Industrial Scale-Up of S-Nicotine: A Novel Glutarate-Based Synthetic Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing high-value chiral intermediates, particularly for the booming electronic cigarette and smoking cessation therapy markets. Patent CN113387925B, published in March 2023, introduces a groundbreaking preparation method for synthesizing S-nicotine starting from glutarate, offering a compelling alternative to traditional, cost-prohibitive routes. This innovation addresses critical pain points in modern chemical manufacturing by replacing expensive halogenated pyridines with widely available dicarboxylic acid esters. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the cost structure of nicotine production while maintaining rigorous purity standards. The disclosed methodology leverages a seven-step sequence that includes Claisen condensation, Hofmann degradation, and a highly stereoselective reduction, ultimately delivering S-nicotine with exceptional optical purity. By shifting the synthetic foundation to glutarate derivatives, manufacturers can bypass the volatility associated with brominated starting materials and achieve a more stable, predictable production cycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of nicotine has relied heavily on routes originating from 3-bromopyridine, a strategy that presents substantial economic and operational hurdles for large-scale manufacturers. As illustrated in the prior art reaction schemes, these conventional pathways often necessitate the use of organolithium reagents at cryogenic temperatures, typically around -78°C, to facilitate nucleophilic addition. Maintaining such extreme low-temperature conditions requires specialized refrigeration equipment and consumes vast amounts of energy, directly inflating the operational expenditure (OpEx) of the facility. Furthermore, 3-bromopyridine itself is a relatively high-cost starting material compared to simple aliphatic esters, and its supply chain can be susceptible to fluctuations in the bromine market. Another significant drawback of earlier methods, such as those utilizing cyclic imines or transition metal catalysts, is the requirement for high-pressure hydrogenation equipment, which introduces safety risks and necessitates expensive infrastructure investments. These factors collectively render many traditional synthetic routes economically unviable for the production of single-configuration, optically active nicotine required for premium applications.
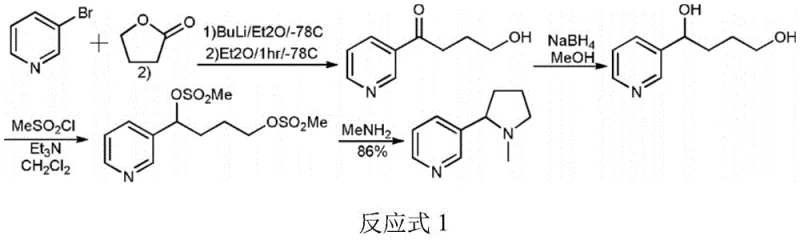
The Novel Approach
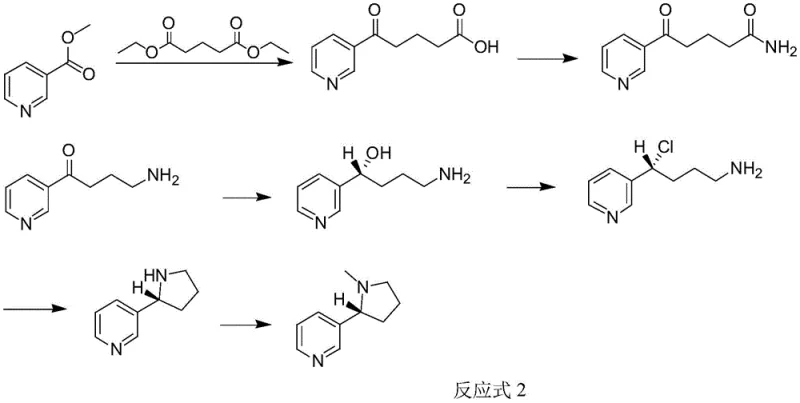
In stark contrast to the harsh conditions of legacy methods, the novel approach detailed in patent CN113387925B utilizes a glutarate-based backbone that fundamentally simplifies the synthetic architecture. This new route initiates with a Claisen condensation between nicotinate and diethyl glutarate, reactions that proceed efficiently at mild temperatures ranging from 0°C to 25°C. By eliminating the need for cryogenic cooling, the process dramatically reduces energy consumption and allows for the use of standard stainless steel reactors rather than specialized low-temperature vessels. The strategic selection of diethyl glutarate as a building block not only lowers raw material costs due to its commodity status but also streamlines the downstream purification processes. The subsequent steps, including amination and Hofmann degradation, are performed under atmospheric pressure and moderate thermal conditions, further enhancing the safety profile and scalability of the operation. This shift from a halogenated aromatic coupling strategy to an aliphatic chain extension strategy represents a paradigm shift in nicotine manufacturing, prioritizing operational simplicity and cost-efficiency without compromising on the stereochemical integrity of the final product.
