Advanced Biocatalytic Synthesis of Chiral Cyclopentene Intermediates for Antiviral APIs
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex antiviral nucleoside analogs, particularly those serving as critical building blocks for HIV treatments. Patent CN1115343C introduces a groundbreaking methodology for the preparation of (1S,4R)- or (1R,4S)-4-(2-amino-6-chloro-9H-purin-9-yl)-2-cyclopentene-1-methanol, a pivotal intermediate in the synthesis of Abacavir (1592U89). This technical disclosure moves beyond traditional chemical resolution methods by integrating a sophisticated biocatalytic kinetic resolution step that leverages specific microbial strains to achieve exceptional stereochemical control. The innovation lies not merely in the final product structure but in the strategic reordering of synthetic steps to maximize yield and minimize the reliance on costly chiral auxiliaries early in the sequence. For R&D directors and process chemists, this patent represents a significant leap forward in green chemistry and process intensification, offering a robust alternative to legacy routes that often suffer from low overall yields due to multiple protection and deprotection cycles. By focusing on enzymatic specificity, the process ensures high optical purity, which is paramount for regulatory approval and clinical efficacy of the final active pharmaceutical ingredient.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral cyclopentene amines required for nucleoside analogs relied heavily on resolution strategies that were inherently inefficient and economically burdensome. Prior art methods, such as those described in WO95/21161, typically necessitated the introduction of expensive protecting groups like the tert-butoxycarbonyl (BOC) group at the very beginning of the synthesis on the bicyclic ketone precursor. This early protection strategy locked the molecule into a specific pathway that limited flexibility and added significant cost due to the price of BOC reagents and the additional synthetic steps required for their subsequent removal. Furthermore, achieving high enantiomeric purity often involved classical resolution techniques using chiral acids or bases, which theoretically cap the maximum yield at 50% unless dynamic kinetic resolution is employed, a feat difficult to achieve with simple chemical reagents. These conventional approaches also generated substantial chemical waste, as the unwanted enantiomer was often discarded rather than recycled, creating both environmental liabilities and supply chain inefficiencies for procurement teams managing raw material budgets. The reliance on stoichiometric chiral reagents also introduced variability in batch-to-batch consistency, posing risks for quality control in large-scale manufacturing environments.
The Novel Approach
The novel process disclosed in the patent fundamentally reimagines the synthetic sequence by deferring the introduction of chirality until a later stage where biocatalysis can be most effectively applied. Instead of immediate protection with expensive groups, the process begins with a simple acylation of the racemic bicyclic ketone using cost-effective acylating agents such as acetyl chloride or butyryl chloride.  This initial modification prepares the substrate for a subsequent reduction step that opens the bicyclic ring to form a cyclopentene derivative, which serves as the ideal substrate for enzymatic recognition. By utilizing inexpensive acyl groups initially, the process drastically reduces the cost of goods sold (COGS) associated with raw materials. The true breakthrough occurs in the third step, where specific microorganisms or isolated enzymes are employed to kinetically resolve the racemic mixture. This biological step is highly selective, hydrolyzing only one enantiomer of the N-acyl amino alcohol while leaving the other intact, or vice versa, depending on the specific strain used. This approach bypasses the need for expensive chiral chromatography and allows for the potential recycling of the unwanted enantiomer through racemization, theoretically pushing yields well beyond the 50% limit of classical resolution. The result is a streamlined, cost-effective pathway that aligns perfectly with the goals of modern sustainable manufacturing.
This initial modification prepares the substrate for a subsequent reduction step that opens the bicyclic ring to form a cyclopentene derivative, which serves as the ideal substrate for enzymatic recognition. By utilizing inexpensive acyl groups initially, the process drastically reduces the cost of goods sold (COGS) associated with raw materials. The true breakthrough occurs in the third step, where specific microorganisms or isolated enzymes are employed to kinetically resolve the racemic mixture. This biological step is highly selective, hydrolyzing only one enantiomer of the N-acyl amino alcohol while leaving the other intact, or vice versa, depending on the specific strain used. This approach bypasses the need for expensive chiral chromatography and allows for the potential recycling of the unwanted enantiomer through racemization, theoretically pushing yields well beyond the 50% limit of classical resolution. The result is a streamlined, cost-effective pathway that aligns perfectly with the goals of modern sustainable manufacturing.
