Revolutionizing Beta-Amino Acid Production: A Scalable One-Step Hydrogenation Strategy for Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of novel peptidomimetics and stable peptide therapeutics has placed alpha-substituted beta-amino acids at the forefront of medicinal chemistry research. Unlike their natural alpha-amino acid counterparts, beta-amino acids offer enhanced metabolic stability and unique conformational properties, making them invaluable building blocks for next-generation drug candidates. However, the commercial viability of these compounds has historically been hindered by complex, multi-step synthetic routes that rely on expensive reagents and difficult purification processes. Patent CN1435408A, filed in 2003, introduces a transformative manufacturing protocol that addresses these critical bottlenecks through a streamlined one-step hydrogenation strategy. This technical insight report analyzes the proprietary methodology disclosed in the patent, evaluating its potential to redefine the supply chain dynamics for high-purity pharmaceutical intermediates. By shifting from laborious classical syntheses to a catalytic hydrogenation model, manufacturers can achieve substantial improvements in process efficiency and cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino acids with substituents at the alpha position has been plagued by significant operational inefficiencies and economic barriers. Prior art, such as the methods described by W. Knight and A. Avenoza, often relies on intricate reaction pathways like Claisen rearrangements or Diels-Alder cycloadditions. As illustrated in the reaction scheme below, these conventional approaches typically involve multiple discrete steps, each requiring specific reagents, stringent temperature controls, and extensive work-up procedures. For instance, the Diels-Alder route necessitates the use of specialized dienophiles and often results in complex stereochemical mixtures that are difficult to separate. Furthermore, many traditional protocols utilize stoichiometric amounts of organolithium reagents or expensive chiral auxiliaries, which not only drive up raw material costs but also generate substantial hazardous waste. The cumulative effect of these factors is a manufacturing process with low overall yield, high environmental impact, and limited scalability, rendering it unsuitable for the production of commercial quantities required by the global pharmaceutical market.
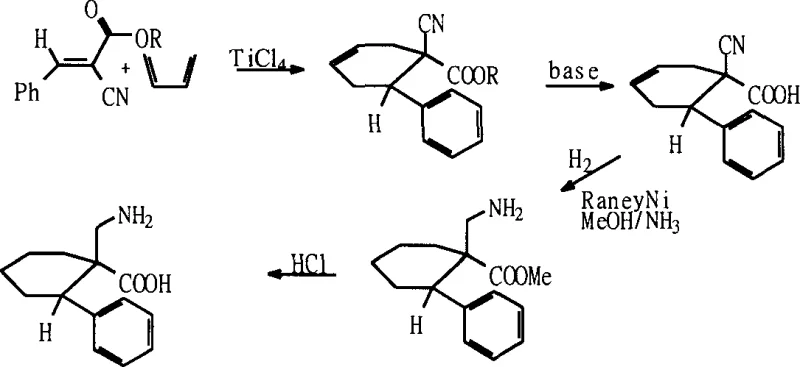
The Novel Approach
In stark contrast to the convoluted pathways of the past, the methodology outlined in CN1435408A presents a remarkably direct and efficient solution. The core innovation lies in the combination of a Knoevenagel-type condensation followed by a simultaneous catalytic hydrogenation. Instead of isolating unstable intermediates or employing precious metal catalysts, this process utilizes readily available aldehydes and ethyl cyanoacetate as starting materials. The reaction sequence proceeds through the formation of an alpha-cyano-alpha,beta-unsaturated ester, which is then subjected to hydrogenation using Raney Nickel. This transition metal catalyst is not only significantly cheaper than palladium or platinum but also demonstrates exceptional activity for the concurrent reduction of both the carbon-carbon double bond and the nitrile group. The result is a robust "one-pot" style transformation that dramatically shortens the synthetic timeline. By eliminating the need for multiple isolation steps and expensive reagents, this novel approach offers a clear pathway to cost reduction in pharmaceutical intermediate manufacturing while maintaining high product integrity.
