Advanced Refining Technology for High-Purity Dabigatran Etexilate API Manufacturing
Advanced Refining Technology for High-Purity Dabigatran Etexilate API Manufacturing
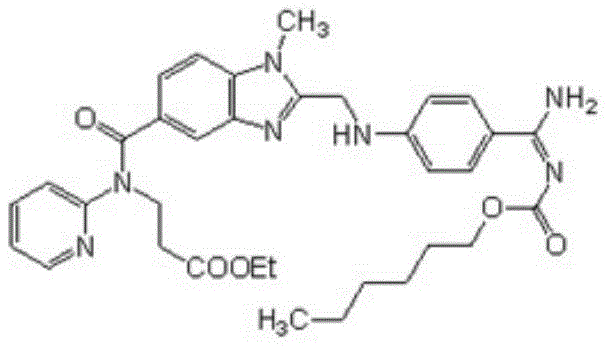
The pharmaceutical landscape for anticoagulant therapies has been fundamentally transformed by the introduction of direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule in modern cardiovascular care. As detailed in the pivotal patent documentation CN111393412A, published in July 2020, a breakthrough refining methodology has been established to address the persistent challenges associated with the purification of this complex active pharmaceutical ingredient (API). The patent outlines a sophisticated two-stage crystallization protocol that leverages specific solvent interactions to achieve unprecedented levels of purity, targeting a critical market need for reliable high-purity API intermediates. This technical advancement is not merely an incremental improvement but represents a paradigm shift in how manufacturers approach the removal of stubborn structural analogs and process-related impurities that have historically plagued the production of Dabigatran Etexilate.
For R&D directors and process chemists, the significance of this patent lies in its ability to consistently deliver product with an HPLC purity exceeding 99.7% and a maximum single impurity content controlled within 0.05%. These specifications are vital for meeting the stringent regulatory requirements of global health authorities, particularly when considering the subsequent conversion to the mesylate salt form. The methodology described eliminates the reliance on labor-intensive and low-yield chromatographic techniques, replacing them with a scalable, robust crystallization sequence that utilizes acetone, water, and ethyl acetate. By optimizing the polarity profile of the solvent system, the process effectively segregates impurities based on their solubility characteristics, ensuring that the final crystal lattice is composed almost exclusively of the target therapeutic molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations disclosed in CN111393412A, the industrial purification of Dabigatran Etexilate was fraught with inefficiencies that severely impacted both cost structures and supply chain reliability. Traditional approaches often relied heavily on silica gel column chromatography, a technique that, while effective for small-scale laboratory synthesis, is notoriously difficult to scale for commercial manufacturing. Data from prior art indicates that silica gel purification typically results in yields hovering around 70%, representing a substantial loss of valuable starting material. Furthermore, the use of large volumes of eluents such as dichloromethane and methanol creates significant environmental burdens and necessitates complex solvent recovery systems, driving up the operational expenditure for cost reduction in API manufacturing.
Alternative recrystallization attempts using solvents like toluene, hexanol, or tert-butyl alcohol have also demonstrated significant shortcomings. For instance, while toluene might offer a theoretical recovery rate of up to 85%, it frequently fails to adequately reduce impurity levels, leaving the product below the required pharmacopeial standards. In many cases, repeated recrystallization cycles in these suboptimal solvent systems lead to a paradoxical increase in impurity content due to the co-crystallization of structurally similar byproducts or the degradation of the sensitive ester linkages within the molecule. These limitations create a bottleneck for procurement managers seeking commercial scale-up of complex pharmaceutical intermediates, as the inability to guarantee consistent purity profiles leads to batch rejections and extended lead times.
The Novel Approach
The novel approach introduced in the patent circumvents these historical barriers through a strategic dual-solvent crystallization design. The process initiates with a primary refinement step using an acetone and water mixture, specifically optimized at a volume ratio of 2:1. This specific solvent combination is engineered to exploit the differential solubility of polar impurities, which are effectively retained in the mother liquor while the target compound precipitates. Following this initial purification, the solid is subjected to a secondary crystallization using ethyl acetate. This second stage is critical for removing non-polar or lipophilic impurities that the aqueous acetone system might miss. The result is a synergistic purification effect that achieves a refining yield of over 83% while simultaneously driving total impurities down to negligible levels.
This methodology offers a distinct advantage in terms of process robustness and scalability. By operating at moderate temperatures—dissolving at 30-50°C for the first stage and 70-80°C for the second—the process minimizes thermal stress on the molecule, preserving its chemical integrity. The use of common, industrially available solvents like acetone and ethyl acetate simplifies the supply chain logistics, reducing the risk of raw material shortages. For supply chain heads, this translates to a more predictable production schedule and reducing lead time for high-purity APIs. The elimination of chromatographic steps not only accelerates the batch cycle time but also drastically reduces the physical footprint required for production, allowing existing facilities to increase throughput without major capital investment in new equipment.
Mechanistic Insights into Solvent-Driven Impurity Segregation
The success of this refining protocol is rooted in a deep understanding of the physicochemical properties of Dabigatran Etexilate and its associated impurities. The molecule, characterized by a benzimidazole core flanked by ester and amidine functionalities, exhibits amphiphilic characteristics that make it susceptible to trapping various types of byproducts during synthesis. The initial acetone/water crystallization acts as a polarity filter. Acetone, being a polar aprotic solvent, effectively dissolves the crude material, while the addition of water modulates the dielectric constant of the medium. This adjustment reduces the solubility of the target compound just enough to induce nucleation, while keeping highly polar synthetic byproducts and inorganic salts in solution. The precise control of the water-to-acetone ratio is paramount; deviations can lead to either oiling out or the co-precipitation of impurities, which is why the patent specifies a narrow operational window.
