Advanced Manufacturing of Chlorantraniliprole Intermediates via Optimized Catalytic Routes
The global demand for high-efficiency, low-toxicity insecticides continues to drive innovation in agrochemical manufacturing, particularly for ryanodine receptor modulators like chlorantraniliprole. Patent CN112552284B introduces a transformative preparation method that addresses critical bottlenecks in the traditional synthesis of this potent insecticide. By shifting the starting material from the notoriously difficult-to-source 2,3-dichloropyridine to the more accessible 2,3,6-trichloropyridine, this technology fundamentally alters the economic and operational landscape of production. The patent details a sophisticated multi-step sequence involving catalytic hydrazinolysis, selective hydrogenation, and a streamlined one-pot cyclization-bromination-hydrolysis protocol. For R&D directors and procurement specialists, this represents a significant opportunity to enhance purity profiles while simultaneously driving down the cost of goods sold through reduced step counts and simplified post-treatment operations. The integration of specialized catalysts, such as Cu-Ag/gamma-Al2O3, ensures high selectivity and yield, making this route highly attractive for commercial scale-up of complex agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chlorantraniliprole has been plagued by reliance on 2,3-dichloropyridine, a raw material characterized by harsh synthesis conditions, low availability, and prohibitive costs. Conventional routes, such as those disclosed in prior art patents, often necessitate the use of hazardous reagents like methylsulfonyl chloride and require tedious recrystallization and purification steps that generate substantial solid waste and wastewater. Furthermore, traditional methods frequently involve multiple discrete reaction steps for the construction of the pyrazole ring, including separate hydrazinolysis, Michael addition, cyclization, and oxidation stages. These fragmented processes not only extend the overall lead time for high-purity agrochemical intermediates but also introduce multiple points of failure where yield losses can accumulate. The reliance on solid oxidants like potassium persulfate in older methodologies further complicates waste management, creating significant environmental compliance burdens for manufacturing facilities striving for green chemistry standards.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages 2,3,6-trichloropyridine as a superior starting feedstock, effectively bypassing the supply chain vulnerabilities associated with 2,3-dichloropyridine. This strategic substitution is coupled with a highly efficient catalytic system that facilitates the direct formation of key hydrazino-intermediates with exceptional yields. The process innovates further by employing a one-pot methodology for the synthesis of the critical pyrazole carboxylic acid intermediate, where bromination and hydrolysis occur sequentially in the same reactor without the need for intermediate isolation. This consolidation of steps drastically simplifies the operational workflow, eliminating energy-intensive drying and filtration procedures that are typical in batch processing. By optimizing solvent systems to allow for oil-water phase separation rather than crystallization, the method achieves a level of process intensification that is rare in fine chemical synthesis, offering a robust pathway for cost reduction in insecticide manufacturing.
Mechanistic Insights into Catalytic Selectivity and One-Pot Cyclization
