Industrial Scale Production of Primary Amines via Selective Ammonia Formaldehyde Reaction
The chemical industry constantly seeks more efficient pathways to synthesize fundamental building blocks, and the production of primary amines remains a critical challenge due to selectivity issues. Patent CN101346345B introduces a groundbreaking method for producing primary amine compounds that addresses these longstanding inefficiencies by utilizing a unique combination of halogen compounds, ammonia, and formaldehyde. This innovative approach allows for the commercially advantageous production of primary amines while effectively suppressing the formation of unwanted secondary and tertiary amine by-products, a common plague in traditional ammonolysis reactions. By leveraging low-cost ammonia and a controlled reaction environment, this technology offers a robust solution for generating high-purity intermediates essential for pharmaceutical and agrochemical applications. The strategic use of formaldehyde acts as a transient directing group, fundamentally altering the reaction kinetics to favor mono-alkylation, thereby providing a significant competitive edge in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the selective synthesis of primary amines has relied on methods that are often economically and operationally burdensome for large-scale manufacturing. Traditional techniques such as the Gabriel reaction, which utilizes phthalimide, or the Delepine reaction using hexamethylenetetramine, involve expensive aminating reagents and require complex, multi-step decomposition operations to liberate the free amine. Furthermore, direct alkylation of ammonia with halogen compounds is notoriously difficult to control; once the primary amine is formed, it often acts as a stronger nucleophile than ammonia itself, leading to rapid over-alkylation and a messy mixture of secondary and tertiary amines. This lack of selectivity necessitates energy-intensive separation processes, such as fractional distillation, which drastically reduces overall yield and increases the carbon footprint of the manufacturing process. Consequently, finding a method that bypasses these thermodynamic pitfalls without resorting to costly protecting groups has been a primary objective for process chemists.
The Novel Approach
The method disclosed in the patent revolutionizes this landscape by introducing formaldehyde into the reaction matrix alongside ammonia and the halogen substrate. This three-component system facilitates the formation of a specific methyleneimine intermediate, which serves as a protected form of the primary amine that resists further alkylation. 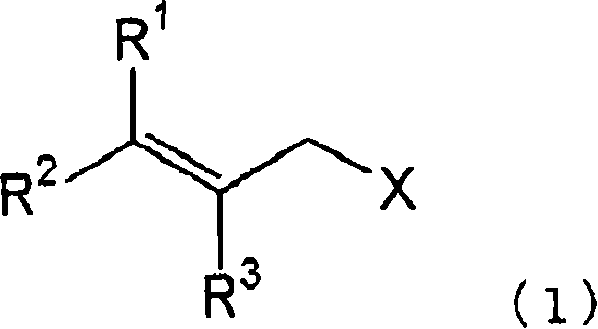 The process begins with the reaction of a halogen compound, represented by Formula (1) where X is a halogen and R groups vary, with ammonia and formaldehyde. This initial step effectively masks the nucleophilic nitrogen, preventing it from reacting further with the halogen compound to form di- or tri-substituted amines. Subsequent treatment of this intermediate with either an aqueous acid solution or hydroxylamine under acidic conditions cleanly cleaves the methylene group, releasing the desired primary amine in high purity. This strategy not only simplifies the synthetic route but also utilizes commodity chemicals, making it highly attractive for cost reduction in pharmaceutical intermediates manufacturing.
