Advanced Photocatalytic Oxidation for High-Purity Imine Intermediates Manufacturing
Advanced Photocatalytic Oxidation for High-Purity Imine Intermediates Manufacturing
The chemical industry is currently witnessing a paradigm shift towards sustainable synthesis methodologies, particularly in the production of high-value nitrogen-containing heterocycles and intermediates. A groundbreaking development in this sector is detailed in patent CN108623494B, which discloses a novel method for synthesizing imines through the visible light-catalyzed oxidation of asymmetric secondary amines. This technology leverages sulfur-containing aza metalloporphyrins as robust photocatalysts, specifically utilizing molecular oxygen as a green oxidant and visible light (λ ≥ 420nm) as the energy driver. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a significant leap forward in process efficiency, offering a pathway to produce critical imine intermediates with exceptional selectivity while drastically reducing the environmental footprint associated with traditional oxidative dehydrogenation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of imines from secondary amines has relied heavily on stoichiometric oxidants such as manganese dioxide, chromium-based reagents, or hypervalent iodine compounds. These conventional routes are fraught with significant drawbacks that impact both the economic viability and the environmental compliance of large-scale manufacturing. Firstly, the use of heavy metal oxidants generates substantial quantities of toxic waste, necessitating complex and costly downstream purification steps to meet stringent regulatory limits on residual metals in active pharmaceutical ingredients (APIs). Secondly, these reactions often require harsh conditions, including elevated temperatures and strong acidic or basic media, which can lead to the degradation of sensitive functional groups on the substrate. Furthermore, achieving high regioselectivity in the oxidation of asymmetric secondary amines is notoriously difficult with traditional methods, often resulting in complex mixtures of oxidative coupling products and dehydrogenation products that are challenging to separate, thereby lowering the overall yield and increasing production costs.
The Novel Approach
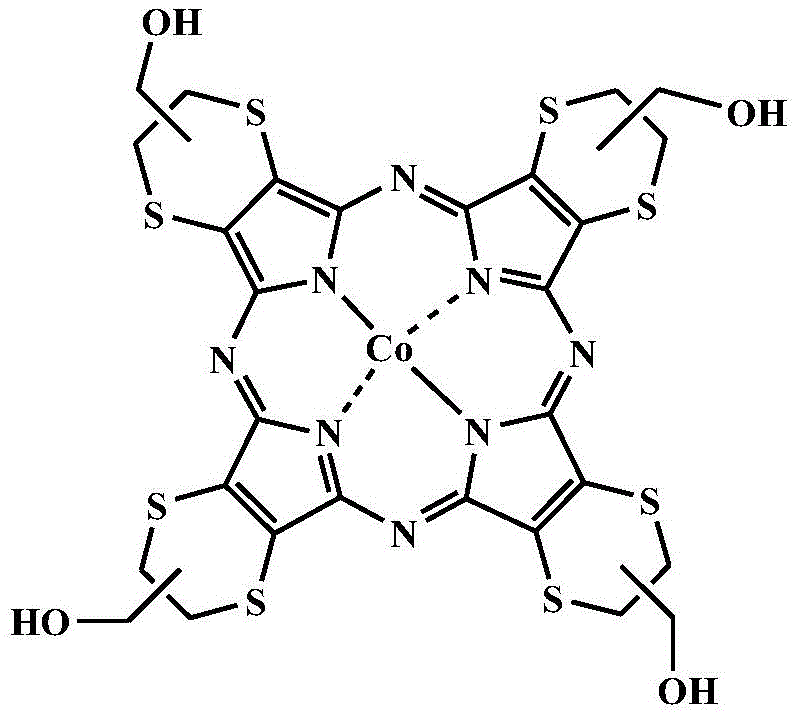
In stark contrast to these legacy methods, the technology described in patent CN108623494B introduces a biomimetic photocatalytic system that operates under remarkably mild conditions. By employing a sulfur-containing aza metalloporphyrin, specifically tetrakis(1,4-dithiine)tetraazacobalt porphyrin (CoPz(hmdtn)4), the process activates molecular oxygen efficiently under visible light irradiation. This approach not only eliminates the need for hazardous stoichiometric oxidants but also utilizes air or pure oxygen, which is abundant and inexpensive. The integration of 1,8-diazabicycloundec-7-ene (DBU) as a crucial auxiliary agent further refines the reaction pathway, ensuring that the oxidative dehydrogenation proceeds with high specificity. This novel strategy transforms the synthesis of imines into a green, atom-economical process that aligns perfectly with modern principles of sustainable chemistry, offering a reliable solution for the cost reduction in fine chemical manufacturing.
Mechanistic Insights into CoPz(hmdtn)4-Catalyzed Photocatalytic Oxidation
The core of this technological breakthrough lies in the unique electronic properties of the sulfur-containing aza metalloporphyrin catalyst. Unlike standard porphyrins, the incorporation of sulfur atoms into the macrocyclic framework enhances visible light absorption and stabilizes the excited states necessary for photocatalysis. Upon irradiation with visible light (λ ≥ 420nm), the CoPz(hmdtn)4 catalyst enters an excited state capable of transferring energy to molecular oxygen. Quenching experiments detailed in the patent reveal that the reaction mechanism involves the generation of reactive oxygen species, specifically superoxide anion radicals (O2·-) and singlet oxygen (1O2). These species are pivotal in initiating the oxidation of the amine substrate. The presence of the cobalt center facilitates the redox cycling required to sustain the catalytic turnover, ensuring that the catalyst remains active over extended periods without significant degradation, which is a critical factor for commercial scalability.
