Advanced Simvastatin Intermediate Production: Overcoming Legacy Acylation Bottlenecks
Advanced Simvastatin Intermediate Production: Overcoming Legacy Acylation Bottlenecks
The pharmaceutical industry's relentless pursuit of efficient statin production has led to significant innovations in synthetic methodology, particularly for high-volume drugs like simvastatin. A pivotal advancement in this domain is detailed in Chinese Patent CN1215064C, which discloses a robust and streamlined process for preparing simvastatin intermediates. This technology addresses critical pain points associated with traditional acylation routes, specifically targeting the inefficiencies of acid chloride handling and the operational hazards of lithium bromide usage. For R&D directors and procurement strategists alike, understanding this shift from legacy methods to in-situ activation represents a major opportunity for optimizing the supply chain of HMG-CoA reductase inhibitors. By leveraging a novel activation system involving triphenylphosphine and halogen compounds, manufacturers can achieve superior conversion rates while drastically simplifying the purification landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key simvastatin intermediates relied heavily on methods described in earlier patents such as U.S. Patent No. 4,444,784. These conventional approaches typically necessitated the use of excess acid chloride derivatives under harsh conditions, including elevated temperatures and prolonged reaction times. Such parameters not only degraded the overall yield of the acylated product but also generated significant quantities of by-products, particularly through the unwanted removal of tert-butyldimethylsiloxy protecting groups. Furthermore, the separation of residual alcohol starting materials and unreacted acid chlorides from the final ester product proved to be a formidable chromatographic challenge, often hindering effective crystallization and compromising final purity specifications.
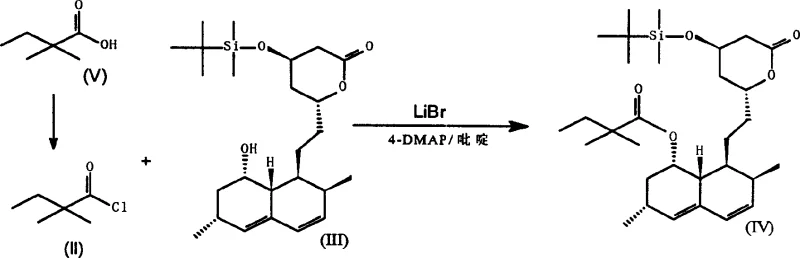
Subsequent attempts to improve upon these deficiencies, such as the method disclosed in U.S. Patent 4,845,237, introduced the use of alkali metals and 4-dialkylaminopyridine (4-DMAP) to activate the acid chloride. While this modification offered some improvements in yield, it introduced a new set of severe logistical burdens centered around the use of lithium bromide (LiBr). The handling of LiBr is notoriously difficult due to its extreme hygroscopic nature, requiring rigorous vacuum drying at 135°C for up to three days prior to use. If not meticulously dried, the presence of moisture leads to catastrophic drops in yield and increased impurity profiles. Additionally, the corrosive nature of wet LiBr necessitates specialized plastic processing equipment, adding capital expenditure and operational complexity to the manufacturing line.
The Novel Approach
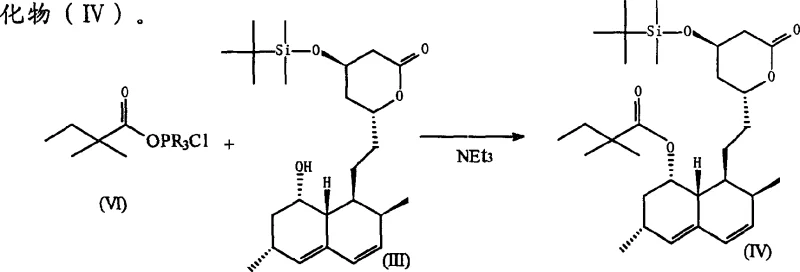
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN1215064C presents a paradigm shift by utilizing a carboxylic acid compound of Formula (VI) directly, bypassing the isolation of acid chlorides entirely. This innovative route employs an in-situ activation strategy where the carboxylic acid is activated by a phosphine derivative (PR3) and a halogen compound, such as hexachloroethane or carbon tetrachloride. This approach effectively eliminates the need for hazardous and difficult-to-handle lithium bromide, thereby removing the extensive drying protocols and specialized containment requirements associated with the prior art. By reacting the activated, unpurified carboxylic acid species directly with the hydroxylated lactone substrate, the process achieves remarkably high conversion efficiencies while maintaining a clean impurity profile, facilitating easier downstream processing and crystallization.
Mechanistic Insights into Triphenylphosphine-Halogen Mediated Acylation
The core chemical innovation driving this process lies in the generation of a highly reactive acylating species without isolating the corresponding acid chloride. As illustrated in the reaction mechanism, the carboxylic acid (VI) interacts with triphenylphosphine and a halogen source to form an activated intermediate, likely an acyloxyphosphonium salt or a mixed anhydride species, in situ. This activated complex is sufficiently electrophilic to attack the hydroxyl group of the lactone substrate (Formula III) under mild thermal conditions, typically ranging from 0°C to 110°C, with optimal performance observed around 83°C. The use of solvents such as ethylene dichloride or acetonitrile further stabilizes these intermediates, ensuring a homogeneous reaction environment that promotes rapid kinetics.
