Advanced Manufacturing of Milnacipran Hydrochloride Intermediates via Novel Phthalimide Strategy
Advanced Manufacturing of Milnacipran Hydrochloride Intermediates via Novel Phthalimide Strategy
The pharmaceutical landscape for antidepressants continues to evolve, driven by the need for safer, more scalable manufacturing processes for active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN112759542B, which discloses a robust synthetic pathway for Milnacipran Hydrochloride, a potent serotonin-norepinephrine reuptake inhibitor used globally for treating fibromyalgia and major depressive disorder. This intellectual property introduces a critical intermediate, designated as Compound III, which serves as the cornerstone for a streamlined production sequence that circumvents the severe safety hazards and cost inefficiencies plaguing legacy methodologies. By leveraging a phthalimide protection strategy, the inventors have established a route that is not only chemically elegant but also industrially viable, offering a compelling value proposition for generic drug manufacturers seeking to optimize their supply chains for high-volume psychiatric medications.
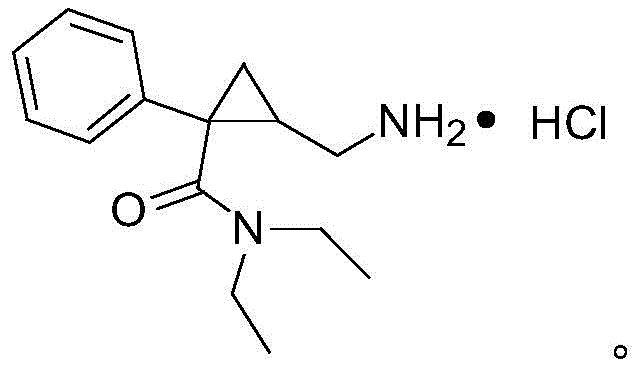
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Milnacipran and its precursors has been fraught with significant operational challenges that hinder large-scale commercialization. As illustrated in prior art literature, such as the route disclosed in Heterocyclic Letters, many conventional pathways rely heavily on the use of sodium azide to introduce the necessary nitrogen functionality. While chemically effective on a small laboratory scale, the use of azides presents unacceptable safety risks in an industrial setting due to their potential for explosive decomposition and high toxicity, necessitating expensive containment infrastructure and rigorous safety protocols. Furthermore, other documented approaches, such as those found in European patent EP200638, suffer from poor atom economy and harsh reaction conditions, often requiring high-temperature reactions in polar aprotic solvents like DMF which result in difficult purification processes and suboptimal yields. Additionally, catalytic methods reported in journals like Advanced Synthesis & Catalysis frequently employ expensive lithium metal complexes or palladium catalysts, which not only inflate the raw material costs but also introduce complex downstream processing requirements to ensure residual metal levels meet stringent regulatory limits for pharmaceutical products.
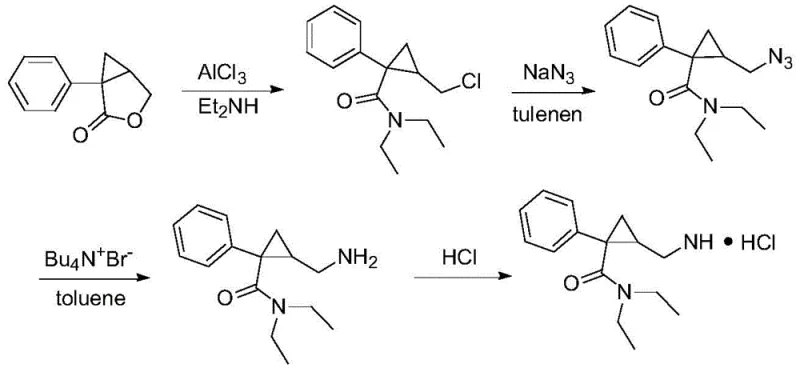
The Novel Approach
In stark contrast to these problematic precedents, the methodology outlined in patent CN112759542B offers a paradigm shift by utilizing a phthalimide-based protection group strategy that effectively eliminates the need for hazardous azides and precious metal catalysts. The core innovation lies in the preparation and utilization of Intermediate III, a stable phthalimide derivative that allows for the safe introduction of the amine moiety through standard nucleophilic substitution reactions. This approach transforms the synthesis into a series of high-yielding, predictable steps that utilize commodity chemicals such as thionyl chloride, potassium phthalimide, and hydrazine hydrate. The elimination of explosive reagents and expensive transition metals fundamentally alters the economic and safety profile of the manufacturing process, making it highly attractive for contract development and manufacturing organizations (CDMOs) aiming to reduce liability and operational expenditures. The transition from a chloromethyl precursor (Compound II) to the protected amine (Compound III) proceeds under mild thermal conditions, ensuring high selectivity and minimizing the formation of difficult-to-remove impurities that often plague multi-step syntheses.
