Advanced Asymmetric Hydrogenation for High-Purity Azepine Intermediates and Commercial Scale-Up
Advanced Asymmetric Hydrogenation for High-Purity Azepine Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable pathways for complex heterocyclic scaffolds, particularly those serving as critical backbones for potent kinase inhibitors. Patent CN1119332C presents a groundbreaking methodology for the preparation of azepine derivatives, specifically targeting compounds with significant biological activity such as Balanol and other phosphokinase inhibitors. This intellectual property outlines a sophisticated dual-route strategy involving either chemical asymmetric hydrogenation or microbial reduction to establish the necessary stereochemistry with exceptional precision. By leveraging transition metal catalysis with specialized chiral ligands, the disclosed process overcomes traditional limitations associated with racemic synthesis and subsequent resolution. For R&D directors and procurement specialists evaluating reliable azepine intermediate suppliers, this technology represents a pivotal shift towards more efficient, high-yielding manufacturing protocols that align with modern green chemistry principles and cost-reduction mandates.
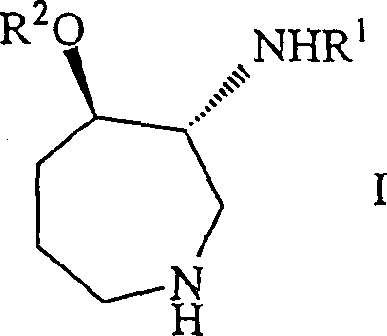
The core innovation lies in the ability to construct the seven-membered azepine ring with defined stereocenters at the 3 and 4 positions directly from simpler ketone precursors. The patent details the synthesis of general formula I compounds where R1 and R2 represent aromatic acyl groups, which are essential for the biological function of the final drug substance. Understanding the structural nuances of these intermediates is crucial for supply chain heads who must ensure the continuity of raw materials for downstream API synthesis. The versatility of this approach allows for the introduction of various substituents, including hydroxy, halogen, and alkoxy groups, providing a flexible platform for generating diverse libraries of analogues during the drug development phase. This adaptability ensures that the manufacturing process remains viable even as molecular structures are optimized for better efficacy or safety profiles in clinical trials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral azepine intermediates relied heavily on the resolution of racemic mixtures or the use of chiral pool starting materials, both of which suffer from inherent inefficiencies. Resolution processes typically cap the maximum theoretical yield at 50%, necessitating the recycling or disposal of the unwanted enantiomer, which drastically increases the cost of goods sold and environmental waste footprint. Furthermore, chiral pool synthesis is often restricted by the availability and price volatility of natural amino acids or sugars, creating supply chain vulnerabilities for large-scale production. Traditional reduction methods using non-chiral catalysts fail to discriminate between enantiotopic faces of the ketone substrate, resulting in racemic products that require additional, costly purification steps to meet the stringent enantiomeric purity standards required for pharmaceutical registration. These legacy methods also often involve harsh reaction conditions that can compromise the integrity of sensitive functional groups on the azepine ring, leading to complex impurity profiles that are difficult to characterize and control.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes asymmetric hydrogenation catalyzed by optically active complexes containing Group VIII metals, particularly rhodium and ruthenium, coordinated with atropisomeric diphosphine ligands. This method enables the direct conversion of achiral 4-oxo-azepine-3-carboxylates into chiral hydroxy-azepines with outstanding stereoselectivity. 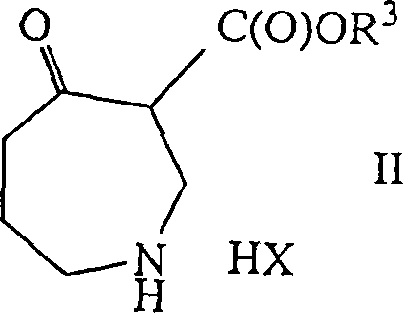
The process operates under relatively mild conditions, typically utilizing hydrogen pressures between 1 bar and 100 bar and temperatures ranging from 0°C to 80°C, which enhances operational safety and energy efficiency. By employing ligands such as MeOBIPHEP, BINAP, or BIPHEMP, the reaction achieves enantiomeric excess values exceeding 99%, effectively eliminating the need for downstream resolution. This catalytic efficiency translates directly into cost reduction in pharmaceutical manufacturing by maximizing atom economy and minimizing solvent usage. Additionally, the patent describes an alternative biocatalytic route using microbial strains like Hanseniaspora uvarum, offering a metal-free option that appeals to manufacturers seeking to avoid heavy metal residues in their final products. This dual-pathway flexibility provides a strategic advantage for supply chain resilience, allowing producers to switch between chemical and biological methods based on raw material availability and regulatory requirements.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise interaction between the transition metal center and the chiral ligand environment, which dictates the facial selectivity of the hydrogen addition. The catalyst system, often generated in situ from precursors like [Ru(OAc)2(R)-MeOBIPHEP], forms a coordinatively unsaturated species that binds the substrate through the carbonyl oxygen and the olefinic or aromatic interactions, locking it into a specific conformation. Upon exposure to hydrogen gas, the metal hydride species transfers hydrogen atoms to the re-face or si-face of the ketone with high fidelity, driven by the steric bulk and electronic properties of the ligand's biphenyl backbone. The patent highlights that the ratio of rhodium to ligand is critical, generally maintained between 0.5 to 2 moles per mole of ligand, to ensure the formation of the active catalytic species while preventing the aggregation of inactive metal clusters. Optimization of the substrate-to-catalyst ratio, which can range from 100 to 30,000, further demonstrates the robustness of the system, allowing for low catalyst loadings that reduce heavy metal costs and simplify purification workflows.