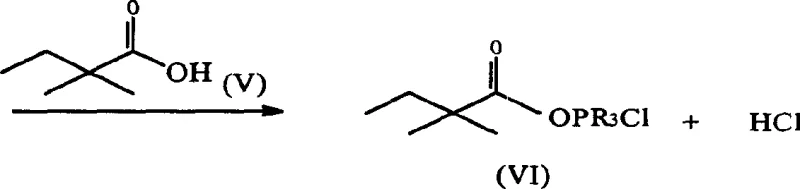
From an impurity control perspective, this mechanism offers distinct advantages over acid chloride routes. Because the activation occurs in the presence of the nucleophile (the lactone alcohol), the concentration of free, highly reactive acid chloride is kept negligible, minimizing side reactions such as self-polymerization or hydrolysis by trace moisture. The patent data indicates that when the activated carboxylic acid reacts with the alcohol substrate, the conversion rate of reactants reaches 99% or higher. Moreover, the ratio of the desired acylated product (IV) to by-products like unsaturated lactones is maintained at an impressive 96-97% versus 1-2%. This high selectivity is crucial for commercial manufacturing, as it reduces the burden on purification columns and recrystallization steps, directly impacting the cost of goods sold (COGS) and environmental waste generation.
How to Synthesize Simvastatin Intermediate Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and addition sequences to maximize the efficiency of the in-situ activation. The process generally begins with the preparation of the activating mixture, followed by the introduction of the carboxylic acid, and finally the coupling with the lactone substrate. Detailed operational parameters, including specific molar equivalents of triphenylphosphine and hexachloroethane, are critical for reproducibility. For a comprehensive guide on executing this transformation with industrial-grade consistency, please refer to the standardized protocol below.
- Activate the carboxylic acid derivative in situ using triphenylphosphine and a halogen compound like hexachloroethane in a solvent such as dichloroethane.
- React the activated species directly with the hydroxylated lactone substrate (Formula III) under controlled temperature conditions to form the acylated intermediate.
- Perform hydrolysis and purification steps, including acidification and crystallization, to isolate the final simvastatin compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this LiBr-free acylation technology translates into tangible operational resilience and cost optimization. The primary value driver is the elimination of the lithium bromide drying cycle, which previously consumed significant energy resources and occupied reactor capacity for multiple days. By removing this bottleneck, manufacturing throughput can be significantly increased, allowing for faster batch turnover and reduced lead times for high-purity pharmaceutical intermediates. Furthermore, the avoidance of corrosive salts reduces maintenance costs for reactor vessels and piping, extending the lifecycle of capital equipment.
- Cost Reduction in Manufacturing: The novel process eliminates the requirement for expensive and energy-intensive vacuum drying of lithium bromide, which previously necessitated heating at 135°C for three days. This removal of a multi-day unit operation drastically reduces utility consumption and labor hours per batch. Additionally, the high conversion rate minimizes the loss of valuable starting materials, ensuring that raw material costs are optimized through superior atom economy and reduced waste disposal fees associated with failed batches.
- Enhanced Supply Chain Reliability: By relying on stable carboxylic acids and common reagents like triphenylphosphine and hexachloroethane, the supply chain becomes less vulnerable to the availability fluctuations of specialized activators like LiBr. The simplified handling requirements mean that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized plastic containment, broadening the pool of eligible contract manufacturing organizations (CMOs) capable of producing this reliable simvastatin intermediate supplier grade material.
- Scalability and Environmental Compliance: The reduction in by-product formation, specifically the suppression of unsaturated lactone impurities, simplifies the wastewater treatment profile and reduces the volume of organic solvents required for purification. This aligns with increasingly stringent environmental regulations regarding solvent emissions and chemical waste. The robustness of the reaction at scale, demonstrated by high yields in pilot examples, ensures that commercial scale-up of complex pharmaceutical intermediates can proceed with minimal risk of batch failure or quality deviation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this acylation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this method is preferred for modern manufacturing.
Q: Why is the elimination of Lithium Bromide (LiBr) significant in this synthesis?
A: Eliminating LiBr removes the need for complex drying procedures (vacuum drying at 135°C for 3 days) and prevents moisture-induced yield losses, significantly simplifying the operational workflow and reducing equipment corrosion risks.
Q: What activation system replaces the traditional acid chloride method?
A: The process utilizes an in-situ activation system comprising triphenylphosphine (PR3) and a halogen compound such as hexachloroethane, which activates the carboxylic acid directly without isolating unstable acid chloride intermediates.
Q: What conversion rates are achievable with this novel acylation method?
A: Experimental data from the patent indicates that conversion rates of the reactants can reach 99% or higher, with the desired acylated product constituting 96-97% of the mixture, minimizing by-product formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN1215064C is essential for maintaining competitiveness in the global statin market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of simvastatin intermediate meets the exacting standards required by top-tier pharmaceutical clients, guaranteeing consistency and regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this LiBr-free acylation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies specific to your facility. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of high-purity pharmaceutical intermediates remains cost-effective and supply-secure.