Mechanistic Insights into Biocatalytic Kinetic Resolution
The core of this technological advancement lies in the precise mechanism of the enzymatic hydrolysis performed in the third step of the synthesis. The patent details the use of microorganisms possessing N-acylamino alcohol hydrolase activity, such as specific strains of Rhodococcus erythropolis, Alcaligenes, and Pseudomonas. These microbes produce enzymes that exhibit profound stereoselectivity towards the N-acyl cyclopentene derivatives generated in the previous reduction step. 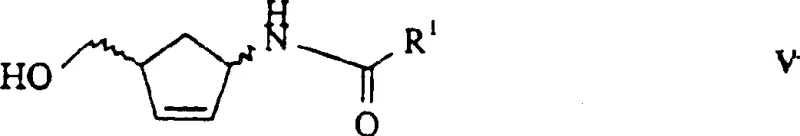 When the racemic substrate is introduced to the fermentation broth or immobilized enzyme system, the biocatalyst selectively recognizes the spatial arrangement of the (1R,4S) or (1S,4R) isomer. The enzyme catalyzes the hydrolysis of the amide bond in one specific enantiomer, releasing the free amine and the corresponding carboxylic acid, while the other enantiomer remains largely unreacted as the N-acyl derivative. This differential reaction rate allows for the physical separation of the two enantiomers based on their differing chemical properties—specifically, the basicity of the free amine versus the neutrality of the amide. The patent highlights that certain strains, like Rhodococcus erythropolis DSM 10686, can achieve enantiomeric excess values exceeding 98%, which is critical for downstream processing. This high level of selectivity minimizes the formation of diastereomeric impurities that are notoriously difficult to remove in later stages of nucleoside synthesis. Furthermore, the use of whole cells or crude extracts simplifies the catalyst preparation process, avoiding the need for extensive enzyme purification which can be a cost-prohibitive bottleneck in industrial biotechnology.
When the racemic substrate is introduced to the fermentation broth or immobilized enzyme system, the biocatalyst selectively recognizes the spatial arrangement of the (1R,4S) or (1S,4R) isomer. The enzyme catalyzes the hydrolysis of the amide bond in one specific enantiomer, releasing the free amine and the corresponding carboxylic acid, while the other enantiomer remains largely unreacted as the N-acyl derivative. This differential reaction rate allows for the physical separation of the two enantiomers based on their differing chemical properties—specifically, the basicity of the free amine versus the neutrality of the amide. The patent highlights that certain strains, like Rhodococcus erythropolis DSM 10686, can achieve enantiomeric excess values exceeding 98%, which is critical for downstream processing. This high level of selectivity minimizes the formation of diastereomeric impurities that are notoriously difficult to remove in later stages of nucleoside synthesis. Furthermore, the use of whole cells or crude extracts simplifies the catalyst preparation process, avoiding the need for extensive enzyme purification which can be a cost-prohibitive bottleneck in industrial biotechnology.
