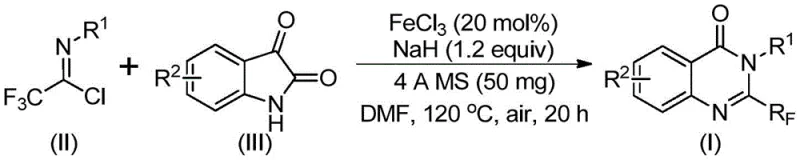
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization for Global Pharmaceutical Supply Chains
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly quinazolinones, which serve as critical scaffolds in drug discovery. Patent CN111675662B, published in late 2021, introduces a significant advancement in the preparation of 2-trifluoromethyl substituted quinazolinone compounds. This technology addresses long-standing challenges in heterocyclic synthesis by utilizing a cost-effective iron-catalyzed cyclization strategy. The introduction of the trifluoromethyl moiety is strategically vital, as it enhances the electronegativity, bioavailability, metabolic stability, and lipophilicity of the resulting molecular entities, making them highly desirable candidates for anti-cancer, anticonvulsant, and anti-inflammatory therapies. For R&D directors and procurement specialists, this patent represents a shift towards more sustainable and economically viable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While these methods are documented in literature, they suffer from significant drawbacks that hinder efficient commercial production. The reaction conditions are often severe, requiring harsh reagents that pose safety risks and complicate process control. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, can be prohibitively expensive and difficult to source in bulk quantities. These conventional routes frequently exhibit narrow substrate scope and low yields, leading to substantial material waste and increased production costs. For supply chain managers, reliance on such inefficient pathways creates vulnerabilities in the availability of key intermediates, potentially delaying drug development timelines and inflating the cost of goods sold.
The Novel Approach
In contrast, the methodology disclosed in CN111675662B offers a transformative solution by employing readily available trifluoroethylimidoyl chloride and isatin as starting raw materials. This novel route leverages a series of cyclization reactions catalyzed by cheap metal iron, specifically ferric chloride, which drastically reduces catalyst costs compared to precious metal alternatives. The process operates under relatively mild conditions, utilizing a two-stage temperature profile that ensures high conversion rates while maintaining functional group tolerance. By shifting the synthetic strategy to utilize abundant isatin derivatives, the method opens up a wide array of structural possibilities, allowing for the design of diverse quinazolinone libraries. This approach not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis, making it an attractive option for both laboratory-scale optimization and industrial manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the unique mechanistic pathway facilitated by the iron catalyst and the base promoter. During the reaction, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring closure. The presence of ferric chloride is critical, as it catalyzes the decarbonylation and cyclization steps required to isomerize the intermediate into the final stable 2-trifluoromethyl-substituted quinazolinone structure. This dual-function mechanism ensures that the reaction proceeds efficiently without the need for exotic ligands or complex catalytic systems. The use of sodium hydride as a base further drives the equilibrium forward, ensuring complete consumption of the starting materials and minimizing the formation of unreacted byproducts.
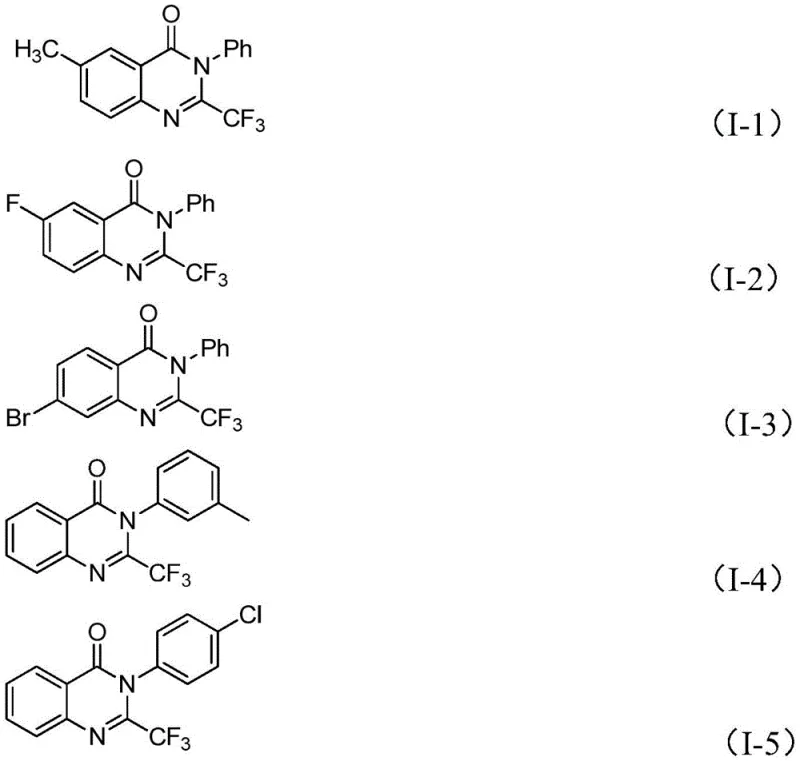
From an impurity control perspective, this mechanism offers distinct advantages for quality assurance teams. The high selectivity of the iron-catalyzed cyclization minimizes the generation of complex side products that are often difficult to separate in traditional methods. The patent data indicates excellent functional group tolerance, meaning that various substituents on the aryl rings (such as methyl, halogen, or methoxy groups) do not interfere with the catalytic cycle. This robustness is evidenced by the successful synthesis of multiple derivatives, including compounds (I-1) through (I-5), with consistently high yields. For R&D directors, this implies a cleaner crude reaction profile, which simplifies downstream purification and reduces the burden on analytical laboratories to identify and quantify trace impurities.