Advanced Electrochemical Manufacturing of 2-Phenylpropionic Acid for Loxoprofen Sodium Production
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for critical intermediates, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) like Loxoprofen Sodium. Patent CN113979854B introduces a groundbreaking electrochemical methodology for the preparation of 2-phenylpropionic acid, a pivotal precursor in this therapeutic class. This technology represents a significant paradigm shift from traditional thermal chemistry to controlled electro-organic synthesis, leveraging magnesium sacrificial anodes and dimethyl carbonate (DMC) to achieve superior outcomes. By operating under mild conditions and utilizing green solvents, this approach addresses the growing regulatory and economic pressures faced by modern chemical manufacturers. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-phenylpropionic acid has been plagued by severe operational constraints and safety hazards that complicate large-scale manufacturing. As illustrated in the comparative reaction schemes below, traditional routes often rely on extreme conditions that demand specialized, capital-intensive infrastructure. One prevalent method involves the reaction of benzyl cyanide with dimethyl carbonate under high temperature (180°C) and high pressure (over 20 kg), necessitating robust autoclaves and rigorous safety protocols to prevent catastrophic failure. Alternatively, carbonylation of styrene using carbon monoxide introduces significant toxicity risks, requiring complex gas handling systems to protect personnel and the environment. These legacy processes not only incur high energy costs but also generate substantial waste streams, making them increasingly untenable in a regulatory landscape focused on green chemistry principles.
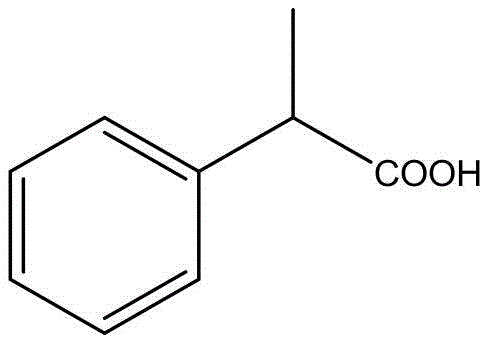
The Novel Approach
In stark contrast to these hazardous legacy protocols, the electrochemical method disclosed in the patent offers a streamlined, ambient-pressure alternative that fundamentally redesigns the reaction landscape. By replacing thermal activation with electrical energy, the process eliminates the need for high-temperature reactors and toxic carbon monoxide feeds. The utilization of 1-phenylchloroethane and dimethyl carbonate allows the reaction to proceed at mild temperatures ranging from 10°C to 40°C, drastically reducing the thermal load on the facility. Furthermore, the substitution of toxic methylating agents like dimethyl sulfate with the benign dimethyl carbonate enhances the safety profile of the operation. This transition not only mitigates risk but also simplifies the engineering requirements for plant scale-up, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Electrochemical Carboxylation and Methylation
The core innovation of this technology lies in the precise orchestration of electrochemical parameters to drive the formation of the carbon-carbon bond. In this system, a magnesium electrode serves as a consumable anode, generating reactive organomagnesium species in situ upon the application of a direct current between 10 mA and 2 A. The presence of a phase transfer catalyst, typically a quaternary ammonium salt such as tetrabutylammonium bromide, facilitates the interaction between the organic substrate and the electrochemically generated species within the dimethyl carbonate medium. This setup ensures that the methylation and subsequent carboxylation steps occur with high regioselectivity, minimizing the formation of side products that typically plague free-radical or thermal ionic reactions. The control over current density allows chemists to fine-tune the reaction rate, ensuring complete conversion of the 1-phenylchloroethane starting material without over-reaction or decomposition.
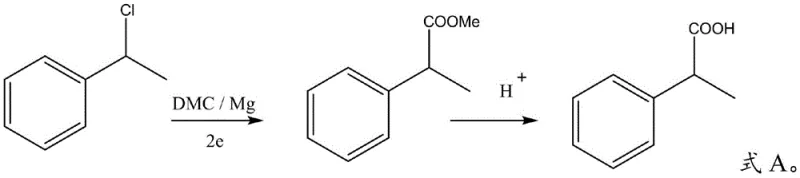
Impurity control is another critical aspect where this electrochemical mechanism excels, directly impacting the downstream purification burden. The reaction pathway avoids the harsh acidic or basic conditions often required in hydrolysis steps of other routes until the final stage, preserving the integrity of the intermediate ester. The specific stoichiometry, maintaining a molar ratio of magnesium to substrate of at least 1:1, ensures that the consumption of the electrode matches the reaction demand, preventing the accumulation of unreacted halides. Subsequent hydrolysis with inorganic acids like hydrochloric or sulfuric acid at moderate temperatures (60-80°C) cleanly converts the ester to the target acid. The result is a crude product with exceptionally high purity, often exceeding 99% by HPLC analysis, which significantly reduces the complexity and cost of final crystallization or distillation steps required to meet stringent pharmacopeial standards.
