Industrial Scale-Up of High-Purity Dabigatran Intermediates via Novel Crystallization Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical anticoagulant agents, and patent CN102985416A presents a transformative approach for synthesizing thrombin-specific inhibitors. This intellectual property details a sophisticated chemical process designed to prepare compounds of formula (I), specifically focusing on the prodrug dabigatran etexilate and its pharmaceutically acceptable salts. The core innovation lies in the strategic isolation of key intermediates in solid crystalline forms, which fundamentally alters the purification landscape compared to traditional solution-phase methods. By shifting the paradigm from chromatographic separation to controlled crystallization, this technology addresses the persistent bottlenecks of scalability and purity that often plague the commercial production of complex cardiovascular medications. 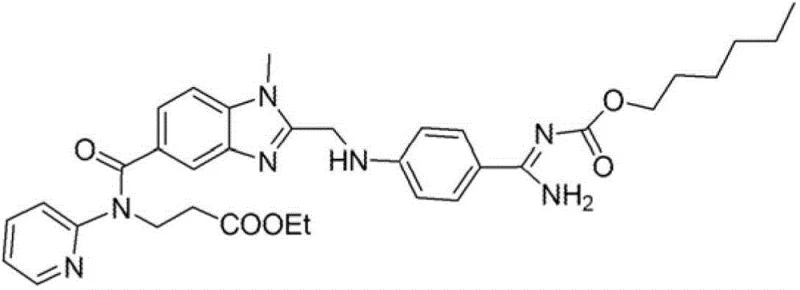
For R&D directors evaluating process viability, the ability to achieve high chemical purity without resorting to labor-intensive chromatography is a decisive factor. The patent elucidates a pathway where intermediates such as the imido-ester hydrochloride are precipitated directly from the reaction medium, allowing for efficient removal of soluble impurities through simple filtration and washing protocols. This methodological shift not only streamlines the operational workflow but also ensures that the final active pharmaceutical ingredient meets stringent regulatory specifications for impurity profiles. Consequently, this process represents a significant leap forward in the manufacturability of high-purity pharmaceutical intermediates, offering a reliable foundation for consistent drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in WO98/37075, rely heavily on the use of excessive amounts of volatile salts and ammonia equivalents to drive the conversion of cyano intermediates to amidino compounds. These conventional routes typically necessitate the use of as many as 10 to 16 equivalents of ammonia sources, which results in the generation of substantial quantities of ammonium chloride salt residues within the reaction mixture. The presence of these inorganic salts creates severe downstream processing challenges, often requiring tedious purification steps such as column chromatography to achieve the necessary purity levels for subsequent reactions. Furthermore, the accumulation of salt waste hinders plant-scale amplification, as the removal of these residues becomes increasingly difficult and costly as batch sizes increase from laboratory to industrial scales. 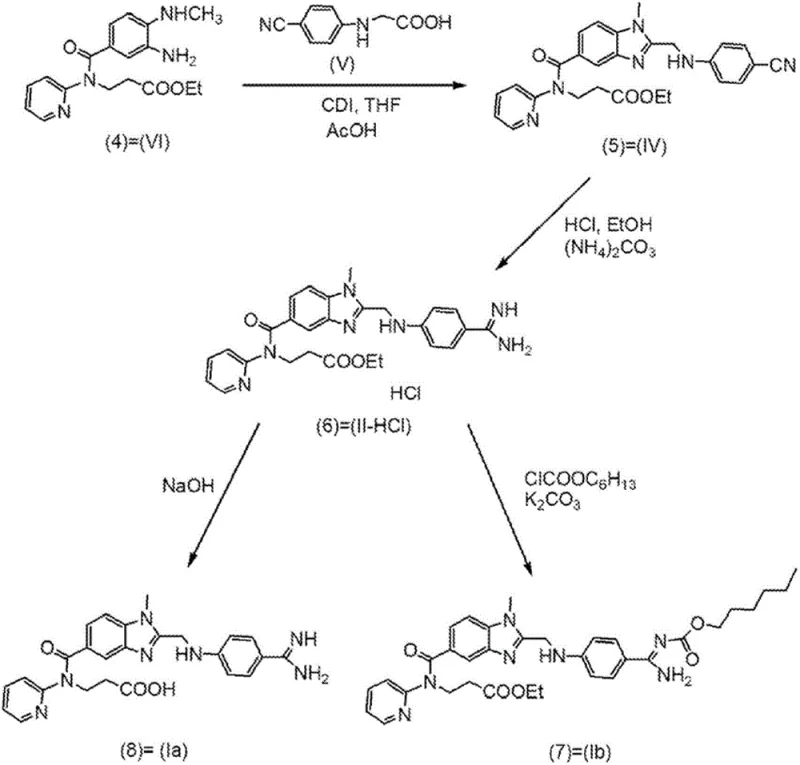
The reliance on chromatographic purification in traditional synthesis routes introduces significant variability and risk into the supply chain, making it unsuitable for the consistent demands of commercial API manufacturing. The solvents required for chromatography add to the environmental burden and operational costs, while the time-consuming nature of column separation extends the overall production cycle time. Additionally, the difficulty in completely removing ammonium salts can interfere with subsequent esterification steps, potentially consuming essential reagents like haloformic esters and reducing the overall yield of the final product. These cumulative inefficiencies highlight the critical need for an alternative synthetic strategy that eliminates chromatography and minimizes salt waste generation.
