Revolutionizing Imine Production: Green Photocatalytic Technology for Commercial Scale-Up
Revolutionizing Imine Production: Green Photocatalytic Technology for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable and energy-efficient processes, driven by the urgent need to reduce environmental footprints while maintaining high product quality. A pivotal advancement in this domain is detailed in patent CN108097323B, which discloses a novel method for preparing imine compounds through the oxidative coupling of primary amines at room temperature. This technology leverages a sophisticated ligand-modified supported gold (Au) catalyst under visible light irradiation, utilizing molecular oxygen as a green oxidant. Unlike traditional thermal methods that require harsh conditions and stoichiometric oxidants, this photocatalytic approach achieves exceptional conversion rates ranging from 92% to 100% and yields between 95% and 99%. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this represents a transformative opportunity to streamline the synthesis of critical nitrogen-containing intermediates. The ability to operate under mild conditions not only enhances safety but also significantly lowers the energy consumption associated with heating and cooling cycles, positioning this method as a superior alternative for the reliable pharmaceutical intermediates supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of imines has relied heavily on the condensation of amines with carbonyl compounds or the oxidation of amines using stoichiometric oxidants, both of which present significant logistical and environmental challenges. Traditional protocols often employ toxic catalysts such as iodine or expensive oxidants like tert-butyl hydroperoxide (TBHP), which necessitate rigorous safety measures and complex waste disposal procedures to handle hazardous byproducts. Furthermore, many existing catalytic systems suffer from poor atom economy and require elevated temperatures to drive the reaction to completion, leading to increased operational expenditures and potential thermal degradation of sensitive functional groups. The reliance on non-recyclable homogeneous catalysts further exacerbates the cost burden, as the separation of trace metal impurities from the final API intermediate often requires additional purification steps that erode overall yield. These inefficiencies create bottlenecks in the supply chain, making it difficult to achieve cost reduction in fine chemical manufacturing without compromising on purity or regulatory compliance standards.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a heterogeneous photocatalytic system that operates efficiently at room temperature, effectively bypassing the thermal energy barriers associated with conventional synthesis. By employing a ligand-modified gold nanoparticle catalyst supported on semiconductors like TiO2 or CeO2, the process harnesses visible light energy to drive the oxidative coupling of primary amines directly. This approach eliminates the need for hazardous stoichiometric oxidants, replacing them with ambient oxygen, which serves as a clean and inexhaustible electron acceptor. The heterogeneity of the catalyst allows for straightforward separation via simple centrifugation, facilitating catalyst recovery and reuse without the need for complex filtration or extraction workflows. As illustrated in the reaction scheme below, the transformation proceeds with high selectivity, minimizing the formation of over-oxidation byproducts such as nitriles or amides, which are common pitfalls in amine oxidation chemistry.
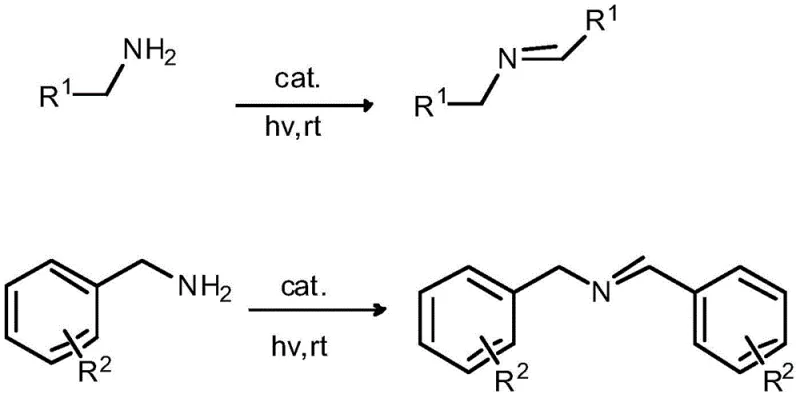
This streamlined workflow not only accelerates the reaction timeline but also ensures a cleaner crude profile, thereby reducing the load on downstream purification units. For supply chain heads, this translates to a more robust and predictable manufacturing process that is less susceptible to raw material volatility associated with specialized oxidants.
Mechanistic Insights into Ligand-Modified Au Photocatalysis
The exceptional performance of this catalytic system is rooted in the intricate electronic interactions facilitated by the dual-ligand modification on the gold nanoparticle surface. The patent elucidates that the simultaneous presence of triphenylphosphine (PPh3) and thiol ligands induces a Ligand-to-Metal Charge Transfer (LMCT) effect upon irradiation with visible light. When the catalyst absorbs photons, photogenerated electrons are excited from the ligand orbitals to the metal conduction band, creating electron-hole pairs that are critical for the redox cycle. The photogenerated electrons are efficiently transferred to the semiconductor support, where they activate molecular oxygen to form superoxide radicals, while the corresponding holes on the gold surface oxidize the adsorbed primary amine substrate. This spatial separation of charge carriers prevents rapid recombination, thereby maximizing the quantum efficiency of the photocatalytic process and ensuring high turnover frequencies even under mild illumination conditions.