Following the successful resolution of the chiral amine, the process proceeds to the construction of the purine ring system, a step that requires careful control of reaction conditions to maintain the integrity of the chiral center. The resolved amine is coupled with N-(2-amino-4,6-dichloro-5-pyrimidinyl)formamide under basic conditions. 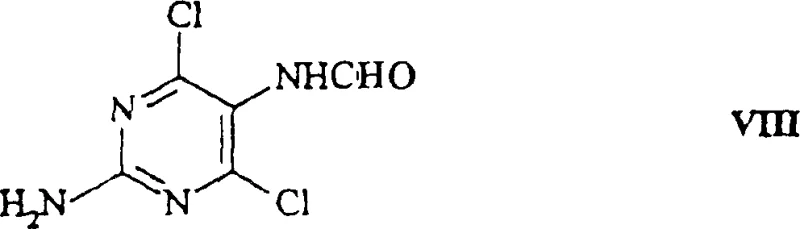 This nucleophilic substitution reaction displaces one of the chlorine atoms on the pyrimidine ring, forming a new carbon-nitrogen bond. The choice of solvent and base is critical here; the patent suggests using lower alkyl alcohols like isobutanol and organic bases like triethylamine to facilitate the reaction while minimizing side reactions. The subsequent cyclization step involves heating the intermediate in the presence of a strong acid and a trialkyl orthoformate, which promotes the closure of the imidazole ring to complete the purine scaffold. Throughout this sequence, the robustness of the chiral center established during the biocatalytic step is maintained, ensuring that the final product retains the high optical purity achieved earlier. This mechanistic understanding is vital for process chemists aiming to scale the reaction, as it highlights the importance of controlling pH and temperature during the enzymatic phase to prevent enzyme denaturation or loss of selectivity, which could compromise the entire batch.
This nucleophilic substitution reaction displaces one of the chlorine atoms on the pyrimidine ring, forming a new carbon-nitrogen bond. The choice of solvent and base is critical here; the patent suggests using lower alkyl alcohols like isobutanol and organic bases like triethylamine to facilitate the reaction while minimizing side reactions. The subsequent cyclization step involves heating the intermediate in the presence of a strong acid and a trialkyl orthoformate, which promotes the closure of the imidazole ring to complete the purine scaffold. Throughout this sequence, the robustness of the chiral center established during the biocatalytic step is maintained, ensuring that the final product retains the high optical purity achieved earlier. This mechanistic understanding is vital for process chemists aiming to scale the reaction, as it highlights the importance of controlling pH and temperature during the enzymatic phase to prevent enzyme denaturation or loss of selectivity, which could compromise the entire batch.
How to Synthesize (1S,4R)-4-(2-amino-6-chloro-9H-purin-9-yl)-2-cyclopentene-1-methanol Efficiently
To implement this advanced synthesis route in a production setting, operators must adhere to a strict sequence of chemical and biological transformations that balance reaction kinetics with stereochemical fidelity. The process begins with the acylation of the bicyclic ketone, followed by reduction to generate the racemic cyclopentene substrate suitable for enzymatic processing. The critical phase involves the fermentation or biotransformation step where specific microbial strains are cultivated and induced to express the necessary hydrolase enzymes. Detailed standard operating procedures regarding substrate feeding rates, pH control, and temperature maintenance during the biotransformation are essential to maximize the enantiomeric excess and conversion yield. Once the resolution is complete, the separation of the free amine from the unreacted amide requires precise extraction and crystallization techniques to isolate the desired isomer. Finally, the coupling and cyclization steps must be monitored closely to ensure complete conversion without epimerization of the chiral center.
- Acylation of (±)-2-aza-bicyclo[2.2.1]hept-5-en-3-one followed by reduction to form racemic N-acyl cyclopentene derivatives.
- Biocatalytic kinetic resolution using specific microorganisms like Rhodococcus erythropolis to separate enantiomers with high ee.
