Advanced Synthetic Route for Dronedarone Intermediates: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced Synthetic Route for Dronedarone Intermediates: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for cardiovascular therapeutics, particularly for complex molecules like Dronedarone. Patent CN103228639A introduces a transformative methodology for the preparation of N-[2-n-butyl-3-{4-[(3-di-n-butylamino)-propoxy]benzoyl}benzofuran-5-yl]-methanesulfonamide, widely known as Dronedarone. This innovation addresses long-standing challenges in impurity control and process scalability by employing a strategic N-acyl protection and selective cleavage mechanism. Unlike traditional linear syntheses that struggle with selective functionalization in late stages, this novel approach utilizes a convergent strategy where the methanesulfonyl group is installed early on the benzofuran core. The core breakthrough lies in the manipulation of the benzofuran derivative of Formula II, where a temporary acyl group is selectively cleaved to reveal the final active pharmaceutical ingredient. 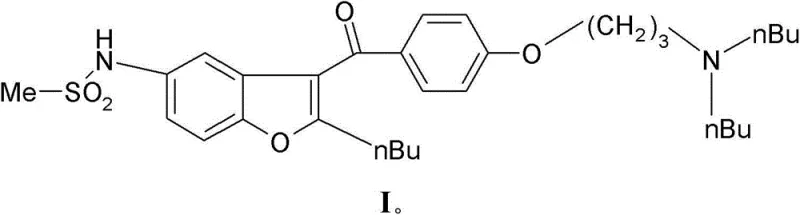 This structural refinement not only mitigates the formation of bis-mesylated byproducts but also streamlines the purification workflow, positioning this technology as a cornerstone for reliable Dronedarone intermediate supplier operations aiming for high-purity outputs.
This structural refinement not only mitigates the formation of bis-mesylated byproducts but also streamlines the purification workflow, positioning this technology as a cornerstone for reliable Dronedarone intermediate supplier operations aiming for high-purity outputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dronedarone has been plagued by significant technical hurdles that impede efficient commercial scale-up of complex benzofuran derivatives. Conventional linear routes, such as those described in EP0471609B1, typically involve the reaction of nitro-substituted benzofurans followed by reduction and a challenging final mesylation step. A critical bottleneck in these legacy processes is the selective methanesulfonylation of the amino group in the presence of the bulky dibutylaminopropoxy side chain. This lack of chemoselectivity frequently leads to the formation of bis-mesylated derivatives, which are structurally similar to the target molecule and extremely difficult to separate without resorting to resource-intensive column chromatography. Furthermore, the reliance on mutagenic nitro-intermediates late in the synthesis poses severe safety and regulatory compliance risks for manufacturing facilities. The cumulative effect of these inefficiencies results in suboptimal yields, often reported around 61.6% after rigorous purification, and generates substantial chemical waste, thereby inflating the cost reduction in cardiovascular API manufacturing efforts.
The Novel Approach
In stark contrast, the methodology disclosed in CN103228639A revolutionizes the synthetic landscape by reversing the order of key functional group installations. This novel approach initiates with the methanesulfonylation of the simpler 2-n-butyl-5-aminobenzofuran scaffold, effectively locking in the sulfonamide motif before introducing the complex side chain. To protect this sensitive moiety during subsequent harsh reactions, the nitrogen is temporarily capped with an acyl group (R-CO), forming the stable intermediate of Formula III. 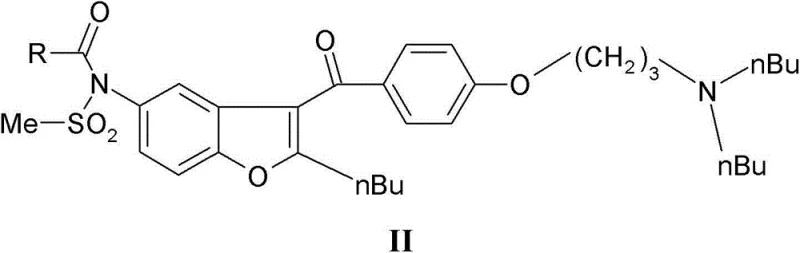 This strategic protection allows the molecule to withstand the vigorous conditions of the Friedel-Crafts acylation with the specialized benzoyl chloride derivative. The culmination of this sequence is the elegant selective cleavage of the acyl protecting group from the Formula II intermediate. By utilizing mild alkaline or acidic conditions, the temporary acyl mask is removed with high precision, yielding the target Dronedarone with exceptional purity profiles. This convergent strategy effectively bypasses the selectivity issues of the past, offering a streamlined pathway that is economically superior and technically more robust for industrial applications.
