Revolutionizing Prothioconazole Production: A Technical Deep Dive into Scalable Agrochemical Manufacturing
Introduction to Next-Generation Prothioconazole Manufacturing
The global demand for high-efficiency triazole fungicides continues to surge, driven by the critical need for crop protection in major agricultural sectors. Within this landscape, the patent CN113185470B represents a significant technological leap forward in the synthesis of prothioconazole, a broad-spectrum fungicide widely used for cereal and oilseed crops. This intellectual property addresses long-standing bottlenecks in chemical manufacturing, specifically targeting the issues of harsh reaction conditions, excessive operational steps, and suboptimal yields that have plagued previous synthetic routes. By introducing a novel one-pot boiling mode that strategically utilizes triethyl orthoformate followed by trimethylsilyl isothiocyanate, the inventors have established a pathway that is not only chemically elegant but also industrially robust. For R&D directors and supply chain leaders, this development signals a shift towards more sustainable and cost-effective production methodologies that align with modern green chemistry principles while maintaining rigorous quality standards.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for reliable agrochemical intermediate supplier networks seeking to optimize their portfolios. The transition from multi-step, hazardous processes to a streamlined, mild-temperature protocol reduces the barrier to entry for large-scale manufacturing. Furthermore, the specific choice of reagents and the precise control over the reaction sequence demonstrate a deep understanding of heterocyclic chemistry, ensuring that the final product meets the stringent purity specifications required by regulatory bodies worldwide. As we delve deeper into the technical specifics, it becomes evident that this methodology provides a competitive edge in the market for high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of prothioconazole has been fraught with significant operational challenges that hinder efficient commercial scale-up. The first generation of methods, such as those disclosed in US5789430, relied heavily on the use of n-butyllithium for hydrogen extraction under ultra-low temperature conditions, necessitating strict anhydrous and anaerobic environments that are both costly and dangerous to maintain on a metric-ton scale. Furthermore, these legacy processes often required a subsequent vulcanization step at extremely high temperatures, reaching up to 200°C, which poses severe safety risks and complicates reactor design. Alternative routes involving formaldehyde and sodium thiocyanate suffered from excessively long reaction sequences, resulting in lower total yields and the generation of substantial amounts of hazardous three wastes, thereby increasing environmental compliance costs. Another prevalent issue in prior art, specifically noted in comparative studies, involves the incorrect feeding sequence where thiocyanate is added prematurely, leading to increased isomer ratios and decreased main product selectivity due to the low activity of traditional thiocyanate salts.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in patent CN113185470B introduces a paradigm shift by employing a one-pot synthesis strategy that dramatically simplifies the operational workflow. The core innovation lies in the sequential addition of reagents: reacting the hydrazine precursor with triethyl orthoformate first to generate a key intermediate, followed by the introduction of trimethylsilyl isothiocyanate to effect ring closure. This approach effectively bypasses the need for cryogenic conditions or extreme thermal inputs, operating instead within a mild temperature range of 20°C to 45°C. The utilization of trimethylsilyl isothiocyanate as the sulfur source offers superior reactivity compared to inorganic thiocyanates, accelerating the reaction rate and minimizing side reactions. Consequently, this novel route achieves a remarkable improvement in overall yield and purity while significantly reducing the generation of waste, making it an ideal candidate for cost reduction in agrochemical intermediate manufacturing.
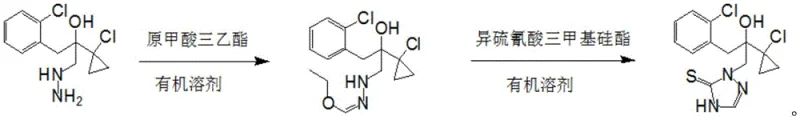
To visualize the superiority of this new pathway, one must examine the specific reaction dynamics compared to older methods. The patent data explicitly contrasts the new method against comparative examples where traditional reagents like ammonium thiocyanate or potassium thiocyanate were used in conjunction with formaldehyde. These older methods resulted in yields ranging merely from 50% to 78%, accompanied by lower purity profiles. The new method, however, leverages the high electrophilicity of the orthoformate and the specific nucleophilic properties of the silyl reagent to drive the reaction to completion with high selectivity. This fundamental change in chemical strategy eliminates the need for complex workup procedures between steps, thereby consolidating the production timeline and reducing the physical footprint required for manufacturing.
Mechanistic Insights into One-Pot Triazole Cyclization
The mechanistic elegance of this synthesis lies in the controlled formation of the 1,2,4-triazole-5-thione ring system through a carefully orchestrated sequence of nucleophilic attacks and cyclization events. Initially, the hydrazine moiety of the starting material, [1-(2-chlorophenyl)-2-(1-chlorocyclopropyl)-2-hydroxy]propylhydrazine, acts as a nucleophile attacking the electrophilic carbon of the triethyl orthoformate. This step generates an unstable amidine-like intermediate which is crucial for the subsequent ring closure. By performing this step first, the system ensures that the nitrogen atoms are correctly positioned and activated before the introduction of the sulfur source. The subsequent addition of trimethylsilyl isothiocyanate allows for the transfer of the thiocarbonyl group to the activated nitrogen center. The trimethylsilyl group acts as a potent leaving group, facilitating the attack of the nitrogen on the sulfur-carbon bond, which triggers the intramolecular cyclization to form the stable triazole ring. This mechanism avoids the formation of linear polymeric byproducts that often plague reactions involving simple thiocyanates and aldehydes.