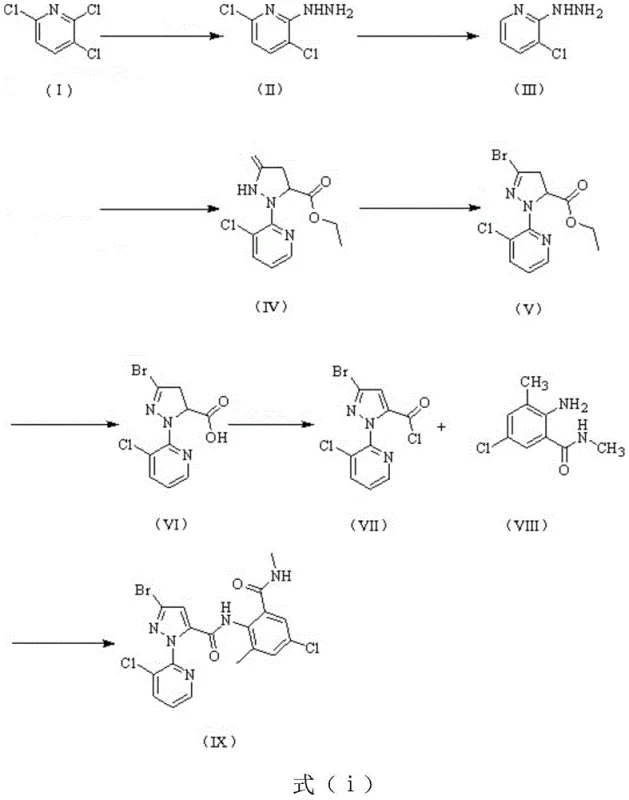
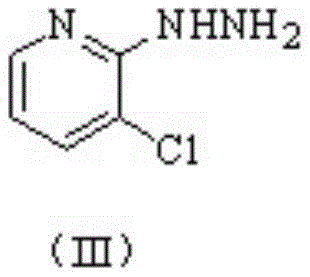
The core of this technological breakthrough lies in the precise control of catalytic selectivity during the hydrogenation and cyclization phases. The utilization of a bimetallic Cu-Ag/gamma-Al2O3 catalyst (Catalyst B) is pivotal for the selective dechlorination of 3,6-dichloro-2-hydrazinopyridine to form Intermediate I, chemically known as 3-chloro-2-hydrazinopyridine. This catalyst system is engineered to prevent over-reduction or unwanted side reactions that could compromise the integrity of the pyridine ring, ensuring that the chlorine atom at the 3-position remains intact while the 6-position is selectively reduced. The mechanistic efficiency is further enhanced by the use of specific phase transfer catalysts (Catalyst A), such as tetrabutylammonium chloride, which accelerate the initial hydrazinolysis reaction, shortening reaction times from over 20 hours in conventional methods to merely 4-6 hours. This acceleration is not merely a matter of speed but reflects a fundamental improvement in reaction kinetics that allows for tighter control over impurity profiles.
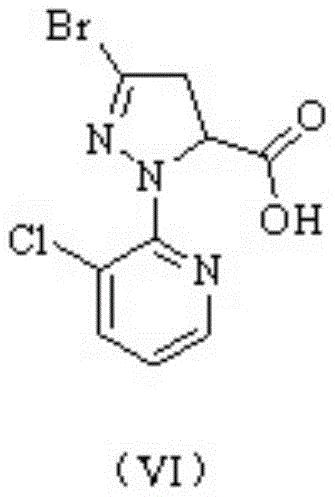
Following the formation of Intermediate I, the synthesis proceeds through a complex cascade involving cyclization with diethyl maleate and subsequent bromination. The patent highlights the critical role of solvent engineering in this stage, where the selection of specific solvent mixtures (e.g., dichloromethane and water) allows the intermediate ester to exist in an oil phase. This physical state enables direct oil-water phase separation, a technique that circumvents the need for traditional crystallization and suction filtration, thereby minimizing product loss and solvent retention. The subsequent conversion to Intermediate II, 3-bromo-1-(3-chloropyridin-2-yl)-4,5-dihydro-1H-pyrazole-5-carboxylic acid, is achieved through a seamless one-pot hydrolysis. The ability to manipulate the pH and temperature to induce phase separation or crystallization of Intermediate II provides manufacturers with flexible downstream processing options tailored to their specific equipment capabilities. This level of mechanistic understanding allows for the production of high-purity chlorantraniliprole precursors with minimal impurity carryover into the final amidation step.
How to Synthesize Chlorantraniliprole Efficiently
The synthesis of chlorantraniliprole via this patented route requires strict adherence to catalytic parameters and solvent protocols to maximize the benefits of the one-pot design. The process begins with the reflux of 2,3,6-trichloropyridine and hydrazine hydrate, followed by a controlled hydrogenation step that demands precise pressure management between 0.8-1.4 MPa. The subsequent cyclization and bromination steps are sensitive to pH adjustments and temperature gradients, necessitating real-time monitoring via HPLC to ensure complete conversion before proceeding to hydrolysis. For process engineers looking to implement this technology, the detailed standardized synthesis steps outlined below provide a comprehensive roadmap for replicating the high yields and purity levels reported in the patent examples. These guidelines are essential for translating laboratory-scale success into reliable commercial production.
- Synthesize 3,6-dichloro-2-hydrazinopyridine by reacting 2,3,6-trichloropyridine with hydrazine hydrate using a phase transfer catalyst.
- Perform selective hydrogenation using Cu-Ag/gamma-Al2O3 catalyst to obtain 3-chloro-2-hydrazinopyridine (Intermediate I).
