Scalable Production of High-Purity Watermelon Ketone via Advanced Hypervalent Iodine Oxidation
Introduction to Advanced Watermelon Ketone Manufacturing
The global demand for high-quality olfactory ingredients in the fine fragrance and daily chemical sectors continues to drive innovation in synthetic methodology. A pivotal advancement in this domain is detailed in Chinese Patent CN113651795A, which discloses a robust and efficient synthesis method for Watermelon Ketone (3,4-dihydro-7-methyl-2H-1,5-benzoxazol-3-one). This precious spice, renowned for its unique marine odor characteristics and fresh, melon-like sweetness, has historically faced production bottlenecks due to complex multi-step syntheses and the use of hazardous reagents. The disclosed technology addresses these critical pain points by introducing a streamlined oxidation strategy using periodic acid series reagents. By shifting from traditional, harsh oxidative conditions to a controlled hypervalent iodine-mediated transformation, this method achieves exceptional product purity exceeding 99% while maintaining high reaction yields. For R&D directors and procurement specialists seeking a reliable watermelon ketone supplier, understanding the mechanistic advantages and commercial viability of this route is essential for securing a stable supply of this high-value flavor intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Watermelon Ketone has been plagued by significant safety and efficiency challenges that hinder cost-effective manufacturing. Early synthetic routes, such as the Williams etherification followed by Dieckmann condensation, necessitated the use of strong bases like sodium hydride under rigorous anhydrous conditions, often resulting in mediocre yields around 60% and requiring expensive quenching procedures. More critically, alternative pathways relied heavily on 1,3-dichloroacetone as a key building block. This reagent is notoriously dangerous; it is a potent lachrymator that releases toxic, tear-inducing vapors at ambient temperatures and is classified as a potential mutagen and carcinogen. Handling such hazardous materials imposes severe burdens on supply chain heads regarding worker safety, specialized containment infrastructure, and waste disposal compliance. Furthermore, other reported methods involving chloroacetonitrile or multi-step protection-deprotection sequences utilizing vanadium pentoxide and hydrogen peroxide introduced excessive operational complexity. These legacy processes typically involved four or more distinct reaction steps, leading to cumulative yield losses where the overall efficiency dropped to approximately 52%, thereby inflating the cost of goods sold and limiting the availability of high-purity material for premium perfume applications.
The Novel Approach
The innovative methodology presented in the patent data fundamentally reengineers the synthesis landscape by decoupling the hazardous precursor formation from the final oxidative step. Instead of relying on toxic dichloroacetone, the process utilizes the safer and more commercially accessible 1,3-dichloropropanol to generate the intermediate alcohol precursor, 3,4-dihydro-7-methyl-2H-1,5-benzoxazol-3-ol. The core breakthrough lies in the subsequent one-step oxidation of this secondary alcohol to the target ketone using periodic acid derivatives. Unlike traditional oxidants such as potassium permanganate or sodium hypochlorite, which can be non-selective and degrade the sensitive benzoxazole ring system, periodic acid reagents like Dess-Martin periodinane, 2-iodobenzoic acid (IBX), or 2-iodobenzenesulfonic acid offer precise chemoselectivity. This novel approach allows the reaction to proceed under mild conditions, often at room temperature or with moderate heating below 95°C, eliminating the need for cryogenic cooling or extreme thermal inputs. The result is a drastic simplification of the workflow, where the target molecule is obtained in a single oxidative transformation with yields soaring up to 95%, representing a paradigm shift in cost reduction in flavors & fragrances manufacturing.
Mechanistic Insights into Hypervalent Iodine Oxidation
The success of this synthesis hinges on the unique electronic properties of hypervalent iodine species, which act as powerful yet selective oxygen transfer agents. In the context of Watermelon Ketone production, the substrate contains a secondary alcohol adjacent to a heterocyclic oxygen atom, creating a steric and electronic environment that resists oxidation by common chromium-based or metal-catalyzed systems. The periodic acid oxidants function by forming a transient alkoxy-iodinane intermediate with the hydroxyl group of the precursor alcohol. This intermediate undergoes a concerted elimination reaction, effectively removing two hydrogen atoms to form the carbonyl double bond while reducing the iodine species. This mechanism is particularly advantageous because it avoids the generation of free radical species that could otherwise attack the electron-rich aromatic ring or the ether linkages within the benzoxazole scaffold. Consequently, the reaction profile is exceptionally clean, minimizing the formation of over-oxidized byproducts or ring-opened impurities that are difficult to separate via standard distillation or crystallization techniques.