How to Synthesize 2-Phenylpropionic Acid Efficiently
Implementing this electrochemical protocol requires careful attention to electrode configuration and electrolyte composition to maximize yield and reproducibility. The process begins with the preparation of an electrolytic cell containing the magnesium anode and a carbon cathode, charged with the substrate and dimethyl carbonate solvent. Operators must maintain strict control over the direct current supply and temperature to ensure the steady generation of the reactive intermediate. Following the electrolysis, a straightforward workup involving pH adjustment and aqueous washing removes inorganic salts, preparing the organic phase for the final hydrolysis step. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Perform electrochemical reaction using 1-phenylchloroethane and dimethyl carbonate with a magnesium consumable electrode and quaternary ammonium catalyst.
- Adjust pH to neutral, separate organic phase, and wash to remove magnesium salts and methanol byproducts.
- Hydrolyze the resulting ester with inorganic acid at 60-80°C, followed by distillation to isolate pure 2-phenylpropionic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits regarding cost stability and operational resilience. The shift away from high-pressure and high-temperature requirements means that manufacturing can be conducted in standard glass-lined or stainless steel reactors rather than specialized autoclaves, significantly lowering capital expenditure barriers for contract manufacturing organizations. Additionally, the reliance on dimethyl carbonate as both solvent and reagent simplifies the raw material inventory, reducing the logistical complexity associated with managing multiple volatile organic compounds. This consolidation of materials not only streamlines purchasing but also minimizes the storage footprint and associated safety compliance costs, driving substantial cost savings in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and toxic gases like carbon monoxide removes the need for complex recovery systems and hazardous waste disposal protocols. By utilizing electricity as the primary reagent driver and magnesium as a low-cost consumable, the variable cost of goods sold is optimized. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to byproducts, improving the overall mass balance and economic efficiency of the production line without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically 1-phenylchloroethane and dimethyl carbonate, are commodity chemicals with robust global supply chains, reducing the risk of feedstock shortages. Unlike processes dependent on specialized catalysts that may have long lead times or single-source suppliers, this method utilizes widely available reagents. This accessibility ensures consistent production scheduling and reduces the vulnerability of the supply chain to geopolitical or logistical disruptions, guaranteeing a steady flow of high-purity pharmaceutical intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions (10-40°C) and atmospheric pressure operation make this process inherently safer and easier to scale from pilot plants to commercial tonnage. The absence of toxic emissions and the use of a green solvent system align perfectly with increasingly strict environmental regulations, minimizing the permitting hurdles for facility expansion. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand reputation of the supply chain partners by demonstrating a commitment to sustainable and responsible chemical production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 2-phenylpropionic acid. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of this modern synthetic route. Understanding these details is crucial for technical teams evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this electrochemical method over traditional high-pressure routes?
A: Unlike conventional methods requiring temperatures up to 180°C and pressures exceeding 20 kg, this electrochemical process operates at mild temperatures between 10-40°C and atmospheric pressure. Furthermore, it eliminates the need for toxic carbon monoxide gas or highly toxic dimethyl sulfate, significantly reducing operational hazards and containment costs.
Q: How does the use of Dimethyl Carbonate (DMC) contribute to process efficiency?
A: In this novel protocol, Dimethyl Carbonate serves a dual function as both the methylation reagent and the reaction solvent. This dual role simplifies the reaction matrix by avoiding the need for additional volatile organic solvents, thereby streamlining downstream separation processes and enhancing the overall environmental profile of the manufacturing cycle.
Q: What purity levels can be achieved with this electrochemical synthesis route?
A: The method demonstrates exceptional selectivity, consistently yielding 2-phenylpropionic acid with an HPLC purity greater than 99%. The specific control over the electrochemical parameters and the efficient removal of magnesium salts during the workup phase ensure a clean impurity profile suitable for sensitive pharmaceutical applications like Loxoprofen Sodium synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylpropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value pharmaceutical intermediates with unmatched efficiency. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced electrochemical reactors capable of maintaining stringent purity specifications, guaranteeing that every batch of 2-phenylpropionic acid meets the exacting standards required for Loxoprofen Sodium synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project benefits from the highest levels of quality, safety, and economic performance available in the market.
