Advanced Enzymatic Resolution Strategies for High-Purity Taxane Intermediates Manufacturing
Advanced Enzymatic Resolution Strategies for High-Purity Taxane Intermediates Manufacturing
The pharmaceutical industry's relentless pursuit of potent anticancer agents has placed taxanes, such as paclitaxel and its analogs, at the forefront of oncology therapeutics. Central to the synthesis of these complex molecules is the efficient preparation of chiral intermediates, specifically beta-lactam compounds that form the C-13 side chain. Patent CN1100144A introduces a groundbreaking enzymatic process for the resolution of enantiomeric mixtures of these critical intermediates. This technology represents a significant paradigm shift from traditional chemical resolution methods, leveraging the exquisite stereoselectivity of biocatalysts to produce high-purity compounds essential for drug efficacy. By utilizing specific enzymes or microorganisms, manufacturers can achieve superior optical purity while operating under milder reaction conditions, thereby addressing key challenges in the supply chain of high-value pharmaceutical intermediates.
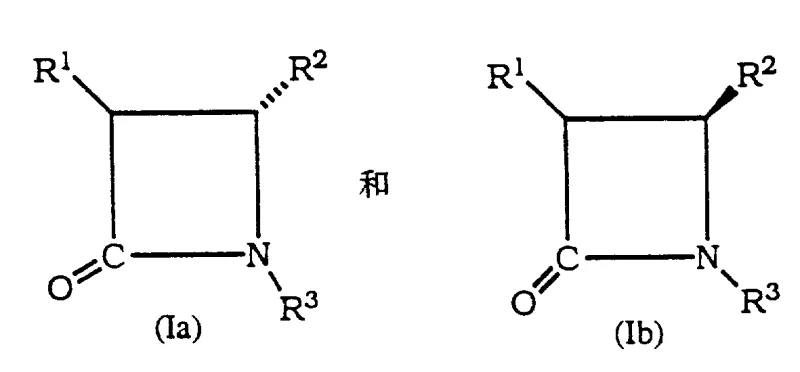
For R&D directors focused on process chemistry, the structural versatility offered by this patent is paramount. The invention covers a broad range of beta-lactam structures where substituents can vary from hydroxyl and halogen groups to acyloxy functionalities, allowing for the customization of intermediates for various taxane analogs. The core innovation lies in the ability to distinguish between enantiomers (Ia and Ib) based on their spatial configuration, specifically targeting cis or trans relationships between substituents. This level of precision ensures that the resulting intermediates possess the exact stereochemistry required for coupling with the taxane nucleus, minimizing the formation of inactive or toxic diastereomers that could complicate downstream purification and regulatory approval processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic beta-lactam mixtures relied heavily on classical chemical methods involving chiral resolving agents or asymmetric synthesis using transition metal catalysts. These conventional approaches often suffer from significant drawbacks, including the requirement for harsh reaction conditions, such as extreme temperatures or strong acids and bases, which can degrade sensitive functional groups present in the molecule. Furthermore, chemical resolution typically yields a maximum theoretical recovery of 50% for the desired enantiomer unless dynamic kinetic resolution is employed, which adds complexity and cost. The use of heavy metal catalysts also introduces the risk of metal contamination, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical standards for residual metals, ultimately impacting the overall cost of goods and manufacturing timeline.
The Novel Approach
In contrast, the enzymatic process described in the patent offers a sustainable and efficient alternative that overcomes these traditional bottlenecks. By employing biocatalysts such as lipases and esterases, the reaction proceeds under mild physiological conditions, typically near neutral pH and moderate temperatures, preserving the integrity of the delicate beta-lactam ring and other sensitive moieties. The inherent chirality of the enzyme's active site allows for highly specific recognition of one enantiomer over the other, facilitating reactions like stereospecific hydrolysis or esterification with exceptional selectivity. This biological approach not only improves the yield of the desired optically active compound but also simplifies the workflow by eliminating the need for stoichiometric amounts of chiral auxiliaries, thereby reducing waste generation and aligning with green chemistry principles increasingly demanded by global regulatory bodies.
Mechanistic Insights into Lipase-Catalyzed Stereoselective Hydrolysis
The mechanistic foundation of this technology rests on the precise interaction between the enzyme's active site and the substrate's stereochemical features. In the case of lipase-catalyzed hydrolysis, the enzyme selectively binds to one enantiomer of the acylated beta-lactam, positioning the ester bond for nucleophilic attack by water or a hydroxide ion. This binding is governed by a lock-and-key or induced-fit model where the spatial arrangement of substituents, such as the furyl or cycloalkyl groups at the C-4 position, dictates whether the molecule fits into the catalytic pocket. For instance, Lipase PS-30 demonstrates a profound preference for hydrolyzing the acetoxy group of specific cis-enantiomers while leaving the mirror image untouched, effectively converting a racemic mixture into a pair of separable compounds with distinct physical properties.