The Novel Approach
The innovative process disclosed in CN102985416A overcomes these historical limitations by introducing a pivotal isolation step involving the imido-ester intermediate (III-HCl). By converting the cyano precursor into a solid imido-ester hydrochloride form, the inventors have created a checkpoint that allows for the physical separation of the desired intermediate from reaction byproducts before the amidination step occurs. This strategic intervention means that the subsequent conversion to the amidino compound can be performed with a drastically reduced amount of ammonia or ammonium salt, typically between 3 to 5 equivalents instead of the 11 or more required by older methods. The reduction in reagent usage directly correlates to a decrease in the formation of insoluble ammonium salt residues, thereby simplifying the workup procedure and eliminating the need for chromatographic purification entirely. 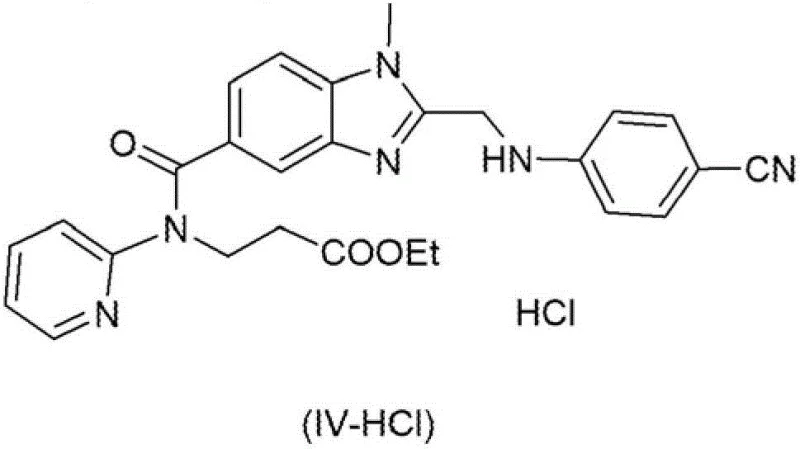
Furthermore, this novel approach facilitates the production of the amidino compound (II-HCl) in specific crystalline forms, such as Type I or Type V, which possess superior mechanical properties for industrial handling. These crystalline forms exhibit excellent filtration characteristics and stability, allowing them to be isolated as free-flowing solids that are substantially free of other polymorphic contaminants. The ability to obtain the intermediate in a defined solid state not only enhances the purity profile, often exceeding 99% area/area by HPLC, but also ensures batch-to-batch consistency which is paramount for regulatory compliance. This method effectively transforms a complex, waste-generating process into a streamlined, scalable operation suitable for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Imido-Ester Hydrochloride Isolation
The mechanistic cornerstone of this improved synthesis is the controlled precipitation of the imido-ester hydrochloride (III-HCl) from a solution of the cyano intermediate (IV). This transformation is achieved by reacting the cyano compound with hydrochloric acid in a solvent system comprising ethanol, optionally mixed with a cosolvent like toluene. The choice of solvent mixture is critical, as it modulates the solubility of the resulting imido-ester salt, promoting its crystallization while keeping impurities in the mother liquor. The reaction is typically conducted at moderate temperatures between 10°C and 50°C, allowing for the gradual formation of well-defined crystals that can be easily filtered. This solid-state isolation acts as a powerful purification gate, ensuring that only the desired chemical species proceeds to the next stage of the synthesis, thereby protecting the integrity of the final molecule.
