Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access nitrogen-containing heterocycles, particularly those bearing fluorine motifs which are critical for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses a long-standing challenge in organic synthesis by providing a direct route to these valuable scaffolds using a novel molybdenum and copper co-catalytic system. The relevance of this chemical structure cannot be overstated, as the 1,2,4-triazole core is a privileged structure found in numerous blockbuster drugs, including the antidiabetic agent Sitagliptin and the HIV inhibitor Maraviroc, as illustrated in the structural diversity of bioactive molecules below.
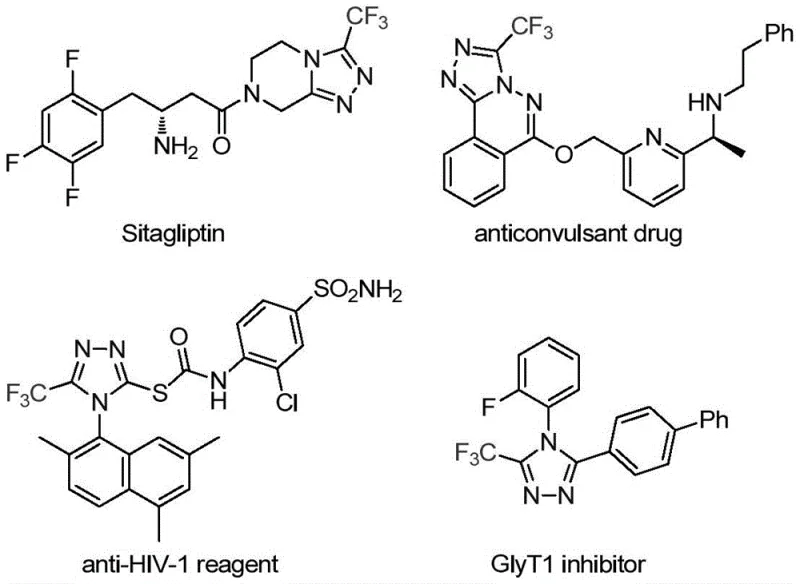
For R&D directors and process chemists, the ability to introduce a trifluoromethyl group regioselectively at the 3-position of the triazole ring opens up vast opportunities for lead optimization. The patent highlights that while many methods exist for fully substituted triazoles, efficient routes to 3,4-disubstituted variants remain scarce. This new methodology fills that gap by utilizing functionalized isonitriles and trifluoroethylimidoyl chloride as key building blocks. The reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C, avoiding the extreme temperatures or hazardous reagents often associated with traditional triazole synthesis. This technological advancement positions the process as a viable candidate for integration into complex API manufacturing workflows where purity and safety are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on synthetic strategies that are often fraught with operational difficulties and safety concerns. Traditional literature reports primarily describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical approaches frequently suffer from poor atom economy, requiring multiple steps to generate the necessary precursors, which inevitably drives up the cost of goods and increases waste generation. Furthermore, alternative copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane present significant safety hazards due to the explosive nature of diazo compounds, making them unsuitable for large-scale commercial production. The reliance on such unstable intermediates creates substantial supply chain risks and necessitates specialized equipment for safe handling, thereby limiting the accessibility of these valuable intermediates for broader pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN113307778A introduces a streamlined, one-pot cycloaddition strategy that fundamentally simplifies the manufacturing process. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary substrates, the reaction bypasses the need for hazardous diazonium species entirely. The core innovation lies in the synergistic use of molybdenum hexacarbonyl and cuprous acetate, which together facilitate the formation of the triazole ring with high efficiency. As depicted in the general reaction scheme below, this transformation allows for the direct assembly of the heterocyclic core from readily available starting materials. The operational simplicity is further enhanced by the use of common organic solvents like THF and the absence of stringent moisture-free requirements beyond standard Schlenk techniques, making this protocol highly attractive for process development teams aiming to reduce lead times for high-purity pharmaceutical intermediates.
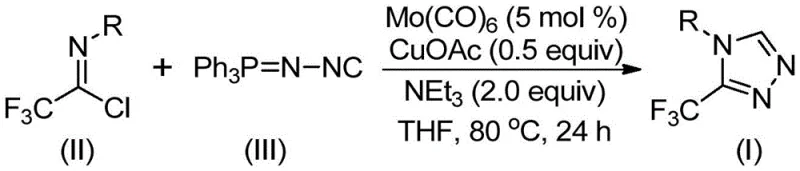
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring consistent quality in a GMP environment. The proposed mechanism suggests a sophisticated interplay between the two metal centers. Initially, molybdenum hexacarbonyl acts as a metal activator, coordinating with the functionalized isonitrile to form a reactive metal-isocyanide complex. This activation step is critical as it enhances the nucleophilicity of the isonitrile carbon, preparing it for the subsequent cyclization event. Following this activation, the copper species, specifically cuprous acetate, promotes a [3+2] cycloaddition reaction between the activated isonitrile complex and the trifluoroethylimidoyl chloride. This step constructs the five-membered triazole ring intermediate with high regioselectivity, ensuring that the trifluoromethyl group is positioned correctly at the 3-position of the ring.