This strategic protection allows the molecule to withstand the vigorous conditions of the Friedel-Crafts acylation with the specialized benzoyl chloride derivative. The culmination of this sequence is the elegant selective cleavage of the acyl protecting group from the Formula II intermediate. By utilizing mild alkaline or acidic conditions, the temporary acyl mask is removed with high precision, yielding the target Dronedarone with exceptional purity profiles. This convergent strategy effectively bypasses the selectivity issues of the past, offering a streamlined pathway that is economically superior and technically more robust for industrial applications.
Mechanistic Insights into Selective Acyl Cleavage and Friedel-Crafts Acylation
The chemical elegance of this process is rooted in the precise orchestration of protection and deprotection chemistry coupled with classic electrophilic aromatic substitution. The synthesis begins with the formation of the N-acyl-N'-mesyl benzofuran (Formula III), which serves as a nucleophile in the subsequent Friedel-Crafts reaction. When reacted with 4-(3-di-n-butylaminopropoxy)benzoyl chloride (Formula IV) in the presence of a Lewis acid catalyst such as iron(III) chloride or aluminum chloride, the benzofuran ring undergoes acylation at the 3-position. 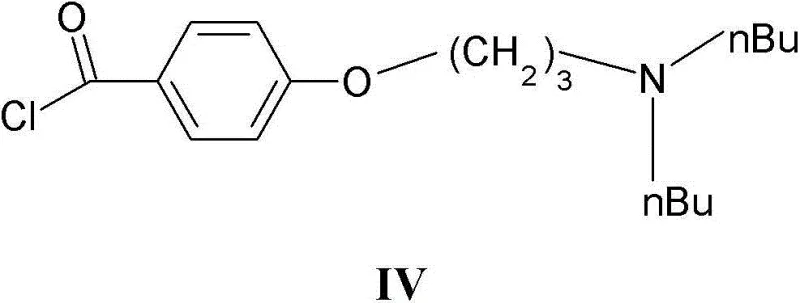 The presence of the N-acyl protecting group is mechanistically crucial; it reduces the electron density on the sulfonamide nitrogen, preventing it from coordinating with the Lewis acid catalyst or undergoing unwanted side reactions that could degrade the molecular integrity. Following the acylation, the resulting Formula II intermediate contains both the complex side chain and the protected sulfonamide. The final transformation involves the hydrolysis or alcoholysis of the amide bond linking the acyl group to the nitrogen. Using reagents like sodium methoxide in methanol or dilute mineral acids, the carbonyl carbon of the protecting group is attacked, leading to the cleavage of the C-N bond. This step regenerates the free sulfonamide proton while leaving the robust sulfonamide S-N bond intact, demonstrating remarkable chemoselectivity that is essential for maintaining high product quality.
The presence of the N-acyl protecting group is mechanistically crucial; it reduces the electron density on the sulfonamide nitrogen, preventing it from coordinating with the Lewis acid catalyst or undergoing unwanted side reactions that could degrade the molecular integrity. Following the acylation, the resulting Formula II intermediate contains both the complex side chain and the protected sulfonamide. The final transformation involves the hydrolysis or alcoholysis of the amide bond linking the acyl group to the nitrogen. Using reagents like sodium methoxide in methanol or dilute mineral acids, the carbonyl carbon of the protecting group is attacked, leading to the cleavage of the C-N bond. This step regenerates the free sulfonamide proton while leaving the robust sulfonamide S-N bond intact, demonstrating remarkable chemoselectivity that is essential for maintaining high product quality.
Impurity control is inherently built into this mechanistic design, addressing the primary concerns of R&D directors regarding杂质谱 (impurity profiles). In conventional routes, the free amine is susceptible to over-reaction, leading to bis-acylation or polymerization. By pre-installing the mesyl group and then protecting it, the reactive sites on the molecule are strictly defined. The selective cleavage step acts as a final "gatekeeper," ensuring that any unreacted acylated species can be hydrolyzed to the product, while the stable sulfonamide core remains untouched. The use of specific solvents like dichloromethane during the acylation and methanol during the cleavage further optimizes the solubility of intermediates, preventing precipitation of impurities and facilitating homogeneous reaction kinetics. This level of control ensures that the final crude product often exceeds 95% purity by HPLC, significantly reducing the burden on downstream purification units and aligning with the stringent purity specifications required for global regulatory submissions.
How to Synthesize Dronedarone Efficiently
The practical implementation of this novel synthesis route requires careful attention to reaction parameters to maximize yield and minimize impurity formation. The process generally follows a three-stage sequence: preparation of the protected benzofuran core, coupling with the side-chain precursor, and final deprotection. Operators must ensure strict temperature control during the Friedel-Crafts acylation, typically maintaining conditions between 5°C and 40°C depending on the specific Lewis acid employed, to prevent thermal degradation of the sensitive dibutylamino side chain. The choice of base for the final cleavage step is also critical; alkali metal alcoholates like sodium methoxide are preferred for their rapid kinetics and ease of removal. For a comprehensive understanding of the specific stoichiometric ratios, solvent volumes, and workup procedures detailed in the patent examples, please refer to the standardized synthesis guide below.
