Scalable Production of 2-Trifluoromethyl Quinazolinones Using Economical Iron Catalysts for Pharma
Scalable Production of 2-Trifluoromethyl Quinazolinones Using Economical Iron Catalysts for Pharma
The pharmaceutical industry continuously seeks robust and cost-effective synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery. Patent CN111675662B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing long-standing challenges in organic synthesis regarding catalyst cost and reaction efficiency. This technology leverages a readily available iron-based catalytic system to facilitate the cyclization of trifluoroethylimidoyl chloride and isatin derivatives, offering a sustainable alternative to traditional precious metal-catalyzed processes. For R&D directors and procurement managers, this innovation represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates while maintaining rigorous purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl groups has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. These conventional pathways are frequently plagued by severe reaction conditions that necessitate stringent temperature controls and specialized equipment, thereby increasing operational expenditures. Furthermore, the reliance on expensive fluorinating agents often results in narrow substrate scopes and inconsistent yields, creating bottlenecks in the production of diverse compound libraries needed for medicinal chemistry campaigns. The economic burden of these legacy methods is compounded by the difficulty in removing residual impurities, which can compromise the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN111675662B utilizes trifluoroethylimidoyl chloride and isatin as starting materials in a reaction catalyzed by inexpensive ferric chloride. This novel approach not only simplifies the operational procedure but also significantly broadens the functional group tolerance, allowing for the efficient synthesis of various substituted quinazolinones. By shifting from costly reagents to abundant iron catalysts, the process inherently lowers the barrier to entry for large-scale manufacturing. The reaction proceeds through a tandem sequence involving alkali-promoted bond formation followed by iron-catalyzed decarbonylation, ensuring high conversion rates under relatively mild conditions compared to prior art.
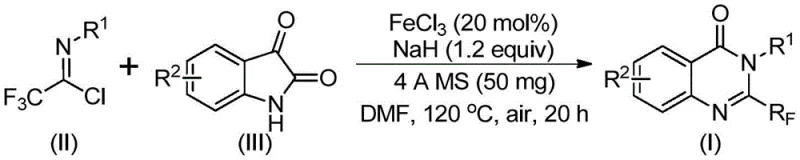
The visual representation of this transformation highlights the elegance of the design, where complex heterocyclic structures are assembled from simple precursors with high atom economy. This strategic shift in synthetic design directly addresses the pain points of cost and scalability that have hindered the widespread adoption of trifluoromethylated quinazolinones in drug development pipelines.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the ferric chloride and sodium hydride system. Initially, the strong base sodium hydride promotes the formation of a carbon-nitrogen bond between the trifluoroethylimidoyl chloride and the isatin substrate, generating a trifluoroacetamidine intermediate. Subsequently, the iron catalyst orchestrates a decarbonylation event that triggers the cyclization, effectively closing the ring to form the stable quinazolinone core. This dual-function catalytic system is remarkable for its ability to drive the reaction to completion without the need for exotic ligands or inert atmospheres, as the reaction can proceed efficiently even in the presence of air.

Understanding the specific roles of the reactants, such as the trifluoroethylimidoyl chloride shown above and the isatin derivative, is crucial for optimizing the reaction parameters. The presence of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging moisture, which is essential for maintaining the activity of the sodium hydride and preventing hydrolysis of the sensitive imidoyl chloride species. This careful control of the reaction environment ensures that side reactions are minimized, leading to a cleaner crude product profile that simplifies downstream purification efforts.
From an impurity control perspective, the mild nature of the iron catalysis prevents the degradation of sensitive functional groups that might occur under harsher acidic or basic conditions typical of older methods. The high selectivity of the cyclization step means that regioisomers and over-reacted byproducts are kept to a minimum, which is a critical factor for R&D teams focused on developing robust analytical methods for quality control. The ability to tolerate diverse substituents on the aromatic rings, including halogens and electron-donating groups, further underscores the versatility of this mechanistic pathway for generating diverse chemical libraries.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward mixing of reagents followed by a controlled heating ramp, eliminating the need for complex addition protocols or cryogenic conditions. Detailed standardized synthetic steps for replicating this high-yielding transformation are provided in the technical guide below, ensuring that process chemists can rapidly adopt this methodology for their specific targets.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- Stir the mixture at 40°C for 8 to 10 hours to initiate the alkali-promoted carbon-nitrogen bond formation.
- Heat the reaction mixture to 120°C and maintain for 18 to 20 hours to complete the iron-catalyzed decarbonylation and cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed process offers tangible benefits that extend beyond mere technical feasibility. The replacement of precious metal catalysts with commodity chemicals like ferric chloride fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with rhodium or palladium markets. Additionally, the use of commercially available starting materials ensures a stable supply chain, reducing the risk of production delays caused by raw material shortages. The simplicity of the workup procedure, which involves standard filtration and chromatography, further contributes to operational efficiency by reducing labor hours and solvent consumption.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with ferric chloride results in a drastic reduction in catalyst costs, which is a major component of the overall bill of materials for fine chemical synthesis. Furthermore, the high yields reported across a broad range of substrates mean that less raw material is wasted, improving the overall material throughput and reducing the cost per kilogram of the final product. The elimination of specialized ligands and the ability to run the reaction under air atmosphere also remove the need for costly inert gas systems and specialized glassware, leading to substantial capital expenditure savings.
- Enhanced Supply Chain Reliability: The reliance on isatin and trifluoroethylimidoyl chloride, which are widely produced and stocked by global chemical suppliers, mitigates the risk of supply disruptions that often plague proprietary or niche reagents. This abundance ensures that production schedules can be maintained consistently, even during periods of global market fluctuation. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality, such as minor fluctuations in cooling water temperature or nitrogen purity, further stabilizing the supply chain.
- Scalability and Environmental Compliance: The demonstrated scalability of this method to gram levels indicates a clear path toward ton-scale production, making it suitable for commercial API manufacturing. The use of iron, a non-toxic and environmentally benign metal, aligns with green chemistry principles and simplifies waste disposal protocols compared to processes generating heavy metal waste. This environmental compatibility reduces the regulatory burden and associated costs of waste treatment, facilitating smoother approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of using ferric chloride over precious metal catalysts?
A: Ferric chloride is significantly cheaper and more abundant than precious metals like palladium or ruthenium, drastically reducing raw material costs while maintaining high catalytic efficiency for this specific cyclization.
Q: Can this synthesis method accommodate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with methyl, halogen, methoxy, and nitro substituents without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly states the method is scalable to the gram level and potentially industrial scale due to simple operation, readily available starting materials, and straightforward post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards for safety and efficacy.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative synthesis method can enhance your competitive advantage in the global marketplace.