The process begins with the reaction of a halogen compound, represented by Formula (1) where X is a halogen and R groups vary, with ammonia and formaldehyde. This initial step effectively masks the nucleophilic nitrogen, preventing it from reacting further with the halogen compound to form di- or tri-substituted amines. Subsequent treatment of this intermediate with either an aqueous acid solution or hydroxylamine under acidic conditions cleanly cleaves the methylene group, releasing the desired primary amine in high purity. This strategy not only simplifies the synthetic route but also utilizes commodity chemicals, making it highly attractive for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Formaldehyde-Mediated Selective Amination
Understanding the mechanistic underpinnings of this reaction is crucial for optimizing yield and purity in a commercial setting. The core of this technology lies in the in situ generation of the methyleneimine species, depicted as Formula (3) in the patent documentation. 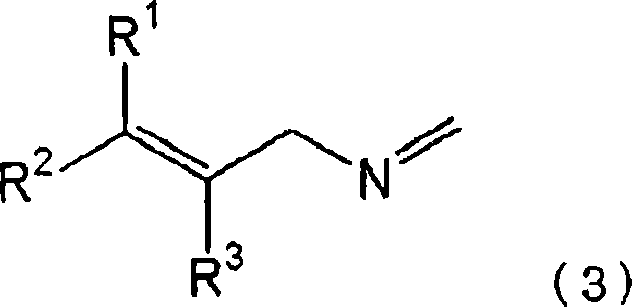 When the halogen compound reacts with ammonia in the presence of formaldehyde, the nitrogen atom attacks the electrophilic carbon of the formaldehyde, forming an imine linkage that stabilizes the mono-alkylated state. This intermediate is kinetically stable enough to prevent attack by a second equivalent of the halogen compound, effectively solving the poly-alkylation problem inherent to direct ammonolysis. The reaction typically proceeds in a sealed pressure reactor at temperatures ranging from 15°C to 100°C, often using alcohol solvents like methanol or ethanol which also serve to solubilize the gaseous ammonia. The precise control of stoichiometry, with ammonia preferably used in excess relative to formaldehyde, ensures that the equilibrium favors the formation of the desired intermediate over potential side products.
When the halogen compound reacts with ammonia in the presence of formaldehyde, the nitrogen atom attacks the electrophilic carbon of the formaldehyde, forming an imine linkage that stabilizes the mono-alkylated state. This intermediate is kinetically stable enough to prevent attack by a second equivalent of the halogen compound, effectively solving the poly-alkylation problem inherent to direct ammonolysis. The reaction typically proceeds in a sealed pressure reactor at temperatures ranging from 15°C to 100°C, often using alcohol solvents like methanol or ethanol which also serve to solubilize the gaseous ammonia. The precise control of stoichiometry, with ammonia preferably used in excess relative to formaldehyde, ensures that the equilibrium favors the formation of the desired intermediate over potential side products.
The second phase of the mechanism involves the decomposition of this methyleneimine intermediate to release the free primary amine. This can be achieved through two distinct pathways: acid hydrolysis or reaction with hydroxylamine. In the acid hydrolysis pathway, the addition of an aqueous acid such as hydrochloric or sulfuric acid cleaves the C=N bond. A critical aspect of this step is the management of the released formaldehyde; the patent suggests conducting this hydrolysis in the presence of a lower alcohol to convert the liberated formaldehyde into a dialkyl acetal. This scavenging of formaldehyde drives the hydrolysis equilibrium forward and prevents the formaldehyde from re-reacting with the newly formed amine. Alternatively, the hydroxylamine pathway reacts with the formaldehyde moiety to form formaldoxime or its trimer, which precipitates or remains in the aqueous layer, allowing for easy separation of the organic primary amine layer. Both mechanisms demonstrate exceptional impurity control, ensuring that the final product meets stringent purity specifications required for high-value applications.
How to Synthesize Primary Amines Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the formaldehyde mediation. The process generally involves charging a pressure reactor with the halogen compound, a source of formaldehyde such as paraformaldehyde or formalin, and an ammonia source, which can be liquid ammonia, aqueous ammonia, or an ammonia-alcohol solution. The mixture is heated, typically between 20°C and 90°C, to facilitate the formation of the methyleneimine intermediate. Following this incubation period, the reaction mixture is subjected to the decomposition step, either by adding acid directly or by introducing hydroxylamine sulfate under acidic conditions. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and workup protocols are essential for reproducibility and safety. For a comprehensive guide on the standardized synthesis steps and specific optimization parameters, please refer to the technical guide below.
- React a halogen compound with ammonia and formaldehyde in a solvent such as methanol at 15-100°C to form a methyleneimine intermediate.