Furthermore, the role of the DBU auxiliary cannot be overstated in controlling the reaction selectivity. Mechanistic studies indicate that the oxidation proceeds via a nitrogen cation radical intermediate. In a neutral or acidic environment, this intermediate might follow multiple pathways, leading to a mixture of products. However, the strong basicity of DBU promotes the rapid deprotonation of the benzylic position of the nitrogen cation radical. This kinetic acceleration directs the reaction exclusively towards the formation of the desired imine product (N-benzylidenemethylamine) rather than oxidative coupling byproducts. The Hammett plot analysis confirms the existence of this cationic intermediate and demonstrates that electron-donating groups on the benzene ring enhance the reaction rate, while electron-withdrawing groups slow it down, providing valuable predictive power for substrate scope optimization. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates with consistent quality.
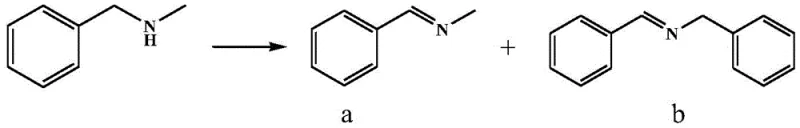
How to Synthesize N-Methylbenzylamine Derivatives Efficiently
The practical implementation of this photocatalytic method is straightforward and adaptable to existing reactor infrastructure. The process typically involves dissolving the CoPz(hmdtn)4 catalyst in a polar aprotic solvent such as acetonitrile, followed by the addition of the asymmetric secondary amine substrate and the DBU auxiliary. The mixture is initially stirred in the dark to ensure homogeneity and equilibrium before being subjected to visible light irradiation under an oxygen atmosphere. The reaction proceeds rapidly at ambient temperature, typically reaching completion within a few hours. This operational simplicity reduces the technical barrier for adoption, allowing manufacturers to transition from batch processes involving hazardous oxidants to continuous flow or batch photocatalytic systems with minimal retooling. For detailed standard operating procedures and safety guidelines regarding the handling of photocatalytic reactors, please refer to the technical documentation below.
- Dissolve the sulfur-containing aza metalloporphyrin catalyst (e.g., CoPz(hmdtn)4) in a suitable solvent like acetonitrile.
- Add the auxiliary agent 1,8-diazabicycloundec-7-ene (DBU) and the asymmetric secondary amine substrate to the solution.
- Stir in the dark, then irradiate with visible light (λ ≥ 420nm) under an oxygen atmosphere to obtain the imine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light photocatalytic technology offers compelling strategic advantages that extend beyond mere technical performance. The shift from stoichiometric heavy metal oxidants to a catalytic system driven by oxygen and light fundamentally alters the cost structure of imine production. By eliminating the purchase of expensive and regulated oxidizing agents, companies can achieve significant raw material cost savings. Moreover, the removal of heavy metals from the process stream drastically simplifies the purification workflow, reducing the consumption of silica gel, solvents, and chelating agents typically required for metal scavenging. This streamlined downstream processing translates directly into reduced cycle times and lower utility costs, enhancing the overall margin profile of the final product.
- Cost Reduction in Manufacturing: The transition to a catalytic aerobic oxidation process removes the dependency on volatile and costly stoichiometric oxidants. Since molecular oxygen serves as the terminal oxidant, the direct material costs are minimized. Additionally, the high selectivity achieved (>99% in optimized conditions) means that less starting material is wasted on byproduct formation, improving the effective yield. The elimination of heavy metal waste disposal fees further contributes to a leaner cost base, making the production of complex imine intermediates more economically competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on specialized oxidants often exposes supply chains to geopolitical risks and availability fluctuations. In contrast, the reagents for this process—cobalt porphyrin catalysts, DBU, and oxygen—are commodity chemicals with robust and diversified supply bases. The stability of the CoPz(hmdtn)4 catalyst ensures consistent batch-to-batch performance, reducing the risk of production delays caused by reagent variability. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental discharge intensify, processes that minimize toxic waste generation are increasingly valued. This photocatalytic method produces water as the primary byproduct of oxygen reduction, avoiding the generation of heavy metal sludge. This inherent greenness simplifies environmental permitting and reduces the liability associated with waste treatment. The mild reaction conditions (ambient temperature and pressure) also lower the energy demand for heating and cooling, facilitating easier scale-up from laboratory to multi-ton commercial production without the need for high-pressure autoclaves or specialized high-temperature reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light photocatalytic oxidation technology. These insights are derived directly from the experimental data and mechanistic studies presented in patent CN108623494B, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the primary advantage of using CoPz(hmdtn)4 over traditional oxidants?
A: CoPz(hmdtn)4 enables the use of molecular oxygen and visible light as the oxidant and energy source, respectively, eliminating the need for stoichiometric heavy metal oxidants and reducing hazardous waste generation significantly.
Q: How does the DBU auxiliary agent improve reaction selectivity?
A: DBU creates a strongly alkaline environment that facilitates the formation of nitrogen cation radical intermediates, promoting the removal of benzyl hydrogen and driving the reaction towards a single imine product with over 99% selectivity.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (ambient temperature and pressure) using stable catalysts and common solvents like acetonitrile, making it highly suitable for commercial scale-up in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imine Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green photocatalytic technologies in the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the CoPz(hmdtn)4-catalyzed oxidation can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photocatalytic reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity imine intermediates that adhere to the highest standards of quality and consistency.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating how this green chemistry approach can optimize your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with sustainable and efficient manufacturing solutions.