Following the removal of polar contaminants, the ethyl acetate crystallization targets the remaining lipophilic fraction. Ethyl acetate is a moderately polar solvent that provides an ideal environment for the selective crystallization of the Dabigatran Etexilate free base. At elevated temperatures (70-80°C), the solubility of the API is maximized, allowing for the dissolution of any occluded non-polar impurities. Upon controlled cooling to 15-25°C, the API crystallizes in a highly ordered lattice, excluding the impurities which remain dissolved due to their differing saturation points. This mechanism ensures that the final product not only meets the >99.7% purity threshold but also maintains a maximum single impurity level below 0.05%, a specification that is critical for preventing toxicological issues in the final drug product. This rigorous control over the impurity profile underscores the value of this technology for partners seeking a reliable API supplier capable of meeting global quality standards.
How to Synthesize Dabigatran Etexilate Efficiently
The implementation of this refining strategy requires precise adherence to the thermal and volumetric parameters outlined in the patent to ensure reproducibility at scale. The process begins with the dissolution of the crude Dabigatran Etexilate in acetone, followed by the controlled addition of purified water to induce crystallization of the primary refined product. After filtration and drying, this intermediate is then subjected to the ethyl acetate recrystallization step to achieve final API grade quality. The detailed standardized synthesis steps, including specific stirring rates, cooling gradients, and filtration protocols necessary for GMP compliance, are provided in the technical guide below.
- Dissolve crude Dabigatran Etexilate in acetone at 30-50°C, filter hot, add purified water (acetone/water ratio 2/1), and crystallize at 15-30°C to remove polar impurities.
- Collect the primary refined solid via filtration and vacuum dry at 45°C to constant weight.
- Dissolve the primary refined product in ethyl acetate at 70-80°C, cool to 15-25°C to crystallize, and dry to obtain final high-purity API (>99.7% HPLC).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this refining technology offers substantial benefits that extend far beyond simple yield improvements. For procurement managers and supply chain executives, the shift from chromatographic purification to a purely crystallization-based workflow represents a significant optimization of the cost of goods sold (COGS). The elimination of silica gel and the reduction in solvent diversity simplify the raw material sourcing strategy, mitigating the risks associated with volatile solvent markets. Furthermore, the higher throughput achieved by reducing the number of processing steps directly correlates to improved asset utilization, allowing manufacturers to fulfill large-volume orders with greater agility and reliability.
- Cost Reduction in Manufacturing: The transition away from column chromatography eliminates the recurring cost of stationary phases and significantly reduces solvent consumption volumes. By achieving yields greater than 83% compared to the historical 70% baseline, the process maximizes the utility of expensive starting materials, effectively lowering the unit cost of production. Additionally, the use of acetone and ethyl acetate facilitates easier solvent recovery and recycling due to their favorable boiling points and miscibility profiles, further driving down waste disposal costs and enhancing the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this crystallization method ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply to downstream formulation partners. The simplified process flow reduces the potential for human error and equipment failure, leading to more predictable delivery schedules. By minimizing the reliance on complex separation technologies that often require specialized expertise and maintenance, the supply chain becomes more resilient to operational disruptions, ensuring that critical anticoagulant medications reach patients without delay.
- Scalability and Environmental Compliance: This refining method is inherently designed for industrial scale-up, utilizing standard reactor configurations and filtration equipment found in most multipurpose pharmaceutical plants. The reduction in hazardous solvent usage, particularly the avoidance of chlorinated solvents like dichloromethane in the purification stage, aligns with modern green chemistry principles and stricter environmental regulations. This compliance reduces the regulatory burden on the manufacturer and minimizes the environmental footprint of the production process, making it a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced refining technology. These insights are derived directly from the experimental data and process descriptions found in patent CN111393412A, providing a clear understanding of how this method resolves historical purification bottlenecks. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: What is the primary advantage of the acetone/water and ethyl acetate refining method?
A: This dual-solvent strategy effectively removes both polar and non-polar stubborn impurities that persist in conventional single-solvent recrystallization, achieving a maximum single impurity content of less than 0.05% and overall purity exceeding 99.7%.
Q: How does this process improve yield compared to traditional silica gel purification?
A: Traditional silica gel column chromatography typically yields only around 70% with significant solvent consumption. The patented crystallization method achieves yields greater than 83%, significantly reducing material loss and processing time.
Q: Why is moisture control critical in the refining of Dabigatran Etexilate?
A: Dabigatran Etexilate exhibits poor stability in aqueous solvents, and excessive moisture can complicate the subsequent salt formation reaction to create the mesylate. The optimized acetone/water ratio and final ethyl acetate wash ensure low water content suitable for downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex molecules like Dabigatran Etexilate depends on mastering the delicate balance between chemical purity and process efficiency. Our team of expert process chemists has extensively analyzed the technological advancements presented in CN111393412A and possesses the capability to implement this high-yield refining strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the <0.05% single impurity profile required for this API, guaranteeing that every batch is ready for immediate salt formation and formulation.
We invite global pharmaceutical partners to collaborate with us to leverage this superior refining technology for their anticoagulant portfolios. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how this optimized process can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from development to commercial supply.
