Revolutionizing Beta-Amino Acid Production: A Scalable One-Pot Synthetic Route for Industrial Applications
Revolutionizing Beta-Amino Acid Production: A Scalable One-Pot Synthetic Route for Industrial Applications
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for more efficient, cost-effective, and environmentally sustainable synthetic pathways for critical building blocks. Patent CN101525299B introduces a groundbreaking methodology for the synthesis of beta-amino acids, a class of compounds indispensable in the development of next-generation pharmaceuticals and agrochemicals. This technology addresses the longstanding challenges associated with low yields, complex purification protocols, and harsh reaction conditions that have plagued traditional production methods. By leveraging a novel one-pot strategy involving aldehyde, malonic acid, and benzylamine, this process achieves high conversion rates under mild conditions, representing a significant leap forward for any reliable beta-amino acid supplier seeking to optimize their portfolio. The implications for industrial scalability are profound, offering a robust solution for the commercial scale-up of complex amino acid derivatives that are essential for drugs like Sitagliptin and various antimicrobial agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino acids has been fraught with significant technical and economic hurdles that hinder efficient large-scale production. Traditional routes, such as those utilizing ethyl acetoacetate or requiring Boc-protection strategies, often involve multiple discrete steps that necessitate the isolation and purification of unstable intermediates. For instance, methods relying on oxime formation followed by Raney nickel reduction are not only operationally cumbersome but also generate substantial hazardous waste and utilize expensive, difficult-to-recycle catalysts. Furthermore, approaches involving Michael additions with protected amines frequently suffer from extremely low yields when applied to aliphatic substrates, limiting their utility to purely academic or small-scale laboratory settings. The reliance on silica gel column chromatography for purification in many of these legacy processes creates a severe bottleneck, making the transition from gram-scale research to ton-scale manufacturing economically unviable due to excessive solvent consumption and time delays.
The Novel Approach
In stark contrast to these inefficient legacy techniques, the methodology disclosed in the patent utilizes a streamlined cascade reaction sequence that dramatically simplifies the operational workflow. By employing benzylamine as a nitrogen source and protecting group simultaneously, the process enables a telescoped synthesis where the conjugated olefinic acid intermediate reacts directly without isolation. This one-pot design effectively merges the Knoevenagel condensation, decarboxylation, and Michael addition into a cohesive flow, significantly reducing the total processing time and equipment footprint required. The use of pyridine as a solvent and secondary amines as base catalysts creates an optimal environment for the reaction to proceed with high selectivity, avoiding the formation of complex byproduct mixtures. Consequently, the final N-benzyl beta-amino acid product precipitates directly from the reaction mixture upon cooling, allowing for recovery via simple suction filtration rather than energy-intensive distillation or chromatographic separation.
Mechanistic Insights into the Cascade Condensation and Hydrogenation Sequence
The core of this innovative synthetic route lies in the precise orchestration of a Knoevenagel condensation followed by a thermal decarboxylation and a subsequent Michael-type addition. Initially, the aldehyde reacts with malonic acid in the presence of a base catalyst, such as diethylamine or piperidine, to form a conjugated diene acid intermediate at temperatures ranging from -5°C to 50°C. Upon heating to between 35°C and 100°C, this intermediate undergoes spontaneous decarboxylation to yield the reactive conjugated olefinic acid species. This electrophilic species then engages in a nucleophilic attack by benzylamine at elevated temperatures of 50°C to 130°C, forming the stable N-benzyl beta-amino acid framework. The elegance of this mechanism is its ability to proceed sequentially in a single vessel, minimizing the exposure of reactive intermediates to degradation and ensuring high atom economy throughout the transformation.
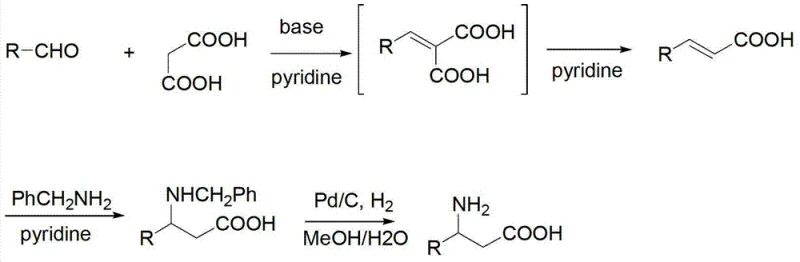
Following the construction of the carbon-nitrogen backbone, the final critical step involves the removal of the benzyl protecting group to reveal the free amine functionality. This is achieved through catalytic hydrogenation using palladium on carbon (Pd/C) under mild pressure conditions of 1 to 5 atmospheres. The choice of Pd/C is strategic, as it offers high activity for debenzylation while tolerating the carboxylic acid moiety without reduction, ensuring the integrity of the beta-amino acid structure. This step typically proceeds in a methanol/water mixed solvent system, which facilitates the solubility of the polar intermediates and allows for easy removal of the catalyst via filtration. The result is a high-purity free beta-amino acid with minimal impurity profiles, as the hydrogenation step effectively saturates any remaining olefinic impurities that might have carried over from the previous condensation stages, thereby enhancing the overall quality of the final API intermediate.