Mechanistic Insights into Asymmetric Reduction and Chiral Control
The cornerstone of this synthetic achievement lies in the precise control of stereochemistry during the reduction phase, specifically in Step S4 of the disclosed process. The conversion of 4-amino-1-(pyridine-3-yl) butanone into the chiral alcohol intermediate is executed using (+)-B-diisopinocampheyl chloroborane, a powerful chiral reducing agent known for its high enantioselectivity. Mechanistically, this borane derivative coordinates with the carbonyl oxygen of the ketone substrate, creating a rigid transition state that directs the hydride delivery to a specific face of the molecule. This steric guidance ensures the formation of the (S)-enantiomer with an enantiomeric excess (ee) value reaching up to 98%, a critical specification for pharmaceutical-grade nicotine. The choice of tetrahydrofuran (THF) as the solvent in this step is also pivotal, as it stabilizes the boron complexes and facilitates the smooth progression of the reduction at 0°C. By securing the chiral center early in the sequence through this asymmetric reduction, the process avoids the need for difficult and yield-loss-prone resolution steps (such as chiral chromatography or salt formation) at the end of the synthesis, thereby maximizing overall throughput.
Impurity control is another vital aspect managed through the careful modulation of reaction parameters throughout the seven-step sequence. For instance, during the initial Claisen condensation, the pH of the system is meticulously adjusted to between 2 and 5 before the amination step to ensure the complete protonation of the intermediate acid, preventing side reactions that could lead to polymeric byproducts. Similarly, the Hofmann degradation step utilizes sodium hypochlorite under controlled temperature ramps (0°C to 71°C) to ensure clean rearrangement without over-oxidation of the sensitive pyridine ring. The final cyclization and methylation steps are conducted under basic conditions that favor intramolecular nucleophilic substitution, minimizing the formation of open-chain oligomers. This rigorous attention to reaction kinetics and thermodynamics ensures that the final S-nicotine product meets stringent purity specifications (≥95%) with minimal contamination from regioisomers or over-alkylated species, satisfying the rigorous quality demands of global regulatory bodies.
How to Synthesize S-Nicotine Efficiently
The synthesis of S-nicotine via this glutarate route offers a robust framework for laboratory and pilot-scale operations, providing a clear pathway from commodity esters to high-value chiral amines. The process begins with the condensation of methyl nicotinate and diethyl glutarate in the presence of sodium hydride, followed by a sequence of functional group transformations that build the pyrrolidine ring characteristic of nicotine. Detailed standard operating procedures for each stage, including precise molar ratios and temperature profiles, are essential for replicating the high yields reported in the patent data. While the general workflow is straightforward, attention to detail in the workup procedures, particularly during the extraction and drying phases, is crucial for maintaining product integrity. For a comprehensive breakdown of the exact experimental conditions and stoichiometry required to achieve optimal results, please refer to the standardized synthesis guide below.
- Perform Claisen condensation between nicotinate and glutarate using an alkaline catalyst like NaH to form 5-carbonyl-5-(pyridine-3-yl) valeric acid.
- Convert the acid to valeramide using ammonia, followed by Hofmann degradation with hypohalite to yield 4-amino-1-(pyridine-3-yl) butanone.