- Preparation of N-acyl-N'-mesyl benzofuran intermediate (Formula III) via acylation of 2-n-butyl-5-methanesulfonamidobenzofuran.
- Friedel-Crafts acylation of Formula III with 4-(3-di-n-butylaminopropoxy)benzoyl chloride (Formula IV) using Lewis acids like FeCl3 or AlCl3 to yield Formula II.
- Selective cleavage of the N-acyl protecting group from Formula II using alkali metal alcoholates or mineral acids to afford high-purity Dronedarone (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible operational efficiencies and risk mitigation. The primary economic driver is the elimination of chromatographic purification, which is notoriously expensive and difficult to scale in a GMP environment. By achieving high crude purity through chemical selectivity rather than physical separation, manufacturers can drastically reduce solvent consumption and processing time. This simplification of the downstream process directly contributes to cost reduction in cardiovascular API manufacturing, allowing for more competitive pricing structures without compromising on quality. Furthermore, the avoidance of mutagenic nitro-intermediates in the final stages of synthesis reduces the need for specialized containment equipment and extensive safety monitoring, lowering the overall capital expenditure required for production facilities.
- Cost Reduction in Manufacturing: The strategic use of a removable acyl protecting group eliminates the formation of hard-to-separate bis-mesylated byproducts, which traditionally necessitated costly column chromatography. By replacing this batch-intensive purification with simple crystallization or extraction steps, the process significantly lowers solvent usage and waste disposal costs. Additionally, the ability to use common Lewis acids like iron(III) chloride instead of more exotic or expensive catalysts further optimizes the raw material bill of materials, ensuring a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply continuity by reducing the risk of batch failures due to impurity spikes. The high selectivity of the cleavage reaction ensures consistent yields across different scales, from pilot plant to commercial tonnage. Moreover, the starting materials, such as 2-n-butyl-5-aminobenzofuran and the benzoyl chloride derivative, are accessible via established supply chains, minimizing the risk of raw material shortages. This reliability is crucial for maintaining steady inventory levels and meeting the demanding delivery schedules of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental perspective, the process offers substantial benefits by reducing the E-factor (mass of waste per mass of product). The avoidance of column chromatography and the use of recyclable solvents like dichloromethane and methanol align with green chemistry principles. The simplified workup procedures, often involving aqueous washes and phase separations, are easily adaptable to large-scale reactor systems. This scalability ensures that the transition from laboratory development to industrial production is seamless, supporting the commercial scale-up of complex benzofuran derivatives with minimal environmental footprint and regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Dronedarone synthesis process. These insights are derived directly from the experimental data and claims within CN103228639A, providing clarity on the feasibility and advantages of this method for potential partners and stakeholders.
Q: How does the novel process improve upon conventional Dronedarone synthesis methods?
A: The novel process described in CN103228639A overcomes the critical issue of selective mesylation found in linear routes. By introducing the methanesulfonyl group early on the simple benzofuran core and protecting it with a removable acyl group prior to the complex Friedel-Crafts step, the method avoids the formation of difficult-to-remove bis-mesylated byproducts and eliminates the need for column chromatography, significantly enhancing industrial feasibility.
Q: What are the key advantages of the selective acyl cleavage step?
A: The selective cleavage of the acyl group from the benzofuran derivative (Formula II) allows for the use of harsh Lewis acid conditions during the preceding acylation step without degrading the sensitive sulfonamide moiety. This protection-deprotection strategy ensures that the final deprotection can be performed under mild conditions (e.g., using sodium methoxide in methanol), yielding the final API with exceptional purity (up to 100% after salt formation) and high recovery rates suitable for commercial production.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial applicability. It utilizes common reagents such as iron(III) chloride or aluminum chloride for acylation and standard solvents like dichloromethane and methanol. The avoidance of mutagenic nitro-intermediates in the final steps and the elimination of complex purification techniques like column chromatography make this route highly scalable, cost-effective, and compliant with stringent GMP requirements for cardiovascular drug manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dronedarone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our technical team has thoroughly analyzed the innovative route described in CN103228639A and is fully equipped to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of the selective acyl cleavage and Friedel-Crafts steps are perfectly controlled at scale. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest international standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior manufacturing process. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements. Together, we can secure a more efficient, cost-effective, and reliable supply of high-quality Dronedarone intermediates for the global healthcare community.