How to Synthesize Beta-Amino Acids Efficiently
To implement this sophisticated yet practical synthesis in a production environment, operators must adhere to strict control parameters regarding temperature ramping and reagent stoichiometry to maximize yield and safety. The process begins with the careful addition of base catalysts to the aldehyde and malonic acid mixture, followed by a controlled temperature increase to drive the decarboxylation before introducing the benzylamine. Detailed standard operating procedures regarding the specific molar ratios, such as the 1:1:1 ratio of aldehyde, malonic acid, and benzylamine, are critical for reproducibility. For a comprehensive breakdown of the exact experimental conditions, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Condense aldehyde and malonic acid with a base catalyst in pyridine at mild temperatures to form conjugated diene acid.
- Heat the intermediate to induce decarboxylation, yielding the conjugated olefinic acid without isolation.
- React the olefinic acid with benzylamine at elevated temperatures to form N-benzyl beta-amino acid.
- Perform catalytic hydrogenation using Pd/C to remove the benzyl group and obtain the free beta-amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic methodology offers transformative benefits that directly address the pain points of cost volatility and supply chain fragility in the fine chemicals sector. By eliminating the need for expensive protecting group reagents like Boc-anhydride and removing the requirement for chromatographic purification, the overall cost of goods sold (COGS) is substantially reduced. The ability to perform the reaction in a one-pot fashion significantly decreases the consumption of organic solvents and reduces the burden on waste treatment facilities, aligning with increasingly stringent environmental regulations. Furthermore, the simplicity of the isolation procedure, which relies on precipitation and filtration, enhances the throughput capacity of existing manufacturing assets without requiring capital-intensive new equipment installations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts like Raney nickel, which require specialized handling and disposal, alongside the removal of silica gel purification steps, leads to drastic operational expenditure savings. The use of commodity chemicals such as malonic acid and benzylamine as starting materials ensures a stable and low-cost raw material supply chain, insulating the production process from the price volatility often associated with specialized chiral auxiliaries or complex reagents. Additionally, the high overall yield of approximately 60% across the multi-step sequence minimizes material loss, further driving down the unit cost of the final high-purity beta-amino acids.
- Enhanced Supply Chain Reliability: Because the process utilizes widely available bulk chemicals and avoids reliance on bespoke catalysts that may have long lead times, the risk of supply disruption is significantly mitigated. The robustness of the reaction conditions, which tolerate a wide range of temperatures and pressures, ensures consistent batch-to-batch quality even in varying production environments. This reliability is crucial for maintaining continuous manufacturing schedules for downstream pharmaceutical clients who depend on just-in-time delivery of critical intermediates for their own drug substance production lines.
- Scalability and Environmental Compliance: The telescoped nature of the reaction reduces the number of unit operations, which inherently lowers the energy consumption and carbon footprint associated with the manufacturing process. The avoidance of chlorinated solvents and the use of aqueous workups in the final hydrogenation step simplify wastewater treatment and reduce the generation of hazardous organic waste streams. This green chemistry profile not only facilitates easier regulatory approval but also future-proofs the supply chain against tightening global environmental standards regarding industrial emissions and solvent residues.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, providing clarity for potential partners evaluating its feasibility for their specific applications. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for assessing the fit of this technology within your existing R&D pipeline or manufacturing infrastructure.
Q: What are the primary advantages of using benzylamine protection over Boc protection in beta-amino acid synthesis?
A: The benzylamine protection strategy described in patent CN101525299B eliminates the need for complex protecting group manipulations and harsh hydrolysis conditions often required for Boc groups. This approach allows for a true one-pot synthesis where intermediates do not require isolation, significantly reducing solvent usage and processing time compared to traditional multi-step protection strategies.
Q: Can this synthetic method be applied to aliphatic aldehydes effectively?
A: Yes, unlike previous methods such as EP1621529 which were limited primarily to aromatic aldehydes, this novel process demonstrates high efficacy with aliphatic aldehydes. The patent data confirms successful synthesis of various aliphatic beta-amino acids, including 3-aminopentanoic acid and 3-amino-4-methyl-pentanoic acid, with robust yields suitable for industrial scaling.
Q: How does the purification process impact the overall cost of goods for these intermediates?
A: The process drastically lowers costs by avoiding silica gel column chromatography, which is a major bottleneck in laboratory-to-plant translation. The N-benzyl beta-amino acid intermediate precipitates directly from the reaction system upon cooling, allowing for simple filtration and washing, thereby minimizing waste generation and maximizing throughput efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a promising patent to a commercially viable product requires deep technical expertise and robust manufacturing capabilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the laboratory are fully realized at the plant level. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including high-pressure hydrogenation reactors and advanced filtration systems, all governed by stringent purity specifications and rigorous QC labs to guarantee product consistency. We are committed to delivering high-purity beta-amino acids that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project, unlocking significant value through improved efficiency and reduced costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your timeline to market and strengthen your supply chain resilience.