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these results in a production environment. The process involves mixing ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF. The reaction is initiated at a lower temperature of 40°C for 8-10 hours to facilitate the initial coupling, followed by heating to 120°C for 18-20 hours to drive the cyclization to completion. Post-reaction processing is straightforward, involving filtration to remove solids, mixing with silica gel, and standard column chromatography purification. This detailed procedure ensures reproducibility and allows for precise control over reaction parameters, which is essential for maintaining batch-to-batch consistency in a GMP setting.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents compelling economic and logistical benefits. The shift away from expensive precious metal catalysts and harsh fluorinating agents directly translates to a reduction in raw material expenditure. Additionally, the use of commodity chemicals like isatin and simple aromatic amines ensures a stable and reliable supply chain, mitigating the risk of shortages associated with specialized reagents. The simplicity of the workup procedure, which avoids complex extraction or distillation steps, further contributes to operational efficiency and reduced utility consumption. These factors collectively enhance the cost-effectiveness of manufacturing high-purity pharmaceutical intermediates, allowing companies to maintain competitive pricing while ensuring supply continuity.
- Cost Reduction in Manufacturing: The utilization of ferric chloride as a catalyst represents a significant departure from costly noble metal systems, offering substantial savings on catalyst procurement. Since iron salts are abundant and inexpensive, the overall cost of goods is drastically lowered without compromising reaction efficiency. Furthermore, the ability to use excess amounts of relatively cheap aromatic amines ensures that the more valuable isatin substrate is fully converted, maximizing yield and minimizing waste. This economic efficiency is critical for scaling production volumes where even marginal savings per kilogram can result in significant financial impact.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including isatin and various substituted aromatic amines, are widely available from global chemical suppliers. This abundance reduces dependency on single-source vendors and shortens lead times for raw material acquisition. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations. For supply chain planners, this reliability ensures that production schedules can be met consistently, supporting just-in-time manufacturing strategies and reducing inventory holding costs.
- Scalability and Environmental Compliance: The protocol is designed with scalability in mind, having been demonstrated to work effectively from gram levels up to potential industrial scales. The use of iron, a non-toxic and environmentally benign metal, aligns with green chemistry principles and simplifies waste disposal compliance. Unlike processes generating heavy metal waste streams, this method produces effluent that is easier to treat, reducing the environmental footprint of the manufacturing facility. This alignment with sustainability goals not only meets regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the FeCl3-catalyzed method over traditional trifluoroacetylation?
A: The FeCl3-catalyzed method utilizes readily available isatin and trifluoroethylimidoyl chloride, avoiding the severe reaction conditions and expensive substrates like trifluoroacetic anhydride often required in conventional routes, resulting in higher yields and better functional group tolerance.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to simple operation, cheap catalysts, and convenient post-treatment procedures.
Q: What is the role of the trifluoromethyl group in these quinazolinone compounds?
A: The introduction of the trifluoromethyl group significantly improves the electronegativity, bioavailability, metabolic stability, and lipophilicity of the target molecule, enhancing its potential as a bioactive pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex heterocyclic intermediates. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into reliable industrial processes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest standards required for pharmaceutical applications. Our commitment to quality and consistency makes us an ideal partner for companies seeking to secure their supply of critical drug intermediates.
We invite you to collaborate with us to leverage this advanced iron-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with our superior manufacturing capabilities.