The final stage of the catalytic cycle involves the elimination of triphenylphosphine oxide, driven by the presence of water within the system or during workup, to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This mechanistic pathway explains the broad substrate tolerance observed in the experimental data, as the metal coordination steps are generally less sensitive to electronic variations on the aromatic rings compared to purely organic cyclization mechanisms. For quality control purposes, this mechanism implies that impurity profiles will be dominated by unreacted starting materials or hydrolysis byproducts rather than complex rearrangement side-products, simplifying the purification strategy. The ability to tune the electronic properties of the R-group on the imidoyl chloride without compromising the cyclization efficiency underscores the versatility of this catalytic system for generating diverse libraries of analogues.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed to be straightforward, minimizing the need for specialized equipment while maximizing yield. The standard protocol involves charging a reaction vessel with the catalyst system and substrates in a defined molar ratio, typically favoring a slight excess of the isonitrile to drive the reaction to completion. The reaction mixture is then heated to a moderate temperature range of 70°C to 90°C for a duration of 18 to 30 hours. Post-reaction processing is equally simple, involving filtration to remove solid residues followed by standard silica gel column chromatography to isolate the pure product. This ease of operation significantly lowers the barrier to entry for laboratories looking to adopt this technology.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling advantages that directly impact the bottom line and operational resilience. The shift away from hazardous diazonium chemistry not only improves safety but also drastically simplifies the regulatory compliance landscape, reducing the administrative burden associated with handling controlled or explosive precursors. Furthermore, the reliance on commercially available starting materials such as aromatic amines and trifluoroacetic acid derivatives ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to原料 shortages. The robustness of the reaction conditions means that the process can be transferred to manufacturing sites with varying levels of technical infrastructure without requiring extensive re-validation or capital investment in specialized containment systems.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and abundant catalysts like cuprous acetate and molybdenum hexacarbonyl, which are used in relatively low loadings. By eliminating the need for expensive, custom-synthesized precursors or hazardous reagents that require special disposal protocols, the overall cost of goods sold is significantly reduced. Additionally, the high reaction efficiency and yield reported across a wide range of substrates mean that less raw material is wasted, further enhancing the cost-effectiveness of the manufacturing process. The simplified workup procedure also reduces solvent consumption and labor hours associated with purification, contributing to substantial operational savings.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including trifluoroethylimidoyl chloride and various functionalized isonitriles, are derived from commodity chemicals that are widely produced by the global chemical industry. This widespread availability ensures that supply chains are less vulnerable to geopolitical disruptions or single-source supplier failures. The ability to synthesize a wide variety of derivatives using the same core protocol allows manufacturers to maintain a flexible inventory strategy, producing different intermediates on the same production lines with minimal changeover time. This flexibility is crucial for responding rapidly to fluctuating market demands in the pharmaceutical sector.
- Scalability and Environmental Compliance: The patent data confirms that the reaction performs well on the gram scale, indicating a clear path towards kilogram and ton-scale production. The mild reaction temperatures and the use of THF, a solvent with well-established recovery and recycling protocols, align with green chemistry principles. By avoiding the generation of heavy metal waste associated with stoichiometric oxidants or the toxic byproducts of diazonium decomposition, this method facilitates easier waste treatment and lower environmental compliance costs. The scalability is further supported by the homogeneous nature of the catalytic system, which ensures consistent heat and mass transfer characteristics when scaling up from laboratory reactors to industrial vessels.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope and optimization. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development planning. Understanding these nuances is essential for successfully integrating this methodology into existing manufacturing pipelines.
Q: What represents the key advantage of this Mo/Cu co-catalytic system over traditional methods?
A: Unlike traditional methods requiring harsh cyclization conditions or unstable diazonium salts, this method utilizes cheap, commercially available starting materials under mild thermal conditions (70-90°C) with high functional group tolerance.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the reaction efficiency remains high and the operation is simple enough to be expanded from milligram scale to gram-level reactions, indicating strong potential for commercial scale-up.
Q: What solvents are compatible with this trifluoromethyl-triazole synthesis?
A: While various aprotic solvents can dissolve the raw materials, tetrahydrofuran (THF) is identified as the preferred organic solvent due to its ability to effectively promote the reaction and ensure high conversion rates compared to acetonitrile or DMF.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the Mo/Cu co-catalytic pathway described in CN113307778A and is prepared to leverage this technology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to commercial supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific drug candidates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both your development timeline and budget.