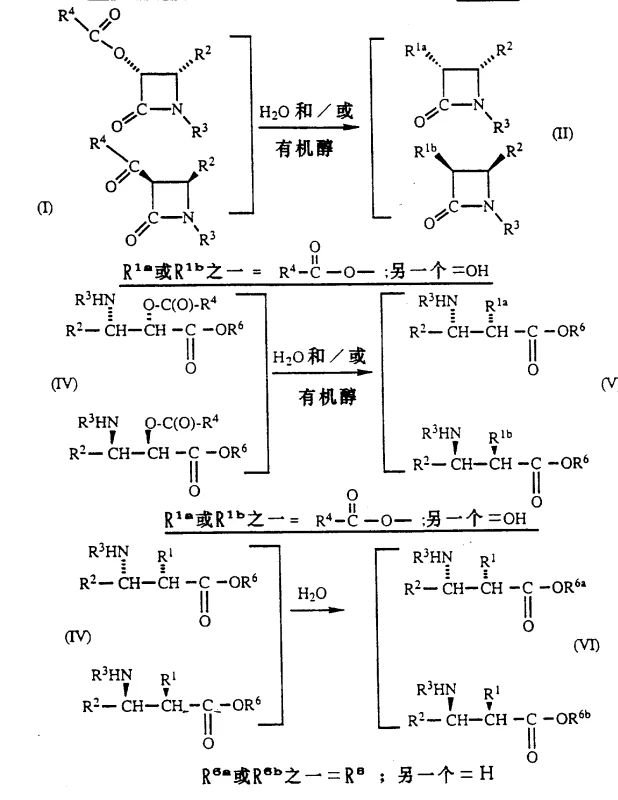
Understanding the impurity profile is crucial for scaling this process, and the patent provides detailed insights into controlling side reactions. The stereoselectivity ensures that unwanted diastereomers are minimized, but the process also accounts for potential over-hydrolysis or non-specific cleavage. By optimizing parameters such as water content in organic solvents and pH control using buffers, manufacturers can steer the reaction towards the desired product with high fidelity. The use of immobilized enzymes further enhances this control by restricting the conformational flexibility of the biocatalyst, often leading to improved stability and reduced leaching of proteinaceous material into the product stream. This rigorous control over the reaction environment is essential for maintaining the stringent purity specifications required for intermediates destined for clinical applications.
How to Synthesize Beta-Lactam Intermediates Efficiently
The synthesis of these high-value intermediates involves a streamlined sequence that integrates enzymatic resolution seamlessly into the production workflow. The process begins with the preparation of the racemic beta-lactam substrate, often via Staudinger cycloaddition, followed by the critical enzymatic step where the mixture is contacted with the selected biocatalyst. Detailed operational parameters, including solvent selection, enzyme loading, and reaction monitoring via HPLC, are critical for success. The patent outlines specific protocols for both hydrolysis and esterification pathways, providing a flexible toolkit for chemists to adapt the method to specific substrate variations. For a comprehensive guide on executing these steps with precision, please refer to the standardized synthesis protocol below.
- Prepare a mixture of enantiomeric beta-lactam compounds containing heterocyclic or cycloalkyl groups suitable for taxane C-13 side chain formation.
- Contact the mixture with a stereospecific enzyme or microorganism, such as lipase PS-30, under controlled pH and temperature conditions to catalyze selective hydrolysis or esterification.
- Separate the resulting diastereomeric products or unreacted substrates using standard purification techniques like extraction or chromatography to isolate the optically active compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift from chemical to enzymatic resolution fundamentally alters the cost structure of manufacturing taxane intermediates by removing the dependency on expensive chiral reagents and heavy metal catalysts. This transition not only lowers the direct material costs but also reduces the burden on waste management systems, as the process generates fewer hazardous by-products. Furthermore, the robustness of modern industrial enzymes, which can often be recycled or used in immobilized forms, ensures a consistent supply of catalyst, mitigating the risks associated with raw material volatility and ensuring long-term production stability.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral resolving agents and transition metal catalysts significantly reduces the raw material expenditure per kilogram of product. Additionally, the milder reaction conditions lower energy consumption for heating and cooling, while the simplified downstream processing reduces the need for complex purification columns, collectively driving down the overall cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: Enzymes are commercially available from multiple global suppliers, reducing the risk of single-source dependency that often plagues specialized chemical reagents. The ability to use whole cells or crude enzyme preparations further diversifies the supply base, ensuring that production schedules remain uninterrupted even in the face of market fluctuations for specific fine chemicals.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's examples ranging from gram to multi-kilogram scales, using standard reactor equipment. The aqueous-organic biphasic systems employed are easier to handle and dispose of compared to harsh chemical solvents, facilitating compliance with increasingly strict environmental regulations and reducing the carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this process into existing manufacturing lines and anticipate potential operational requirements.
Q: What specific enzymes are preferred for the resolution of cis-beta-lactam enantiomers?
A: The patent highlights lipases such as Lipase PS-30 from Pseudomonas species, Lipase P-30, and Lipase R from Rhizopus species as highly effective catalysts for stereospecific hydrolysis and esterification.
Q: How does this enzymatic process improve the purity of taxane intermediates compared to chemical methods?
A: Enzymatic resolution offers superior stereoselectivity, often achieving optical purities exceeding 99%, which is critical for the pharmacological activity of final taxane products, avoiding the complex recrystallization steps often needed in chemical resolution.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the use of immobilized enzymes on carriers like polyacrylic resins or diatomite enhances stability and reusability, making the process robust and suitable for large-scale commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Lactam Taxane Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical tools to guarantee that every batch of beta-lactam intermediate meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain resilience and drive value for your organization.