- Coupling the resolved chiral amine with N-(2-amino-4,6-dichloro-5-pyrimidinyl)formamide followed by cyclization to form the final purine scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic process offers transformative advantages that extend far beyond simple yield improvements. The most significant benefit is the drastic reduction in raw material costs associated with chiral reagents. By replacing expensive stoichiometric chiral auxiliaries and protecting groups with catalytic amounts of renewable microbial enzymes, the process decouples production costs from the volatile pricing of specialty fine chemicals. This shift creates a more stable and predictable cost structure, allowing for better long-term budget planning and margin protection. Additionally, the use of fermentation-based biocatalysis aligns with global sustainability mandates, reducing the environmental footprint of the manufacturing process by minimizing hazardous waste generation and solvent usage. This green chemistry profile not only lowers waste disposal costs but also enhances the brand value of the final pharmaceutical product in markets that prioritize environmentally responsible sourcing. The scalability of the fermentation process further ensures supply continuity, as microbial strains can be propagated indefinitely, eliminating the risk of supply shortages associated with finite natural sources or complex synthetic catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive chiral protecting groups like BOC in the early stages of synthesis results in substantial direct material savings. Instead of purchasing high-cost chiral reagents, the process utilizes commodity acylating agents and biocatalysts that can be produced in-house or sourced at a fraction of the cost. This structural change in the bill of materials significantly lowers the variable cost per kilogram of the intermediate, improving overall gross margins for the manufacturing entity. Furthermore, the high selectivity of the enzymatic step reduces the burden on downstream purification, lowering the consumption of solvents and chromatography media which are often major cost drivers in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: Relying on biocatalytic resolution mitigates the risks associated with the supply of specialized chiral chemicals, which can be subject to geopolitical disruptions or single-source bottlenecks. Microbial strains are stable assets that can be stored and regenerated, providing a secure and continuous source of catalytic activity. This biological redundancy ensures that production schedules are not compromised by external supplier failures. Moreover, the robust nature of the selected microorganisms allows for flexible manufacturing across different geographic locations, enabling a distributed production network that enhances resilience against regional logistical challenges and ensures consistent delivery to global customers.
- Scalability and Environmental Compliance: The transition from chemical resolution to biocatalysis facilitates easier scale-up from pilot to commercial volumes without the linear increase in waste treatment costs typical of traditional chemistry. Fermentation processes are well-understood and easily scaled in standard stainless steel bioreactors, allowing for rapid capacity expansion to meet market demand. From a regulatory perspective, the reduction in hazardous waste and the use of aqueous systems for the key resolution step simplify environmental compliance and permitting processes. This operational efficiency accelerates time-to-market for new drug filings and reduces the administrative burden on EHS teams, making the facility more agile and responsive to changing regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own supply chains. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the enzymatic resolution improve cost efficiency compared to traditional chiral separation?
A: The patented process utilizes highly specific microbial enzymes to kinetically resolve racemic mixtures, eliminating the need for expensive chiral chromatography or stoichiometric chiral resolving agents, thereby significantly reducing raw material costs.
Q: What represents the critical quality attribute in this synthesis route?
A: The enantiomeric excess (ee) of the intermediate amine is the critical quality attribute, as the biocatalytic step is designed to achieve greater than 98% ee, ensuring the final API meets stringent regulatory stereochemical specifications.
Q: Can this biocatalytic process be scaled for commercial manufacturing?
A: Yes, the process utilizes robust microbial strains and standard fermentation techniques compatible with large-scale bioreactors, allowing for seamless transition from laboratory optimization to multi-ton commercial production without loss of selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abacavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for high-value antiviral intermediates. Our technical team has extensively analyzed the methodologies described in CN1115343C and possesses the expertise to adapt this biocatalytic resolution technology for commercial-scale production. We understand that transitioning from laboratory patents to industrial reality requires more than just chemical knowledge; it demands deep experience in process engineering and quality assurance. Our facilities are equipped with state-of-the-art fermentation suites and chemical synthesis reactors capable of handling diverse pathways, ensuring that we can deliver extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of chiral cyclopentene intermediate meets the exacting standards required for API synthesis.
We invite pharmaceutical partners to collaborate with us to optimize this synthesis route for your specific needs. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this enzymatic process for your supply chain. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and discuss route feasibility assessments tailored to your project timelines. Together, we can secure a reliable, cost-effective, and sustainable supply of critical nucleoside intermediates for the global market.
