Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
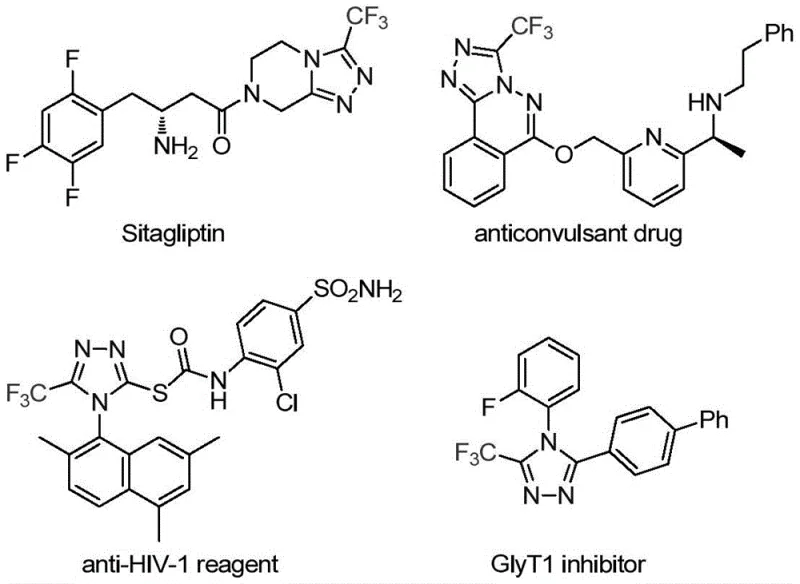
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a significant advancement in this domain by disclosing a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural scaffolds are ubiquitous in modern medicinal chemistry, serving as key intermediates for blockbuster drugs such as Sitagliptin and various antifungal agents. The disclosed technology leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. This approach not only addresses the synthetic challenges associated with introducing the trifluoromethyl group but also offers a streamlined pathway that is highly attractive for industrial scale-up. By operating under relatively mild thermal conditions and utilizing readily accessible reagents, this invention represents a strategic leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status for complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant operational and safety hurdles that impede efficient cost reduction in API manufacturing. Traditional synthetic routes often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently require harsh reaction conditions and generate substantial chemical waste. Furthermore, alternative strategies involving copper-catalyzed multi-component reactions utilizing diazonium salts pose severe safety risks due to the explosive nature of diazonium intermediates, necessitating specialized equipment and rigorous safety protocols that drive up operational expenditures. Another common pathway involves the reaction of trifluoroethylimidoyl chloride with aldehyde hydrazones and hydrazides, which often suffers from limited substrate scope and poor atom economy. These legacy methods typically involve multiple synthetic steps, difficult purification procedures, and the use of expensive or unstable reagents, creating bottlenecks in the commercial scale-up of complex pharmaceutical additives and intermediates.
The Novel Approach
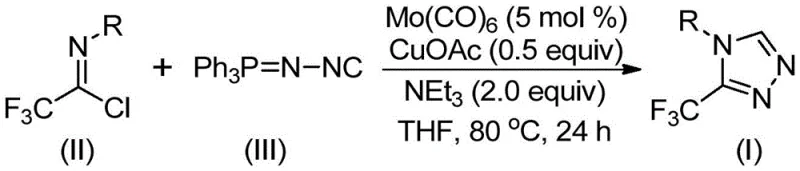
In stark contrast to these cumbersome traditional pathways, the methodology described in patent CN113307778A offers a transformative solution by employing a direct cycloaddition strategy mediated by a dual metal catalyst system. This innovative route utilizes trifluoroethylimidoyl chloride and functionalized isonitrile (specifically Ph3P=N-NC) as the primary building blocks, reacting them in the presence of molybdenum hexacarbonyl and cuprous acetate. The process is characterized by its operational simplicity, requiring only a single reaction vessel where all components are mixed in an organic solvent such as tetrahydrofuran (THF). The reaction proceeds efficiently at moderate temperatures ranging from 70°C to 90°C over a period of 18 to 30 hours, eliminating the need for cryogenic conditions or high-pressure reactors. This streamlined approach significantly reduces the number of unit operations required, thereby minimizing solvent consumption and energy usage while maximizing overall yield. The ability to directly access diverse 3-trifluoromethyl-1,2,4-triazole derivatives through simple substrate design makes this method exceptionally versatile for generating libraries of bioactive compounds.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The efficacy of this synthetic transformation relies on the intricate interplay between the molybdenum and copper catalysts, which orchestrate the activation of distinct reaction partners to facilitate ring closure. Mechanistically, the molybdenum hexacarbonyl serves as a crucial activator for the functionalized isonitrile, likely forming a transient metal-isocyanide complex that enhances the nucleophilicity or electrophilicity of the carbon center. Concurrently, the cuprous acetate acts as a promoter for the [3+2] cycloaddition event, coordinating with the trifluoroethylimidoyl chloride to lower the activation energy barrier for the formation of the five-membered triazole ring. This cooperative catalysis ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 3-trifluoromethyl isomer over potential byproducts. Following the initial cycloaddition, the intermediate undergoes a elimination of triphenylphosphine oxide, driven by the presence of water or moisture within the system, to yield the final aromatic triazole product. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction parameters for specific substrates.