Impurity control is meticulously managed through the selection of reaction solvents and crystallization conditions that favor the precipitation of the desired trans-isomer over the cis-isomer. For instance, the use of ethanol or tetrahydrofuran during the workup phase facilitates the selective crystallization of the (3R, 4R) isomer, leaving the minor (3S, 4R) isomer in the mother liquor. This physical separation complements the kinetic selectivity of the catalyst, ensuring that the final intermediate meets rigorous purity specifications without the need for chromatographic purification. The subsequent protection of the amine functionality with a tert-butoxycarbonyl group and the conversion to an oxazolidinone via Curtius rearrangement serve as effective strategies to lock the stereochemistry and prevent racemization during subsequent acylation steps. Understanding these mechanistic details is vital for commercial scale-up of complex pharmaceutical intermediates, as it allows process engineers to identify critical process parameters (CPPs) that must be monitored to maintain consistent product quality across different batch sizes.
How to Synthesize Chiral Azepine Intermediates Efficiently
The synthesis of these high-value intermediates begins with the preparation of the 4-oxo-azepine-3-carboxylate substrate, followed by the critical asymmetric hydrogenation step that establishes the chiral centers. The process requires careful control of atmospheric conditions, specifically the exclusion of oxygen, to prevent catalyst deactivation and ensure reproducible reaction rates. Following hydrogenation, the crude product undergoes a series of transformations including protection, saponification, and cyclization to yield the versatile oxazolidinone scaffold.
- Perform asymmetric hydrogenation of the 4-oxo-azepine-3-carboxylate precursor using a rhodium or ruthenium catalyst with chiral diphosphine ligands like MeOBIPHEP under high hydrogen pressure.
- Protect the resulting amino group with a tert-butoxycarbonyl (Boc) group and saponify the ester to obtain the corresponding carboxylic acid intermediate.
- Convert the carboxylic acid into an oxazolidinone derivative via Curtius rearrangement, followed by hydrolysis and selective N- and O-acylation to yield the final active azepine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers tangible benefits that extend beyond mere technical feasibility. The elimination of resolution steps fundamentally alters the cost structure of the manufacturing process, removing the need for expensive chiral resolving agents and the associated loss of material. This efficiency gain results in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market. Furthermore, the use of widely available starting materials such as piperidine derivatives and common aromatic acids reduces dependency on niche suppliers, thereby enhancing supply chain reliability and mitigating the risk of shortages. The robustness of the hydrogenation process, which tolerates a wide range of substrates and conditions, ensures consistent output quality, reducing the frequency of batch failures and the associated financial penalties.
- Cost Reduction in Manufacturing: The asymmetric hydrogenation route significantly lowers production costs by achieving high yields and stereoselectivity in a single step, avoiding the material losses inherent in racemic resolution. The ability to use low catalyst loadings with high turnover numbers further reduces the expense associated with precious metals like rhodium and ruthenium. Additionally, the simplified downstream processing, which relies on crystallization rather than chromatography, decreases solvent consumption and waste disposal costs, contributing to a leaner and more economical manufacturing operation.
- Enhanced Supply Chain Reliability: By utilizing standard chemical feedstocks and established unit operations like high-pressure hydrogenation, the process minimizes supply chain bottlenecks. The availability of multiple ligand options and the possibility of switching to a biocatalytic route provide redundancy, ensuring that production can continue even if specific reagents face temporary scarcity. This flexibility is crucial for maintaining continuous supply to downstream API manufacturers and meeting strict delivery schedules without compromise.
- Scalability and Environmental Compliance: The process is designed for scalability, with reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The reduced generation of waste streams, particularly the avoidance of large volumes of mother liquor from resolution steps, aligns with increasingly stringent environmental regulations. This eco-friendly profile not only reduces compliance costs but also enhances the corporate sustainability image, which is becoming a key factor in supplier selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of azepine intermediates using the methods described in patent CN1119332C. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear understanding of the technology's capabilities and limitations.
Q: What is the primary advantage of the asymmetric hydrogenation method for azepine synthesis?
A: The primary advantage is the ability to directly generate chiral centers with high enantiomeric excess (ee > 99%) without the need for inefficient resolution steps, significantly improving overall yield and reducing waste.
Q: Can this process be scaled for commercial manufacturing of kinase inhibitors?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and robust catalysts like Rh-MeOBIPHEP complexes, making it highly suitable for commercial scale-up from kilogram to multi-ton production.
Q: What specific impurities are controlled during this synthesis?
A: The method effectively controls cis/trans isomer ratios and enantiomeric purity through optimized crystallization steps and the use of highly selective chiral ligands, ensuring stringent purity specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azepine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of new therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of azepine intermediate meets your exact requirements. Our commitment to technical excellence and regulatory compliance makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to contact our technical procurement team to discuss your specific needs and explore how our manufacturing capabilities can support your project goals. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into potential efficiencies and budget optimizations for your synthesis route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our dedication to quality and reliability as your preferred supplier of complex pharmaceutical intermediates.
