Advanced Manufacturing of Dronedarone Intermediates: A Cost-Effective Three-Step Route for Global API Production
Advanced Manufacturing of Dronedarone Intermediates: A Cost-Effective Three-Step Route for Global API Production
The global pharmaceutical landscape is witnessing a surge in demand for effective antiarrhythmic agents, particularly for the management of atrial fibrillation, a condition affecting millions worldwide. At the forefront of this therapeutic class is Dronedarone, a novel medication that offers a superior safety profile compared to its predecessor, amiodarone, by minimizing extra-cardiac toxicity while retaining potent electrophysiological properties. As the market for cardiovascular therapeutics expands, the imperative for efficient, scalable, and cost-effective manufacturing processes for Dronedarone and its key intermediates has never been more critical for supply chain stability. Patent CN102180848A discloses a groundbreaking preparation method that addresses these industrial needs through a streamlined three-step synthetic sequence. This technical insight report analyzes the proprietary methodology detailed in the patent, highlighting its potential to redefine the supply chain dynamics for reliable pharmaceutical intermediates suppliers and API manufacturers alike.
The disclosed innovation focuses on the synthesis of N-(2-butyl-3-(4-(3-dibutylamino propoxy-) benzoyl) cumarone-5-yl) methanesulfonamide, commonly known as Dronedarone. The core of this technological advancement lies in its ability to bypass the economic and operational bottlenecks associated with legacy synthesis routes. By leveraging a strategic combination of nucleophilic substitution, catalytic hydrogenation, and sulfonylation, the process achieves high purity and operational simplicity. For R&D directors and procurement managers, understanding the nuances of this pathway is essential for evaluating potential partnerships and optimizing the cost reduction in API manufacturing. The following analysis dissects the chemical engineering principles, reaction mechanisms, and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of the methodology described in CN102180848A, the industrial synthesis of Dronedarone was often hindered by reliance on complex and economically burdensome pathways. One prominent example found in the background art, specifically referenced in patent US20050049302, involves a synthetic route that necessitates the use of significantly more expensive raw materials. These conventional approaches frequently rely on intricate Friedel-Crafts acylation strategies or multi-step functional group manipulations that require harsh reaction conditions and specialized reagents. Such methods not only inflate the direct material costs but also introduce significant challenges in impurity control and waste management. The reliance on costly precursors creates a fragile supply chain, where fluctuations in the availability of niche starting materials can lead to substantial production delays and increased lead times for high-purity drug substances.
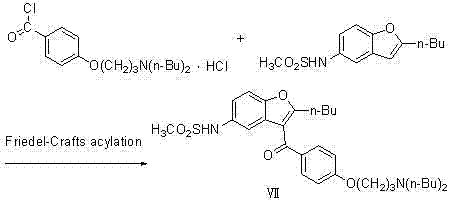
Furthermore, traditional routes often suffer from poor atom economy and generate substantial quantities of hazardous by-products, complicating the downstream purification processes. The need for rigorous chromatographic separations or multiple recrystallization steps to meet stringent pharmaceutical purity standards adds layers of operational complexity and reduces overall yield. For a commercial scale-up of complex pharmaceutical intermediates, these inefficiencies translate directly into higher capital expenditure and operating costs. The environmental footprint of these older methods is also a growing concern, as regulatory bodies increasingly demand greener chemistry solutions. Consequently, there is a pressing industry-wide demand for alternative synthetic strategies that can deliver the same high-quality active pharmaceutical ingredients without the associated economic and environmental penalties of legacy technologies.
The Novel Approach
In stark contrast to the limitations of the prior art, the novel approach presented in CN102180848A introduces a robust and economically viable three-step synthesis that fundamentally alters the production economics of Dronedarone. This innovative route begins with a nucleophilic substitution reaction where a mesylate-containing precursor (Compound IV) reacts with dibutylamine to form the key intermediate Compound V. This step is advantageous because it utilizes readily available and cost-effective amines, avoiding the need for exotic or prohibitively expensive building blocks. The reaction conditions are flexible, allowing for operation in common organic solvents such as toluene, tetrahydrofuran, or dichloromethane across a broad temperature range of 30°C to 180°C. This flexibility provides process engineers with significant latitude to optimize reaction kinetics and throughput based on specific plant capabilities.