- Execute a one-pot cyclization, bromination, and hydrolysis sequence to generate the key pyrazole carboxylic acid intermediate.
- Complete the synthesis by coupling the acid chloride derivative with substituted anthranilamide to form the final insecticide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic advantages that extend beyond simple yield improvements. The primary value driver is the substitution of the raw material backbone; by utilizing 2,3,6-trichloropyridine, manufacturers can decouple their supply chains from the volatile market dynamics surrounding 2,3-dichloropyridine. This shift not only secures a more stable flow of starting materials but also leverages a feedstock that is inherently more cost-effective due to its broader availability in the chloropyridine market. Furthermore, the elimination of hazardous reagents like methylsulfonyl chloride removes a significant regulatory and safety burden from the procurement portfolio, reducing the costs associated with handling, storage, and disposal of controlled substances. The streamlined nature of the process directly translates to lower operational expenditures, as fewer unit operations mean reduced labor, energy, and maintenance requirements across the production facility.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the consolidation of reaction steps and the elimination of expensive auxiliary reagents. By adopting a one-pot method for the formation of the pyrazole acid intermediate, the process removes the need for intermediate isolation, which traditionally consumes vast quantities of solvents and energy for drying and purification. The patent explicitly notes that compared to conventional industrial routes, this method saves a significant portion of raw material costs, largely due to the higher overall yield and the avoidance of costly oxidants like potassium persulfate. Additionally, the recovery and reuse of hydrazine hydrate solutions and the ability to recycle solvent fractions from the bromination step further compound these savings, creating a leaner and more financially resilient manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche intermediates with limited supplier bases. This new route mitigates that risk by relying on 2,3,6-trichloropyridine, a commodity chemical with a more robust and diversified global supply network. The simplification of the synthesis also reduces the dependency on complex, multi-step toll manufacturing arrangements, allowing for more agile in-house production or sourcing from a wider range of qualified contract manufacturers. The reduction in process complexity means that production schedules are less susceptible to delays caused by equipment bottlenecks or extended purification cycles, ensuring that delivery commitments to downstream formulators can be met with greater consistency and reliability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is designed for scalability with minimal ecological footprint. The avoidance of solid waste generation, particularly from oxidants and acid-binding agents, simplifies waste treatment protocols and reduces disposal costs. The tail gas profile is significantly cleaner, consisting mainly of hydrogen and hydrogen chloride, the latter of which can be captured and utilized for pH adjustment in earlier synthesis stages, creating a closed-loop system that minimizes emissions. This alignment with green chemistry principles not only facilitates easier permitting for capacity expansion but also enhances the brand value of the final agrochemical product in markets that increasingly prioritize sustainable sourcing and manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorantraniliprole synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting product in the global agrochemical market.
Q: Why is 2,3,6-trichloropyridine preferred over 2,3-dichloropyridine in this synthesis?
A: The use of 2,3,6-trichloropyridine overcomes the difficulties associated with obtaining 2,3-dichloropyridine, which often suffers from harsh synthesis conditions and low yields. This raw material substitution significantly improves the total reaction yield of the critical hydrazino-intermediate.
Q: How does the one-pot method improve the manufacturing process?
A: The one-pot method allows for the direct conversion of the pyrazole ester to the carboxylic acid intermediate without isolating the brominated ester. This eliminates multiple post-treatment steps such as crystallization, extraction, and drying, thereby reducing solvent consumption and operational time.
Q: What are the environmental benefits of this new catalytic route?
A: This route avoids the use of hazardous reagents like methylsulfonyl chloride and reduces solid waste by eliminating solid oxidants like potassium persulfate. Additionally, the tail gas consists mainly of clean hydrogen and hydrogen chloride, which can be absorbed and reused, minimizing environmental pollution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorantraniliprole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical industrial setting. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of monitoring the complex impurity profiles associated with pyrazole and pyridine chemistry. We are committed to delivering high-purity chlorantraniliprole intermediates that meet the exacting standards of global crop protection companies, leveraging our infrastructure to support both pilot-scale validation and full-scale commercial supply.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the specific economic benefits applicable to your operation. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and supply security. Together, we can drive the next generation of efficient and sustainable agrochemical manufacturing.