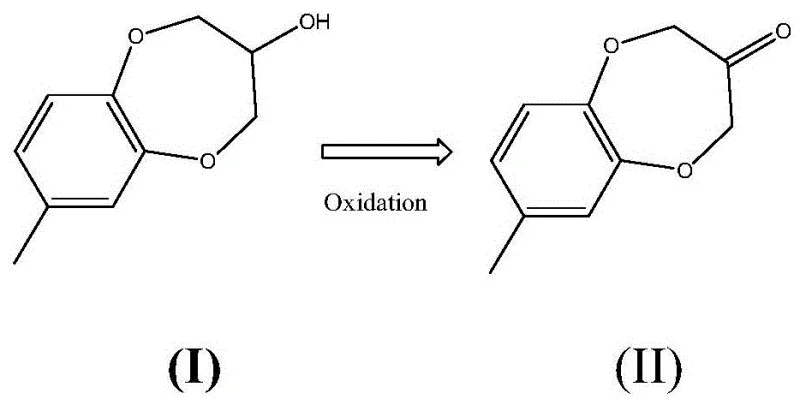
Furthermore, the choice of oxidant allows for fine-tuning of the reaction kinetics to match specific production scales. For instance, when using o-iodobenzenesulfonic acid, the reaction exhibits rapid kinetics even at lower temperatures, completing within 3 to 7 hours with minimal exothermic risk. The solubility profiles of the reduced iodine byproducts also facilitate easy removal; in many embodiments, the spent oxidant precipitates out of the reaction mixture or remains in the aqueous phase during workup, allowing for simple filtration to isolate the crude ketone. This inherent ease of purification is a critical factor for R&D teams focused on impurity control, as it ensures that the final recrystallization step yields a product with purity levels consistently above 99%. The ability to utilize diverse solvent systems, ranging from polar aprotic solvents like DMSO and DMF to greener options like ethyl acetate and tert-butanol, further enhances the versatility of this mechanistic pathway, enabling manufacturers to optimize for both performance and environmental sustainability without compromising the integrity of the catalytic cycle.
How to Synthesize Watermelon Ketone Efficiently
Implementing this advanced oxidation protocol requires careful attention to reagent stoichiometry and solvent selection to maximize the economic and technical benefits described in the patent literature. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or inert atmosphere gloveboxes, which makes it highly attractive for commercial scale-up of complex flavor intermediates. The standard procedure involves dissolving the periodic acid oxidant in a selected solvent, followed by the controlled addition of the alcohol precursor to manage any exothermicity. Following the reaction period, the workup is straightforward, typically involving filtration to remove insoluble iodine species and liquid-liquid extraction to isolate the organic product. For a comprehensive understanding of the specific parameters required to replicate these high-yield results in a GMP environment, please refer to the standardized synthesis guide below.
- Prepare the reaction vessel by adding a suitable polar aprotic or ester solvent along with the periodic acid oxidant (such as Dess-Martin reagent or IBX) at room temperature.
- Dropwise add the solution of watermelon ketone precursor alcohol (3,4-dihydro-7-methyl-2H-1,5-benzoxazol-3-ol) dissolved in the same solvent into the reaction mixture while stirring continuously.