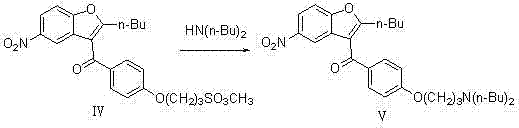
Following the substitution, the process employs a catalytic hydrogenation step to reduce the nitro group of Compound V to an amine (Compound VI). This transformation is critical as it sets the stage for the final sulfonylation. The use of heterogeneous palladium on carbon (Pd/C) catalysts ensures high selectivity and ease of catalyst recovery, which is a major advantage for continuous processing. Finally, the intermediate amine undergoes sulfonylation with methanesulfonyl chloride to yield the final Dronedarone product. This sequence is characterized by its operational simplicity, high controllability, and the use of standard industrial solvents. By eliminating the need for expensive raw materials and simplifying the purification workflow, this novel approach offers a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing, ensuring a more resilient and competitive supply chain.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Substitution
The chemical elegance of this synthesis lies in the precise control of reaction mechanisms to ensure high fidelity in molecular construction. The initial nucleophilic substitution (Compound IV to V) proceeds via an SN2 mechanism where the lone pair of electrons on the nitrogen atom of dibutylamine attacks the electrophilic carbon attached to the mesylate leaving group. The choice of solvent plays a pivotal role here; polar aprotic solvents or moderately polar solvents like toluene facilitate the dissolution of reactants while stabilizing the transition state. The reaction temperature, optimized between 39°C and 145°C in various embodiments, provides the necessary activation energy to overcome the steric hindrance of the dibutyl groups, ensuring complete conversion without significant degradation of the sensitive benzoyl-cumarone scaffold. This step effectively installs the basic side chain required for the drug's pharmacological activity.
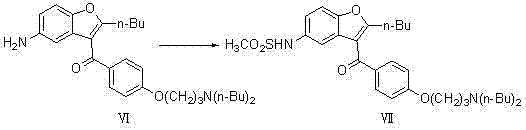
The subsequent reduction of the nitro group in Compound V to the amine in Compound VI is a masterpiece of chemoselective catalysis. Utilizing Pd/C under hydrogen pressures ranging from 0.1 to 3 MPa, the reaction selectively targets the nitro functionality while leaving the ketone, ether, and furan ring intact. This selectivity is paramount; non-selective reduction could lead to the formation of hydroxylamines or the reduction of the carbonyl group, generating difficult-to-remove impurities that would compromise the safety profile of the final API. The heterogeneous nature of the Pd/C catalyst allows for simple filtration post-reaction, minimizing metal contamination in the product stream—a critical quality attribute for regulatory compliance. The final sulfonylation step involves the reaction of the newly formed amine with methanesulfonyl chloride. This exothermic reaction is carefully managed by controlling the addition rate and temperature (30°C to 150°C), ensuring that the sulfonamide bond is formed efficiently. The use of bases or scavengers, implied by the workup procedures involving aqueous extraction, helps neutralize the HCl by-product, driving the equilibrium towards the desired product and facilitating high-purity isolation.
How to Synthesize Dronedarone Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and minimize impurity formation. The patent outlines specific embodiments that serve as a blueprint for process optimization, detailing solvent choices, molar ratios, and thermal profiles. For instance, the use of toluene in the substitution step not only acts as a solvent but can also facilitate the removal of water formed during the reaction if azeotropic distillation is employed, although the primary mechanism here is substitution of a mesylate. The hydrogenation step benefits from the use of alcohols like ethanol or methanol, which provide excellent solubility for the nitro intermediate while being compatible with the Pd/C catalyst. Detailed standardized synthesis steps are crucial for technology transfer and scale-up, ensuring that the laboratory success translates seamlessly to commercial production environments.