- Execute asymmetric reduction using (+)-B-diisopinocampheyl chloroborane to establish chirality, followed by chlorination, cyclization, and methylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this glutarate-based synthesis route offers transformative benefits that extend far beyond simple yield improvements. The primary advantage lies in the radical simplification of the raw material portfolio; by substituting expensive, specialty halogenated pyridines with bulk commodity chemicals like diethyl glutarate and methyl nicotinate, manufacturers can significantly insulate their production costs from the volatility of the fine chemical market. This substitution effect creates a more predictable cost of goods sold (COGS) structure, allowing for more competitive pricing strategies in the final API or intermediate market. Furthermore, the elimination of cryogenic processing requirements translates directly into reduced utility costs, as facilities no longer need to maintain energy-intensive low-temperature loops. The reliance on standard solvents like THF and ethyl acetate, which are readily available in multi-ton quantities globally, ensures that supply chain disruptions are minimized, guaranteeing continuity of supply even during periods of regional logistical stress.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of high-cost inputs and energy-intensive unit operations. By avoiding the use of expensive chiral transition metal catalysts and high-pressure hydrogenation equipment, the capital expenditure (CapEx) required to set up production lines is drastically lowered. Additionally, the mild reaction conditions (0-25°C) eliminate the need for specialized cryogenic reactors, allowing production to occur in standard glass-lined or stainless steel vessels. This reduction in equipment complexity not only lowers depreciation costs but also reduces maintenance overheads. The high atom economy of the Claisen condensation and the efficient Hofmann degradation further contribute to waste minimization, reducing the costs associated with effluent treatment and disposal. Collectively, these factors result in a leaner, more cost-effective manufacturing model that enhances profit margins without sacrificing quality.
- Enhanced Supply Chain Reliability: From a logistics perspective, the shift to glutarate derivatives strengthens the resilience of the supply chain. Diethyl glutarate and methyl nicotinate are produced on a massive scale for various industrial applications, ensuring a deep and diversified supplier base that mitigates the risk of single-source dependency. Unlike specialized chiral catalysts which may have long lead times and limited global availability, these starting materials can be sourced locally in most major chemical hubs. The simplified process flow also reduces the number of intermediate isolation steps, shortening the overall cycle time from raw material intake to finished goods. This agility allows manufacturers to respond more rapidly to fluctuations in market demand, ensuring that inventory levels can be adjusted dynamically without the bottleneck of complex, multi-week synthesis campaigns.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with the increasing regulatory pressure for green chemistry practices. The absence of heavy metal catalysts eliminates the risk of toxic metal residues in the final product, simplifying the purification process and reducing the burden on wastewater treatment facilities. The use of aqueous workups and common organic solvents facilitates easier solvent recovery and recycling, promoting a circular economy within the plant. Moreover, the mild thermal profile of the reaction reduces the generation of greenhouse gases associated with heating and cooling utilities. These environmental benefits not only ensure compliance with stringent international regulations like REACH and TSCA but also enhance the corporate sustainability profile, making the product more attractive to environmentally conscious downstream partners in the pharmaceutical and consumer goods sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel S-nicotine synthesis route. These insights are derived directly from the experimental data and beneficial effects described in patent CN113387925B, providing clarity on how this technology can be integrated into existing manufacturing frameworks. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this synthetic pathway.
Q: What are the key advantages of the glutarate route over traditional 3-bromopyridine methods?
A: The glutarate route utilizes significantly cheaper and more abundant raw materials compared to expensive 3-bromopyridine. Furthermore, it operates under mild temperatures (0-25°C) rather than harsh cryogenic conditions (-78°C), drastically reducing energy costs and equipment requirements for industrial scale-up.
Q: How is high optical purity (ee value) achieved in this synthesis?
A: High optical purity is secured during the asymmetric reduction step (Step S4) using (+)-B-diisopinocampheyl chloroborane. This chiral reducing agent selectively produces the (S)-enantiomer with an ee value reaching up to 98%, eliminating the need for complex resolution processes later in the workflow.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial viability. It avoids high-pressure hydrogenation equipment and expensive transition metal catalysts. The use of commodity chemicals like diethyl glutarate and standard solvents like THF ensures a robust and scalable supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO specializing in complex pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with the versatile reactor systems necessary to handle the specific conditions of the glutarate route, including precise temperature control for the asymmetric reduction step and efficient handling of volatile solvents. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of S-nicotine meets the highest standards of optical purity and chemical integrity, ready for immediate integration into your supply chain.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of S-nicotine that drives your business forward.