- Stir the reaction for 3 to 7 hours, filter the mixture to remove solid byproducts, concentrate the filtrate via rotary evaporation, and recrystallize the crude product to obtain pure Watermelon Ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this periodic acid-mediated synthesis route offers profound strategic advantages that extend beyond mere chemical yield. The primary value driver is the substantial enhancement in operational safety and regulatory compliance. By eliminating the requirement for 1,3-dichloroacetone, a substance associated with severe health hazards and strict handling regulations, manufacturers can significantly reduce the costs associated with hazardous waste disposal, specialized ventilation systems, and personal protective equipment. This shift not only mitigates liability risks but also streamlines the logistics of raw material sourcing, as safer precursors are generally more readily available and less subject to shipping restrictions. Additionally, the simplification of the synthetic sequence from multiple steps to a concise oxidation process directly translates to reduced manufacturing lead times. Fewer unit operations mean less equipment occupancy time, lower labor overheads, and decreased energy consumption for heating and cooling cycles, all of which contribute to a more competitive cost structure in the volatile flavors and fragrances market.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the dramatic improvement in overall process efficiency. Traditional multi-step routes suffered from cumulative yield losses, often leaving more than half of the starting material as waste. In contrast, this novel method achieves near-quantitative conversion in the final step, with isolated yields reaching up to 95%. This high atom economy means that less raw material is required to produce the same amount of finished goods, directly lowering the variable cost per kilogram. Furthermore, the use of recyclable or easily separable iodine reagents reduces the expense of consumable chemicals. The simplified workup procedure, which often requires only filtration and evaporation rather than complex chromatographic separations, minimizes solvent usage and waste treatment costs, delivering significant cost savings without the need for capital-intensive new infrastructure.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche or hazardous intermediates that have limited supplier bases. By adopting a synthesis route based on widely available commodities like 1,3-dichloropropanol and standard periodic acid derivatives, manufacturers can diversify their supply base and reduce dependency on single-source vendors. The robustness of the reaction conditions, which tolerate a range of solvents and operate effectively at ambient temperatures, ensures that production is less susceptible to disruptions caused by utility failures or equipment malfunctions. This resilience is crucial for maintaining consistent delivery schedules to downstream perfume houses and cosmetic formulators, reinforcing the manufacturer's reputation as a dependable partner capable of meeting fluctuating market demands without compromise.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry practices, this oxidation method aligns perfectly with modern environmental, social, and governance (ESG) goals. The avoidance of heavy metal catalysts like chromium or vanadium eliminates the risk of toxic metal contamination in the final product, a critical quality attribute for ingredients used in personal care applications. The process generates significantly less hazardous waste compared to traditional methods, simplifying effluent treatment and reducing the environmental footprint of the manufacturing facility. Moreover, the mild reaction conditions facilitate safe scale-up from laboratory benchtop to multi-ton industrial reactors. The exothermic nature of the oxidation is manageable, allowing for precise temperature control even in large batches, which ensures that the high purity and yield observed in small-scale trials can be faithfully reproduced in commercial production runs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of safety, efficiency, and product quality. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is periodic acid preferred over traditional oxidants like potassium permanganate for Watermelon Ketone synthesis?
A: Traditional oxidants often lead to over-oxidation or ring degradation in sensitive heterocyclic structures. Periodic acid reagents, such as Dess-Martin periodinane or o-iodobenzenesulfonic acid, offer superior chemoselectivity, ensuring the secondary alcohol is oxidized to the ketone without damaging the benzoxazole ring, resulting in significantly higher purity (up to 99.1%) and simplified downstream purification.
Q: How does this synthesis method improve supply chain safety compared to previous industrial routes?
A: Previous methods relied on 1,3-dichloroacetone, a potent lachrymator and suspected carcinogen, posing severe handling risks. This patented process utilizes 1,3-dichloropropanol for the precursor synthesis and mild periodic acid oxidants for the final step. This substitution eliminates the need for handling highly toxic volatile gases, drastically reducing workplace hazards and environmental compliance costs for manufacturers.
Q: What are the expected yields and scalability potential for this oxidation process?
A: The patent data demonstrates exceptional efficiency, with isolated yields reaching up to 95% when using o-iodobenzenesulfonic acid. The reaction operates effectively at room temperature or mild heating (below 95°C), which minimizes energy consumption and thermal stress on equipment. These factors, combined with simple filtration workups, make the process highly amenable to commercial scale-up from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Watermelon Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of high-value fragrance ingredients like Watermelon Ketone requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this oxidation chemistry are fully realized in practical manufacturing settings. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required by the global flavor and fragrance industry. Our capability to handle complex heterocyclic synthesis allows us to deliver products with consistent olfactory profiles and minimal impurity levels, safeguarding the integrity of your final formulations.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can provide you with a secure, cost-effective, and high-quality source of Watermelon Ketone, empowering your business to thrive in the competitive landscape of fine chemicals.