Following the isolation of the imido-ester, the conversion to the amidino compound is managed through a carefully controlled ammonolysis reaction. By utilizing the pre-purified solid intermediate, the process requires significantly less ammonia source, which minimizes the generation of ammonium chloride byproducts that are detrimental to downstream processing. The reaction conditions are optimized to favor the formation of specific polymorphs of the amidino hydrochloride, such as the stable Type I form, which can be characterized by distinct X-ray powder diffraction patterns. This level of control over the solid-state chemistry ensures that the intermediate possesses the necessary physical properties for efficient filtration and drying, ultimately leading to a high-purity product that is ready for the final esterification steps without further purification.
How to Synthesize Dabigatran Etexilate Efficiently
The implementation of this synthesis route requires precise adherence to the crystallization parameters defined in the patent to ensure optimal yield and purity. The process begins with the preparation of the cyano intermediate, which is then subjected to acid treatment in an alcoholic solvent to induce the precipitation of the imido-ester hydrochloride. Detailed standardized synthesis steps see the guide below, which outlines the specific solvent ratios, temperature profiles, and stirring times necessary to reproduce the results described in the intellectual property. Operators must pay close attention to the cooling rates and anti-solvent addition strategies, as these factors dictate the crystal habit and purity of the isolated solid.
- React cyano intermediate (IV) with hydrochloric acid and ethanol to precipitate solid imido-ester hydrochloride (III-HCl).
- Treat the isolated solid (III-HCl) with ammonia or ammonium carbonate in alcohol to convert it to amidino compound (II-HCl).
- Convert the purified amidino intermediate into the final dabigatran etexilate via esterification and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the elimination of chromatography and the reduction of salt waste offer profound economic and logistical benefits. Traditional methods that rely on column purification are inherently difficult to scale, often requiring specialized equipment and large volumes of solvents that drive up both capital expenditure and operating costs. By replacing these steps with simple filtration and crystallization unit operations, the new process significantly lowers the barrier to entry for large-scale manufacturing, enabling suppliers to meet high-volume demand with greater efficiency. This operational simplification translates directly into enhanced supply chain reliability, as the risk of production delays associated with complex purification bottlenecks is substantially mitigated.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps eliminates the need for expensive silica gel and large quantities of chromatography-grade solvents, leading to substantial cost savings in raw materials. Additionally, the reduction in ammonia equivalents from over 10 to approximately 3 per mole decreases the consumption of reagents and reduces the volume of waste that requires treatment. These efficiencies collectively lower the cost of goods sold, making the production of high-purity pharmaceutical intermediates more economically viable in a competitive market.
- Enhanced Supply Chain Reliability: The robustness of the crystallization-based process ensures consistent batch quality and shorter cycle times, which are critical for maintaining continuous supply to downstream API manufacturers. The ability to isolate stable solid intermediates allows for inventory buffering, providing flexibility in production scheduling and reducing the risk of stockouts. This reliability is essential for pharmaceutical partners who require guaranteed delivery schedules to support their own clinical and commercial timelines without interruption.
- Scalability and Environmental Compliance: The process is designed for industrial scalability, utilizing standard reactor equipment and avoiding the engineering challenges associated with scaling chromatographic columns. Furthermore, the significant reduction in ammonium salt waste aligns with increasingly stringent environmental regulations, minimizing the ecological footprint of the manufacturing process. This commitment to green chemistry principles not only ensures regulatory compliance but also enhances the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of thrombin inhibitor intermediates. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and advantages. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing needs.
Q: How does this process eliminate chromatography?
A: By isolating the imido-ester intermediate (III-HCl) as a solid crystalline form, impurities are removed via filtration and washing, removing the need for column chromatography.
Q: What are the benefits of the new crystalline forms?
A: The novel crystal forms (Type I and Type V) exhibit superior filtration properties and stability, facilitating easier handling and drying during large-scale production.
Q: Does this method reduce chemical waste?
A: Yes, the process significantly reduces the amount of ammonium salt residues generated during the amidination step compared to conventional methods using excess volatile salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced process technologies like the one described in CN102985416A to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can seamlessly transition this innovative synthesis from the laboratory to full-scale industrial operation. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch of Dabigatran Etexilate intermediate meets the highest standards of quality and consistency required by the pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the production of high-quality cardiovascular therapeutics.