- Treat the reaction mixture with an aqueous acid solution or react with hydroxylamine under acidic conditions to decompose the intermediate.
- Isolate the primary amine product through concentration, extraction, or crystallization, ensuring removal of formaldehyde byproducts.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational reliability. By shifting away from expensive, specialized aminating reagents like phthalimide or hexamine to commodity chemicals like ammonia and formaldehyde, manufacturers can achieve substantial cost savings in raw material acquisition. The elimination of complex protection and deprotection sequences associated with traditional methods significantly reduces the number of unit operations, leading to lower utility consumption and reduced labor costs. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-separate by-products, which simplifies downstream purification and increases the overall throughput of the production facility. These factors combine to create a more resilient and cost-effective supply chain for critical amine intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of high-cost reagents with low-cost ammonia and formaldehyde. Traditional methods often suffer from poor atom economy due to the stoichiometric waste generated by protecting groups; in contrast, this method generates minimal waste, primarily ammonium salts and acetals or oximes, which are easier to manage. The reduction in purification steps, such as the avoidance of extensive chromatography or high-vacuum distillation required to separate close-boiling amine mixtures, translates directly into lower processing costs. Additionally, the ability to run the reaction in common solvents like methanol or water further reduces solvent recovery expenses, making the overall process significantly more economical than conventional alternatives.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks such as ammonia and formaldehyde mitigates the risk of supply disruptions often associated with niche fine chemicals. These raw materials are produced on a massive global scale, ensuring consistent availability and price stability for long-term production planning. The robustness of the reaction conditions, which tolerate a variety of substituents on the halogen compound, allows for the flexible production of a wide range of primary amines using the same core infrastructure. This flexibility enables manufacturers to respond quickly to market demands for different intermediates without requiring significant retooling or new supplier qualification processes, thereby enhancing the agility of the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in pressure reactors that are standard equipment in the fine chemical industry. The use of aqueous workups and the potential to trap formaldehyde as less volatile acetals or oximes simplifies waste treatment and reduces the environmental burden compared to processes generating heavy metal waste or persistent organic pollutants. The high selectivity reduces the volume of organic waste streams, aligning with modern green chemistry principles and regulatory requirements. This ease of scale-up from laboratory to commercial production ensures that lead times for high-purity pharmaceutical intermediates can be minimized, supporting faster time-to-market for downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these details is vital for R&D teams assessing the route for new product development and for procurement officers evaluating supplier capabilities. The clarity provided here aims to bridge the gap between theoretical patent claims and practical industrial application.
Q: How does this method prevent the formation of secondary and tertiary amines?
A: The method utilizes formaldehyde to form a methyleneimine intermediate, which sterically and electronically directs the reaction pathway towards the primary amine upon hydrolysis, significantly suppressing poly-alkylation common in direct ammonolysis.
Q: What are the typical yields for allylamine production using this process?
A: According to patent examples, the yield of allylamine can reach 87.8% with minimal diallylamine (3.5%) and triallylamine (1.2%) byproducts when formaldehyde is used, compared to significantly lower selectivity without it.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses inexpensive and readily available raw materials like ammonia and formaldehyde, operates at moderate temperatures (15-100°C), and avoids complex purification steps associated with traditional methods like the Gabriel reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Primary Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient amine synthesis in the modern pharmaceutical value chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101346345B are realized in tangible commercial output. Our facilities are equipped with advanced pressure reactors and rigorous QC labs capable of meeting stringent purity specifications for complex amine intermediates. We understand that consistency and quality are paramount, and our team is prepared to adapt this selective amination technology to meet your specific project requirements, ensuring a reliable supply of high-quality materials for your drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating exactly how this method can improve your margins. We encourage you to contact us to request specific COA data for similar amine compounds and to discuss route feasibility assessments for your upcoming projects. By partnering with us, you gain access to not just a chemical supplier, but a strategic ally committed to driving innovation and efficiency in your production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