From an impurity control perspective, the specific tuning of the Au:P and Au:S molar ratios plays a pivotal role in dictating the selectivity of the reaction. The patent data suggests that optimizing these ratios, typically within the range of 1:0.1 to 1:5 for phosphorus and 1:0.01 to 1:5 for sulfur, creates a steric and electronic environment that favors the formation of the C=N bond over competing pathways. The support material, whether it be anatase TiO2, rutile, or g-C3N4, further modulates the band structure to align with the redox potentials of the substrates. This precise engineering of the catalyst surface minimizes side reactions, resulting in high-purity imine intermediates that meet the stringent specifications required for downstream pharmaceutical applications. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as light wavelength (400-800 nm) and solvent polarity to maximize yield and minimize waste generation.
How to Synthesize Imine Intermediates Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the preparation of the specialized catalyst and the optimization of the irradiation setup. The process begins with the synthesis of the ligand-protected gold nanoparticles, followed by their deposition onto the chosen semiconductor support to create the active heterogeneous catalyst. Once prepared, the reaction is conducted by suspending the primary amine substrate and the catalyst in a suitable organic solvent, followed by purging the system with oxygen to ensure an adequate supply of the oxidant. The mixture is then subjected to visible light irradiation at room temperature for a duration typically ranging from 1 to 20 hours, depending on the specific substrate reactivity and light intensity. Detailed standard operating procedures regarding catalyst loading, solvent selection, and workup protocols are essential for reproducibility and are summarized in the technical guide below.
- Prepare the ligand-modified supported Au catalyst by modifying gold nanoparticles with triphenylphosphine and thiol ligands, then loading them onto a semiconductor support such as TiO2 or CeO2.
- Combine the substrate primary amine, solvent (e.g., dichloromethane or acetonitrile), and the prepared catalyst in a reaction vessel, then purge the system with oxygen to replace the internal atmosphere.
- Irradiate the mixture with visible light (400-800 nm) at room temperature for 1 to 20 hours, followed by catalyst removal via centrifugation and product purification through column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift from expensive, hazardous oxidants to atmospheric oxygen fundamentally alters the cost structure of imine production, removing the volatility associated with purchasing specialized chemical reagents. Moreover, the recyclability of the heterogeneous gold catalyst means that the effective cost per kilogram of product decreases significantly over multiple batches, as the catalyst does not need to be replenished after every run. This durability enhances supply chain reliability by reducing the frequency of catalyst ordering and mitigating the risk of production stoppages due to reagent shortages. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating infrastructure, leading to substantial operational cost savings and a reduced carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants like TBHP and toxic catalysts like iodine removes significant line items from the bill of materials, directly impacting the gross margin of the final intermediate. Since oxygen is sourced from the air and the catalyst is reusable, the variable costs associated with raw materials are drastically simplified and lowered. Furthermore, the simplified workup procedure, which avoids complex quenching steps required for reactive oxidants, reduces labor hours and solvent consumption during the isolation phase. These cumulative efficiencies result in a more competitive pricing structure for high-purity imine intermediates without sacrificing quality or yield.
- Enhanced Supply Chain Reliability: Relying on commodity gases like oxygen and stable heterogeneous catalysts insulates the production process from the supply disruptions often seen with specialized fine chemical reagents. The robustness of the catalyst, which maintains activity over multiple cycles as demonstrated in the patent examples, ensures consistent batch-to-batch quality and throughput. This stability allows for better production planning and inventory management, reducing the need for large safety stocks of hazardous materials. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, enabling faster response to market demands and customer orders.
- Scalability and Environmental Compliance: The transition to a green chemistry process using visible light and oxygen aligns perfectly with increasingly stringent environmental regulations regarding waste discharge and solvent emissions. The absence of heavy metal waste streams from stoichiometric oxidants simplifies wastewater treatment and reduces the regulatory burden on the manufacturing site. Scalability is facilitated by the use of flow chemistry compatible photocatalytic reactors, which can easily transition from gram-scale R&D to multi-ton commercial production. This ease of scale-up ensures that the commercial scale-up of complex organic syntheses can be achieved rapidly, meeting the growing global demand for sustainable chemical solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic imine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: Is the gold-based photocatalyst reusable in this imine synthesis process?
A: Yes, the patent data indicates that the ligand-modified supported Au catalyst exhibits excellent recyclability. After the reaction, the catalyst can be separated by centrifugation and reused in subsequent cycles without significant loss of catalytic activity or selectivity, which is crucial for reducing long-term production costs.
Q: What are the advantages of using oxygen over traditional oxidants like TBHP?
A: Using molecular oxygen as the terminal oxidant offers substantial safety and economic benefits compared to expensive and hazardous oxidants like tert-butyl hydroperoxide (TBHP) or toxic iodine catalysts. Oxygen is abundant, inexpensive, and generates water as the only byproduct, aligning with green chemistry principles and simplifying waste treatment protocols.
Q: Does this photocatalytic method work for aliphatic amines or only benzylamines?
A: While benzylamines are the primary focus due to their high reactivity, the patent examples demonstrate that the system is also effective for aliphatic primary amines such as butylamine and methylamine. The ligand modification on the gold surface enhances the substrate scope, allowing for the synthesis of a diverse range of imine intermediates required in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imine Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modernizing the production of critical chemical building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for R&D directors, consistency is key, and our manufacturing protocols are designed to deliver high-purity imine intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this green synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
