Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability in modern drug candidates. A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from conventional multi-step syntheses to a streamlined, one-pot cycloaddition strategy co-catalyzed by molybdenum and copper. The relevance of this chemical scaffold cannot be overstated, as the 1,2,4-triazole core is a privileged structure found in numerous blockbuster medications, including antidiabetic agents like Sitagliptin and antiviral drugs, where the trifluoromethyl group plays a pivotal role in modulating lipophilicity and electronic properties.
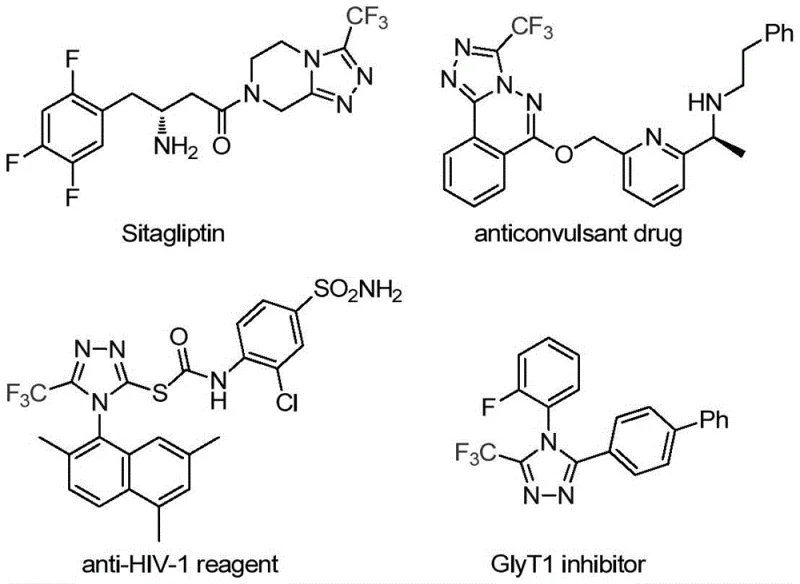
For R&D Directors and Process Chemists, the ability to access these complex heterocycles through a mild, transition-metal catalyzed pathway offers substantial advantages in terms of impurity profile control and process safety. The patent outlines a reaction system that operates effectively at moderate temperatures between 70°C and 90°C, utilizing readily available starting materials such as trifluoroethylimidoyl chloride and functionalized isonitriles. This approach not only circumvents the use of unstable diazonium salts or hazardous hydrazine intermediates often required in legacy protocols but also demonstrates exceptional functional group tolerance, allowing for the synthesis of diverse derivatives suitable for SAR (Structure-Activity Relationship) studies and commercial API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied heavily on classical condensation reactions involving trifluoroacetyl hydrazine and amidine compounds, or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These traditional pathways are fraught with significant operational challenges, including the requirement for harsh reaction conditions, the use of corrosive reagents, and the generation of difficult-to-remove byproducts that complicate downstream purification. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane, while effective, often suffer from safety concerns related to the explosive nature of diazo compounds and the limited stability of the reagents involved. The reliance on hydrazine derivatives also introduces toxicity risks that necessitate stringent containment measures and extensive waste treatment protocols, thereby inflating the overall cost of goods and extending the manufacturing lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN113307778A introduces a sophisticated yet operationally simple cycloaddition reaction between trifluoroethylimidoyl chloride and functionalized isonitriles (NIITP). This novel methodology leverages a synergistic dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate to drive the formation of the triazole ring under remarkably mild thermal conditions. The process eliminates the need for hazardous hydrazine reagents and unstable diazo precursors, replacing them with stable, commercially accessible building blocks that can be handled with standard safety precautions. By shifting the synthetic logic to a metal-promoted [3+2] cycloaddition, the new route achieves high atom economy and significantly reduces the environmental footprint associated with the production of these valuable heterocyclic scaffolds.