- Perform nucleophilic substitution on Compound IV with dibutylamine in an organic solvent like toluene at 30-180°C to obtain Compound V.
- Conduct catalytic hydrogenation of Compound V using Pd/C catalyst under 0.1-3 MPa hydrogen pressure to reduce the nitro group to an amine (Compound VI).
- Execute sulfonylation of Compound VI with methanesulfonyl chloride in a solvent such as dichloroethane to yield the final Dronedarone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN102180848A presents a multitude of strategic advantages that extend beyond mere chemical efficiency. The most significant impact is observed in the realm of cost structure. By replacing expensive starting materials found in legacy routes with commodity chemicals like dibutylamine and methanesulfonyl chloride, the direct material cost is drastically reduced. Furthermore, the elimination of complex purification steps and the use of recoverable heterogeneous catalysts contribute to substantial cost savings in terms of solvent consumption and waste disposal. This economic efficiency allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates, providing a buffer against raw material price volatility and enhancing margin stability for downstream API producers.
- Cost Reduction in Manufacturing: The streamlined three-step process eliminates the need for costly reagents and complex multi-step sequences associated with older methodologies. By utilizing common solvents such as toluene, ethanol, and dichloromethane, the process leverages existing supply chains for bulk chemicals, avoiding the premiums associated with specialty reagents. Additionally, the high selectivity of the Pd/C catalyzed reduction minimizes the formation of by-products, thereby reducing the load on downstream purification units and lowering the overall cost of goods sold. This efficiency translates directly into a more favorable cost structure for the final API, enabling better market positioning.
- Enhanced Supply Chain Reliability: The reliance on widely available raw materials significantly mitigates supply chain risks. Unlike routes that depend on niche intermediates with limited global suppliers, this method uses commodities that are produced at scale by numerous chemical manufacturers worldwide. This diversification of the supply base ensures continuity of supply even in the face of regional disruptions or logistical bottlenecks. Moreover, the robustness of the reaction conditions—tolerating a wide range of temperatures and pressures—means that production is less susceptible to minor equipment variances, further securing the reliability of delivery schedules for critical drug substances.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations such as batch hydrogenation and standard liquid-liquid extraction that are commonplace in modern chemical plants. The use of heterogeneous catalysts simplifies the separation process, reducing the generation of heavy metal waste and aligning with increasingly stringent environmental regulations. The ability to recycle solvents and the high atom economy of the substitution and sulfonylation steps contribute to a greener manufacturing footprint. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing partner, a key consideration for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Dronedarone intermediates. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and quality assurance measures. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This route avoids expensive raw materials used in prior art (like US20050049302) and utilizes common solvents like toluene and ethanol, significantly lowering production costs and simplifying purification.
Q: How is the purity of the intermediate controlled during hydrogenation?
A: By using heterogeneous Pd/C catalysts under controlled pressure (0.1-3 MPa) and temperature (20-120°C), the reaction selectively reduces the nitro group without affecting the sensitive benzoyl or ether linkages.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses standard batch reactor conditions, common organic solvents, and robust catalysts, making it highly scalable from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dronedarone Supplier
As the demand for advanced cardiovascular therapeutics continues to grow, partnering with a technically proficient and commercially agile manufacturer is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in process chemistry to deliver high-quality intermediates and APIs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global clinical and commercial programs. We are committed to maintaining stringent purity specifications through our rigorous QC labs, employing state-of-the-art analytical techniques to guarantee that every batch meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to unlock the full potential of this efficient synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By engaging with us, you gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of switching to this optimized manufacturing process. Contact us today to discuss how we can support your supply chain with reliable, cost-effective, and high-purity Dronedarone intermediates.
