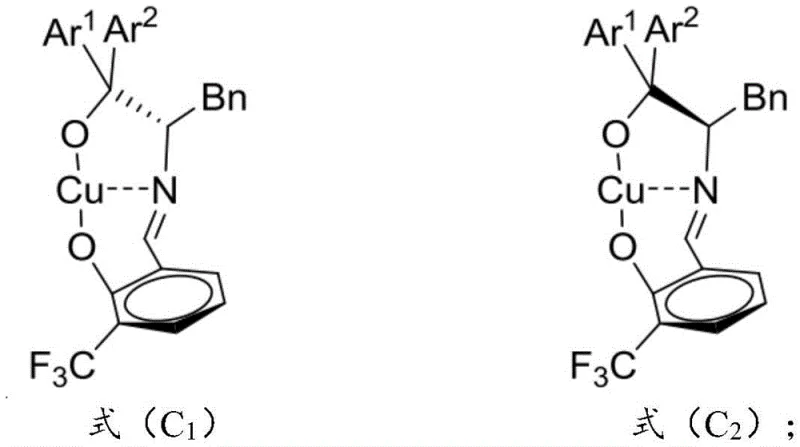
Furthermore, the impurity profile of the final product is tightly controlled by the high diastereoselectivity (dr > 20:1) inherent to this catalytic system. In conventional syntheses, the generation of multiple stereoisomers often necessitates costly and yield-loss-inducing separation steps, such as preparative chiral HPLC or repeated recrystallizations. However, the superior selectivity of the copper catalyst described in this patent minimizes the formation of these impurities at the source, simplifying the downstream purification workflow. The reaction mechanism likely involves a concerted addition pathway where the copper-ligand complex activates the nucleophile while simultaneously coordinating the electrophile, ensuring that the carbon-carbon bond formation occurs with strict stereochemical fidelity. This level of control not only enhances the overall quality of the pharmaceutical intermediate but also reduces the burden on quality control laboratories, allowing for faster batch release times. For technical leaders, this mechanistic robustness provides confidence in the scalability of the process, as the fundamental chemical drivers of selectivity remain consistent regardless of batch size.
How to Synthesize Optically Active Indole Compounds Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins with the preparation of the chiral ligand, followed by the formation of the active copper catalyst complex, and finally the coupling reaction between the indole and isatin components. Detailed protocols emphasize the use of common solvents like ethanol and mild bases such as N-ethylmorpholine, which are readily available and easy to handle on a large scale. The reaction proceeds efficiently at room temperature or slightly below, requiring only standard stirring and monitoring via TLC or HPLC. This operational simplicity is a key factor in facilitating the commercial scale-up of complex pharmaceutical intermediates, as it reduces the need for specialized high-pressure or cryogenic reactors. By following the optimized molar ratios and concentration guidelines provided in the patent, manufacturers can consistently achieve high yields and optical purity, ensuring a reliable supply of critical drug precursors.
- Prepare the chiral ligand by reacting L-phenylalanine derivatives with salicylaldehyde derivatives to form Schiff base ligands (L1/L2).
- Form the chiral copper-based catalyst complex by mixing a divalent copper salt, the prepared ligand, and a nitrogen-containing organic base in a solvent like ethanol at 0-25°C.
- React 3-alkenyl indole compounds with isatin-like compounds in the presence of the catalyst complex to obtain optically active 3-alkenyl-3-substituted oxindoles with high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers profound benefits for procurement and supply chain operations, primarily driven by process intensification and resource efficiency. The ability to conduct reactions under mild conditions significantly lowers energy consumption compared to traditional high-temperature methods, contributing to substantial cost savings in utility expenditures. Moreover, the high atom economy and selectivity of the reaction reduce the volume of waste solvents and byproducts generated, simplifying waste treatment protocols and aligning with increasingly stringent environmental compliance standards. For supply chain heads, the use of commercially available starting materials like L-phenylalanine and simple aryl bromides ensures a stable and resilient raw material base, mitigating risks associated with sourcing exotic or proprietary reagents. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts often used in alternative cross-coupling strategies, combined with the high turnover efficiency of the copper system, leads to a drastic simplification of the cost structure. The process avoids the need for costly chiral separation techniques post-reaction due to the high intrinsic stereoselectivity, which directly translates to lower processing costs per kilogram of product. Additionally, the use of ethanol as a preferred solvent reduces solvent procurement costs and simplifies solvent recovery systems, further enhancing the economic viability of the manufacturing process for large-scale API production.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system allows for flexible manufacturing windows, as the reaction is not overly sensitive to minor fluctuations in temperature or moisture within the specified range. This tolerance reduces the risk of batch failures and reworks, ensuring a more predictable output volume. The scalability demonstrated in the patent, maintaining selectivity from milligram to gram scales, provides assurance that the process can be seamlessly transferred to multi-kilogram or ton-scale production without losing performance, thereby securing long-term supply continuity for key therapeutic programs.
- Scalability and Environmental Compliance: The methodology supports green chemistry principles by utilizing less hazardous solvents and generating fewer toxic byproducts. The simplified workup procedure, involving standard extraction and column chromatography or recrystallization, minimizes the use of auxiliary chemicals. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly a criterion for vendor selection by major pharmaceutical companies. The ability to scale this process while maintaining high purity standards ensures that regulatory filings can be supported with consistent, high-quality data.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral copper catalysis technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN114160206B, providing clarity on performance metrics and operational parameters. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing workflows. The answers reflect the proven capabilities of the catalyst system in delivering high-value chiral intermediates with consistency and reliability.
Q: What level of stereoselectivity can be achieved with this chiral copper catalyst?
A: According to patent CN114160206B, the catalyst achieves an enantioselectivity of greater than 91% ee and a diastereoselectivity (dr) of greater than 20:1, ensuring high optical purity for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the stereoselectivity of the product is maintained even when the reaction is scaled up to the gram scale, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Q: What are the advantages over conventional Lewis acid catalysts?
A: Compared to conventional Lewis acids, this chiral copper-based catalyst offers significant improvements in both selectivity and productivity, operating under milder conditions (0-25°C) while delivering yields exceeding 81%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be successfully translated into industrial reality. We are committed to delivering high-purity optically active indole compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex chiral synthesis allows us to offer customized solutions that address the specific needs of your pipeline, whether for preclinical research or late-stage clinical supply.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic method can be applied to your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and transparency. Let us be your strategic partner in navigating the complexities of chiral intermediate synthesis, ensuring your supply chain remains robust, compliant, and cost-effective.