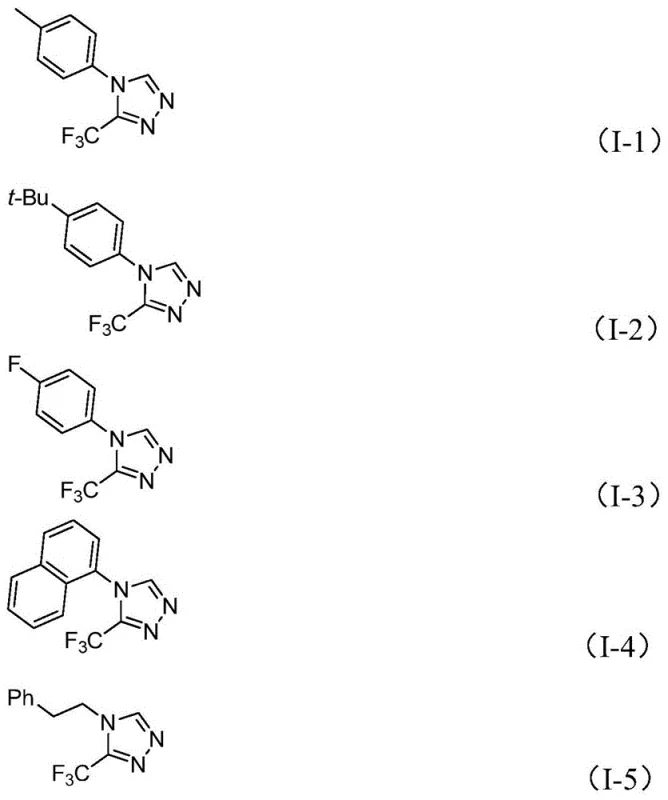
From an impurity control perspective, the choice of reagents and the specific catalytic cycle play a pivotal role in ensuring high product purity. The use of molecular sieves in the reaction mixture helps to manage water content, preventing hydrolysis of the sensitive imidoyl chloride starting material while still allowing for the necessary elimination step. The robustness of the Mo/Cu system tolerates a wide array of functional groups, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, and nitro substituents on the aromatic ring. This broad tolerance minimizes the formation of side products derived from functional group incompatibility, a common issue in heterocycle synthesis. Consequently, the crude reaction mixtures are often cleaner, simplifying the downstream purification process which typically involves standard filtration and silica gel column chromatography. This level of control over the reaction profile is essential for meeting the stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis protocol is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment, making it an ideal candidate for technology transfer. The process begins with the precise weighing and addition of the catalytic system and reagents into a reaction vessel, followed by the introduction of the solvent. The reaction is then heated to the specified temperature range and monitored until completion, after which a simple workup procedure isolates the target molecule. For detailed operational parameters regarding stoichiometry, specific temperature ramps, and purification techniques, please refer to the standardized synthesis guide below.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical yield. The primary advantage lies in the accessibility and cost-effectiveness of the starting materials; trifluoroethylimidoyl chloride and the functionalized isonitrile precursor are either commercially available or can be synthesized from inexpensive bulk chemicals like aromatic amines and trifluoroacetic acid. This reliance on commodity-grade feedstocks insulates the supply chain from the volatility often associated with specialized, high-cost reagents, ensuring a more stable and predictable sourcing strategy. Furthermore, the elimination of hazardous diazonium salts removes the need for expensive safety infrastructure and specialized waste disposal services, contributing to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot reaction significantly reduces the operational complexity typically associated with multi-step heterocycle synthesis. By consolidating the bond-forming events into a single thermal process, manufacturers can drastically cut down on solvent usage, labor hours, and reactor occupancy time. The absence of cryogenic conditions or high-pressure requirements further lowers energy consumption, while the use of relatively inexpensive copper and molybdenum catalysts avoids the financial burden of precious metals like palladium or rhodium. These factors collectively drive down the cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the wide availability of reagents contribute to a highly resilient supply chain. Unlike methods dependent on unstable intermediates that must be generated in situ and used immediately, this process utilizes stable starting materials that can be stockpiled without significant degradation. This stability allows for better inventory management and reduces the risk of production delays caused by reagent spoilage or shipping constraints. Additionally, the high tolerance for functional groups means that a single standardized protocol can be applied to synthesize a diverse range of derivatives, simplifying production planning and reducing the need for multiple specialized manufacturing lines.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated to work effectively from gram-scale experiments to larger batch sizes without loss of efficiency. The post-processing steps are conventional and easily adapted to industrial continuous flow or batch reactors, facilitating the commercial scale-up of complex pharmaceutical additives. Moreover, the reaction generates fewer hazardous byproducts compared to traditional hydrazine-based routes, aligning with increasingly strict environmental regulations. The simplified waste stream, primarily consisting of organic solvents and metal salts that can be recovered or treated, supports sustainable manufacturing practices and reduces the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this triazole synthesis method. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering practical guidance for chemists and engineers evaluating this technology for their own production needs.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using THF as the preferred solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a dual catalyst system of Mo(CO)6 and CuOAc.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with substituents such as methyl, methoxy, fluoro, chloro, and nitro groups on the aromatic ring.
Q: Is this process suitable for large-scale manufacturing?
A: The methodology is designed for scalability, utilizing cheap and commercially available starting materials and simple post-processing steps like filtration and column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay every batch. Our capability to implement advanced catalytic technologies, such as the Mo/Cu co-catalyzed system described here, positions us as a strategic partner capable of solving complex synthetic challenges while maintaining cost competitiveness.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project requirements. Whether you need custom synthesis of novel triazole derivatives or large-scale supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can accelerate your drug development timeline.