From an impurity control perspective, the specific order of reagent addition is the critical factor determining the isomeric purity of the final product. In traditional methods where thiocyanate is present from the beginning, competitive reactions can occur at multiple sites on the hydrazine chain, leading to a mixture of regio-isomers that are difficult to separate. The patent data highlights that adding the orthoformate first creates a specific geometric constraint that directs the subsequent sulfur insertion exclusively to the desired position. This kinetic control minimizes the formation of structural isomers, resulting in a crude product with purity levels exceeding 95% even before recrystallization. For R&D teams, this implies a simplified purification train, potentially eliminating the need for column chromatography and relying solely on crystallization, which is far more scalable and economically viable for industrial production of complex agrochemical intermediates.
How to Synthesize Prothioconazole Efficiently
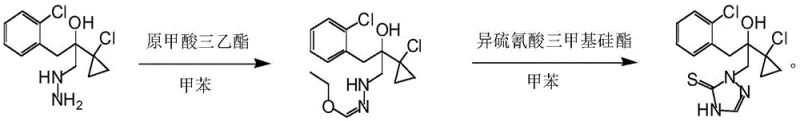
The practical implementation of this synthesis route requires precise adherence to the optimized parameters regarding temperature, stoichiometry, and addition rates to ensure reproducibility and safety. The process begins with the dissolution of the hydrazine precursor in a suitable organic solvent, with toluene being the preferred medium due to its ability to facilitate the reaction while allowing for easy product isolation via crystallization. The reaction is conducted under an inert atmosphere, typically nitrogen, to prevent oxidation of sensitive intermediates. The dropwise addition of triethyl orthoformate must be controlled to manage the exotherm, maintaining the reaction mass within the 20°C to 45°C window. Following the formation of the intermediate, the trimethylsilyl isothiocyanate is introduced slowly to ensure complete conversion without localized overheating. Detailed standardized synthesis steps see the guide below.
- Dissolve the hydrazine precursor [1-(2-chlorophenyl)-2-(1-chlorocyclopropyl)-2-hydroxy]propylhydrazine in an organic solvent like toluene under nitrogen protection.
- Add triethyl orthoformate dropwise at 20-45°C to form the intermediate, reacting for 0.5-3 hours.
- Introduce trimethylsilyl isothiocyanate dropwise to the reaction mixture, maintain temperature for 1-8 hours to complete cyclization, then cool to crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational efficiency and risk mitigation. The elimination of hazardous reagents like n-butyllithium and the removal of extreme temperature requirements significantly lowers the safety profile of the manufacturing process, reducing insurance premiums and the need for specialized cryogenic or high-temperature reactor infrastructure. This simplification of the process equipment requirements allows for greater flexibility in production scheduling and facilitates the rapid scale-up from pilot plant to commercial tonnage without the need for extensive capital expenditure on new machinery. Furthermore, the one-pot nature of the reaction drastically reduces the consumption of solvents and auxiliary materials, as there is no need for intermediate isolation and washing steps, leading to a leaner and more cost-effective material flow.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substantial increase in reaction yield coupled with the reduction in unit operations. By achieving higher conversion rates through the use of highly reactive trimethylsilyl isothiocyanate, the amount of raw material required per kilogram of finished product is significantly decreased. Additionally, the consolidation of multiple reaction steps into a single vessel reduces labor costs, energy consumption for heating and cooling cycles, and the overhead associated with cleaning and preparing reactors between batches. The qualitative improvement in yield means less waste is generated per unit of output, which directly correlates to lower disposal fees and a smaller environmental footprint, providing a dual benefit of economic savings and sustainability compliance.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as triethyl orthoformate and toluene ensures a robust supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents. The mild reaction conditions allow for continuous or semi-continuous processing options, which can dramatically shorten the lead time for high-purity agrochemical intermediates. By removing the dependency on ultra-low temperature logistics and the handling of pyrophoric materials, the supply chain becomes more resilient to disruptions and safer for logistics providers. This stability is crucial for meeting the just-in-time delivery demands of global agrochemical formulators who require consistent quality and timely availability of active ingredients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the exothermic profile is manageable and the reaction mixture remains homogeneous throughout the cycle, avoiding issues with mass transfer that often limit batch sizes in heterogeneous systems. The reduction in three wastes—specifically the minimization of aqueous waste streams containing heavy metals or inorganic salts—simplifies the wastewater treatment process and ensures easier compliance with increasingly stringent environmental regulations. This green chemistry approach not only future-proofs the manufacturing site against regulatory changes but also enhances the brand value of the end product by appealing to markets that prioritize sustainably sourced agricultural inputs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the new route. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists negotiating supply contracts based on these improved specifications.
Q: Why is the feeding sequence critical in prothioconazole synthesis?
A: Adding triethyl orthoformate before the sulfur source prevents the formation of unwanted isomers and ensures the correct cyclization pathway, significantly improving purity compared to traditional thiocyanate-first methods.
Q: What are the safety advantages of this new method over prior art?
A: This method eliminates the need for dangerous reagents like n-butyllithium and avoids extreme conditions such as ultra-low temperatures or high-temperature vulcanization at 200°C, making it safer for industrial scale-up.
Q: How does this process impact overall production costs?
A: By utilizing a one-pot procedure with higher reaction yields and reduced step counts, the process minimizes solvent usage, labor hours, and waste treatment costs, leading to substantial economic efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prothioconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113185470B are fully realized in practical application. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of prothioconazole meets the highest international standards. Our commitment to quality assurance means that we can consistently deliver high-purity intermediates that enable our clients to formulate effective and reliable crop protection solutions.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent and verified performance metrics. Together, we can drive the next generation of efficient and sustainable agrochemical manufacturing.