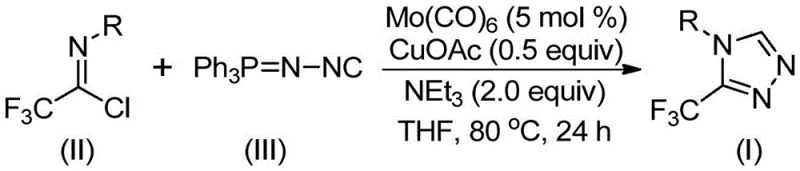
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the cooperative interaction between the molybdenum and copper species, which orchestrate the assembly of the five-membered triazole ring with high precision. The reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isonitrile complex that facilitates the subsequent nucleophilic attack. Concurrently, the cuprous acetate acts as a Lewis acid promoter, coordinating with the imidoyl chloride to enhance its electrophilicity and lower the activation energy for the cycloaddition step. This dual-activation strategy ensures that the reaction proceeds efficiently even with sterically hindered or electronically diverse substrates, resulting in the formation of a five-membered ring intermediate that subsequently undergoes elimination of triphenylphosphine oxide to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
From an impurity control perspective, the mildness of the reaction conditions (70°C to 90°C) is instrumental in suppressing common side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride, which are prevalent in more aggressive acidic or basic environments. The use of triethylamine as a base further buffers the system, neutralizing the HCl generated during the reaction without promoting decomposition of the sensitive triazole core. For Quality Assurance teams, this translates to a cleaner crude reaction profile, reducing the burden on purification steps and ensuring that the final API intermediate meets stringent purity specifications with minimal effort. The robustness of the catalytic cycle allows for consistent performance across different batches, a critical factor for maintaining supply chain reliability in commercial manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for scalability and ease of execution, making it highly attractive for process development teams aiming to transition from laboratory discovery to pilot plant production. The protocol involves a straightforward charging sequence where all reagents, including the catalysts, base, and molecular sieves, are combined in a suitable aprotic solvent such as tetrahydrofuran (THF). The reaction mixture is then heated to the specified temperature range and maintained for a period of 18 to 30 hours, after which the product can be isolated through standard filtration and chromatographic techniques. This simplicity in operation reduces the requirement for specialized equipment and minimizes operator exposure to hazardous chemicals, aligning perfectly with modern green chemistry principles and safety standards.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Mo-Cu co-catalyzed methodology presents a compelling value proposition centered on cost optimization and supply security. The primary drivers of raw material costs in pharmaceutical synthesis are often the complexity of the starting materials and the expense of specialized catalysts; however, this process utilizes trifluoroethylimidoyl chloride and functionalized isonitriles, which are derived from abundant aromatic amines and triphenylphosphine, ensuring a stable and cost-effective supply base. Furthermore, the catalyst loading is kept relatively low, with molybdenum hexacarbonyl used at approximately 5 mol % and cuprous acetate at 0.5 equivalents, both of which are commodity chemicals available from multiple global suppliers, thereby mitigating the risk of single-source dependency and price volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are realized through the elimination of expensive and hazardous reagents typically associated with triazole synthesis, such as hydrazine hydrates or diazonium salts, which require costly disposal and containment measures. By operating under mild thermal conditions in a common solvent like THF, the process significantly reduces energy consumption compared to high-temperature reflux methods or cryogenic reactions. Additionally, the high reaction efficiency and selectivity minimize the formation of byproducts, leading to higher yields and reduced solvent usage during purification, which collectively contribute to a substantial decrease in the overall cost of goods sold for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on commercially available and chemically stable starting materials ensures a robust supply chain that is less susceptible to disruptions caused by the regulatory restrictions often placed on energetic or toxic precursors. The broad substrate scope of the reaction allows for the flexible sourcing of various substituted aromatic amines, enabling manufacturers to adapt quickly to market demands or raw material shortages without compromising the integrity of the synthetic route. This flexibility is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients who demand consistent quality and availability.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable to gram-level reactions with consistent performance, indicating a clear path towards kilogram and ton-scale commercial production without the need for extensive re-optimization. The use of molecular sieves to manage water content and the generation of benign byproducts like triphenylphosphine oxide simplify the waste stream, facilitating easier compliance with increasingly stringent environmental regulations regarding heavy metal discharge and organic solvent emissions. This environmental compatibility not only reduces the cost of waste treatment but also enhances the sustainability profile of the manufacturing site, a key metric for modern corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology, providing clarity on its operational parameters and strategic benefits for potential partners. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this route for their own portfolios.
Q: What are the optimal reaction conditions for the Mo-Cu catalyzed triazole synthesis?
A: According to patent CN113307778A, the optimal conditions involve using THF as the solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a catalyst system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method demonstrates broad substrate tolerance, successfully accommodating R groups that are phenethyl or substituted/unsubstituted aryl rings, including substituents like methyl, methoxy, halogens (fluoro, chloro), and trifluoromethyl groups at ortho, meta, or para positions.
Q: How does this method compare to traditional hydrazine-based cyclization routes?
A: Unlike traditional methods that often require hazardous hydrazine derivatives or harsh cyclization conditions, this novel approach utilizes stable trifluoroethylimidoyl chloride and functionalized isonitriles under mild thermal conditions, significantly improving operational safety and simplifying post-treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in driving innovation within the pharmaceutical sector, and we are uniquely positioned to leverage technologies like the Mo-Cu co-catalyzed triazole synthesis for our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the exacting standards required for API synthesis.
We invite forward-thinking R&D and procurement leaders to collaborate with us to explore how this cutting-edge chemistry can optimize your supply chain and reduce manufacturing costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, along with specific COA data and route feasibility assessments that demonstrate the viability of this approach for your target molecules. Let us partner with you to accelerate your drug development timelines and secure a competitive advantage in the global marketplace through superior chemical manufacturing excellence.