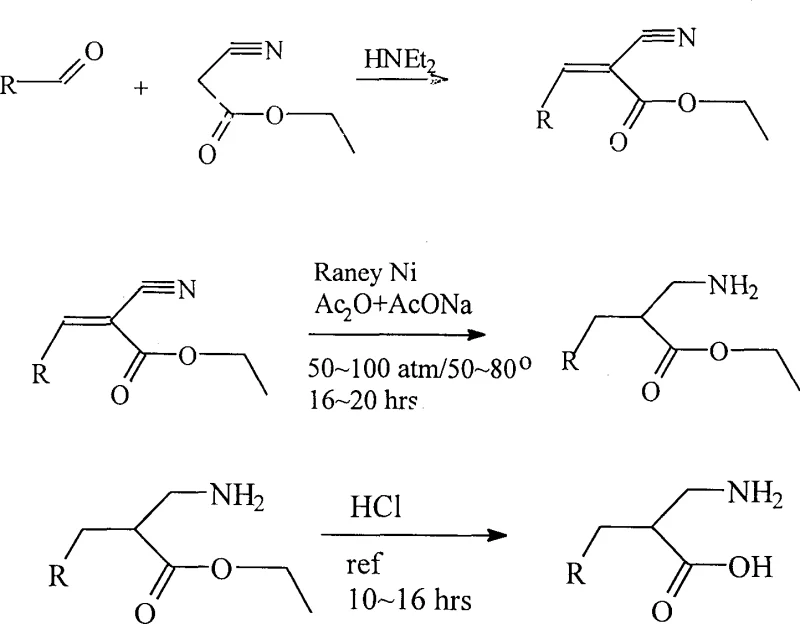
Mechanistic Insights into Raney Ni-Catalyzed Reductive Amination
The chemical elegance of this process is rooted in the dual functionality of the Raney Nickel catalyst under elevated hydrogen pressure. Mechanistically, the reaction begins with the base-catalyzed condensation of the aldehyde and ethyl cyanoacetate to form the unsaturated nitrile intermediate. Upon introduction of hydrogen gas at pressures ranging from 5 MPa to 8 MPa, the Raney Nickel surface facilitates the adsorption and activation of hydrogen atoms. These active species then attack the electron-deficient double bond of the unsaturated system, saturating the carbon backbone. Crucially, under the specific conditions employed—temperatures between 50°C and 80°C in the presence of acetic anhydride and sodium acetate—the nitrile group is simultaneously reduced to a primary amine. The inclusion of acetic anhydride serves a dual purpose: it acts as a solvent modifier and likely participates in the transient protection of the newly formed amine as an acetamide, preventing unwanted side reactions or polymerization during the vigorous hydrogenation phase. This synchronized reduction mechanism ensures that the final product is obtained with high regioselectivity and minimal formation of partially reduced by-products, a common issue in stepwise reduction protocols.
From an impurity control perspective, this mechanism offers distinct advantages for GMP manufacturing. Traditional methods often struggle with the removal of heavy metal residues or complex organic by-products generated during multi-step sequences. In this hydrogenation protocol, the catalyst (Raney Nickel) is heterogeneous, allowing for straightforward removal via filtration post-reaction. Furthermore, the use of acetic acid derivatives as additives ensures that any basic impurities are neutralized or converted into water-soluble salts during the subsequent hydrolysis step. The patent data indicates that the final hydrolysis with concentrated hydrochloric acid effectively cleaves the ester and any transient amide protections, yielding the free beta-amino acid. The simplicity of the work-up—neutralization, crystallization, and washing—means that the final API intermediate can achieve stringent purity specifications without the need for chromatographic purification, which is a major cost driver in fine chemical synthesis.
How to Synthesize Alpha-Substituted Beta-Amino Acids Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is divided into three critical stages: the initial condensation to form the precursor, the high-pressure hydrogenation, and the final hydrolysis. Operators must ensure that the condensation step is allowed sufficient time (3 to 24 hours) to ensure complete conversion of the aldehyde before proceeding to the high-pressure stage. The hydrogenation phase demands rigorous safety protocols due to the use of pyrophoric Raney Nickel and high-pressure hydrogen gas. Detailed standard operating procedures regarding catalyst handling, reactor pressurization, and temperature ramping are essential for safe scale-up. The following guide outlines the fundamental workflow derived from the patent examples, serving as a baseline for process development teams aiming to adopt this technology.
- Condense aldehyde with ethyl cyanoacetate using diethylamine in ethanol to form the unsaturated nitrile intermediate.
- Perform one-step hydrogenation using Raney Nickel catalyst at 50-80°C and 5-8 MPa pressure in the presence of acetic anhydride.
- Hydrolyze the resulting ester with concentrated hydrochloric acid under reflux, followed by neutralization and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1435408A represents a strategic opportunity to optimize the cost structure of amino acid derivative sourcing. The shift away from precious metal catalysts and complex multi-step syntheses directly translates to a more resilient and economical supply chain. By utilizing commodity chemicals like aldehydes and ethyl cyanoacetate, manufacturers can mitigate the risks associated with the volatility of specialty reagent markets. Moreover, the simplified processing requirements reduce the burden on utility consumption and waste treatment facilities, further enhancing the overall economic profile of the production line. This section details the specific commercial benefits that stakeholders can expect when transitioning to this manufacturing paradigm.
- Cost Reduction in Manufacturing: The most immediate impact of this process is the drastic reduction in catalyst costs. By replacing expensive noble metals with Raney Nickel, a widely available and inexpensive industrial catalyst, the direct material cost of the reaction is significantly lowered. Additionally, the ability to recover and reuse the catalyst after activation extends its lifecycle, compounding the savings over multiple batches. The elimination of chromatographic purification steps, which are solvent-intensive and time-consuming, further reduces operational expenditures related to solvent procurement and disposal. This lean manufacturing approach ensures that the final cost per kilogram of the beta-amino acid is competitive, providing a strong margin buffer for downstream drug development.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for exotic reagents. This synthesis route relies on bulk chemicals that are produced globally in massive quantities, ensuring a stable and diversified supply base. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, reducing the rate of batch failures. For supply chain heads, this translates to more predictable lead times and a lower risk of production stoppages. The scalability of the high-pressure hydrogenation step is well-understood in the chemical industry, allowing for seamless transition from pilot plant to full commercial production without the need for bespoke equipment.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route becomes a critical decision factor. This process generates fewer by-products and utilizes a catalyst that can be filtered and recycled, minimizing solid waste generation. The use of hydrogen as a clean reducing agent produces water as the primary by-product of reduction, aligning with green chemistry principles. Furthermore, the simplified work-up procedure reduces the volume of organic solvents required for extraction and purification. These factors collectively ease the burden on environmental health and safety (EHS) departments, facilitating faster regulatory approvals and ensuring long-term compliance with increasingly strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-amino acid synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of this route for their specific target molecules.
Q: What are the primary advantages of using Raney Nickel over noble metal catalysts for this synthesis?
A: According to patent CN1435408A, Raney Nickel offers a significant cost advantage over precious metals like Palladium or Platinum. Furthermore, the patent highlights that the catalyst is easy to prepare, recover, and reuse after activation, which drastically reduces the overall cost of goods sold (COGS) for large-scale manufacturing.
Q: Can this process accommodate diverse aromatic substituents on the beta-amino acid structure?
A: Yes, the methodology is highly versatile. The patent explicitly demonstrates successful synthesis using various aldehydes including benzaldehyde, 3,4-dichlorobenzaldehyde, p-tolualdehyde, and chlorobenzaldehyde. This indicates the process is robust enough to handle electron-withdrawing and electron-donating groups on the aromatic ring.
Q: How does the one-step hydrogenation improve impurity profiles compared to traditional methods?
A: Traditional methods often require separate reduction steps or complex protection/deprotection sequences, increasing the risk of side reactions. This patent utilizes a simultaneous reduction of both the olefinic bond and the cyano group in a single reactor operation. This 'one-pot' approach minimizes intermediate isolation steps, thereby reducing the accumulation of impurities and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Supplier
The technological advancements detailed in patent CN1435408A underscore the immense potential of catalytic hydrogenation in modern pharmaceutical synthesis. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory protocols into robust, commercial-scale manufacturing processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of meeting the most stringent purity specifications required for clinical and commercial API intermediates. We are committed to delivering high-purity beta-amino acids that adhere to global quality standards.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your time to market. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring your next-generation peptide therapeutics to life with efficiency, quality, and reliability.