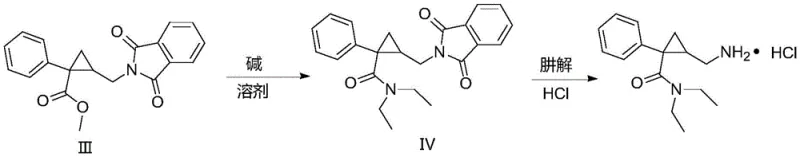
Mechanistic Insights into Phthalimide-Mediated Amine Installation
The chemical elegance of this new route is rooted in the classic Gabriel synthesis modification, adapted here for a sterically demanding cyclopropane scaffold. The process initiates with the ring-opening of the lactone precursor (Compound I) using thionyl chloride in methanol, which efficiently generates the chloromethyl ester (Compound II) with excellent conversion rates. This chloromethyl group serves as a highly reactive electrophile, poised for nucleophilic attack by the potassium salt of phthalimide. In the presence of a suitable solvent system, such as toluene or DMF, the phthalimide anion displaces the chloride ion to form the robust C-N bond found in Intermediate III. This step is crucial as it installs the nitrogen atom in a protected form that is inert to the subsequent amidation conditions, thereby preventing side reactions that could occur if a free amine were present during the formation of the diethylamide moiety. The stability of the phthalimide group allows the synthesis to proceed through the condensation with diethylamine under alkaline conditions without compromising the integrity of the newly formed amine linkage.
Following the formation of the diethylamide structure (Compound IV), the final liberation of the primary amine is achieved through hydrazinolysis, a reaction where hydrazine hydrate acts as a nucleophile to cleave the phthalimide ring, releasing phthalhydrazide as a byproduct and freeing the desired aminomethyl group. This deprotection step is highly specific and proceeds cleanly, avoiding the harsh acidic or basic hydrolysis conditions that might otherwise degrade the sensitive cyclopropane ring or cause racemization of the chiral centers. The mechanism ensures that the final product, Milnacipran Hydrochloride, is obtained with high chemical purity, as the phthalhydrazide byproduct can be easily separated from the amine salt during the workup procedure. This mechanistic pathway demonstrates a sophisticated understanding of orthogonal protecting group chemistry, applying fundamental organic principles to solve complex process development challenges in the manufacture of psychotropic drugs.
How to Synthesize Milnacipran Hydrochloride Efficiently
The practical implementation of this synthetic route requires careful attention to reaction parameters to maximize yield and purity, particularly during the substitution and amidation steps. The patent provides detailed experimental embodiments that serve as a blueprint for scaling this chemistry from gram-scale laboratory experiments to multi-kilogram pilot batches. Operators must strictly control the stoichiometry of the phthalimide salt and the reaction temperature to prevent over-alkylation or hydrolysis of the ester functionality. The following guide outlines the critical operational phases derived from the patent data, serving as a foundational reference for process engineers tasked with technology transfer.
- Preparation of Compound II: React the lactone precursor (Compound I) with thionyl chloride in methanol at room temperature to form the chloromethyl ester.
- Formation of Intermediate III: Perform a nucleophilic substitution using potassium phthalimide in toluene or DMF at 80-90°C to install the protected amine.
- Final Conversion: Condense Intermediate III with diethylamine under alkaline conditions to form the amide, followed by hydrazinolysis and salification to yield Milnacipran Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route represents a strategic opportunity to mitigate risk and enhance the economic viability of Milnacipran production. By shifting away from hazardous azide chemistry, manufacturers can significantly reduce the capital expenditure associated with specialized safety equipment and explosive-proof facilities, leading to a leaner cost structure. Furthermore, the reliance on commodity reagents rather than bespoke catalysts insulates the supply chain from the volatility of the precious metals market, ensuring more predictable raw material pricing and availability. The high yields reported in the patent examples suggest a substantial reduction in waste generation, which not only lowers disposal costs but also aligns with increasingly stringent environmental regulations governing pharmaceutical manufacturing. This process intensification allows for a more efficient use of reactor volume, potentially increasing overall plant throughput without the need for significant infrastructure expansion.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and lithium complexes removes a major cost driver from the bill of materials, while the avoidance of azides reduces the hidden costs associated with hazardous waste disposal and safety compliance. The use of inexpensive reagents like thionyl chloride and hydrazine hydrate further drives down the variable cost per kilogram, enabling competitive pricing in the generic pharmaceutical market. Additionally, the high selectivity of the phthalimide route minimizes the formation of byproducts, reducing the solvent and energy consumption required for purification steps such as chromatography or extensive recrystallization.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key reagents, including potassium phthalimide and diethylamine, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in raw material quality, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API formulators, thereby strengthening the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory flasks to industrial reactors without the need for complex engineering controls typically required for exothermic azide reactions. The waste stream is primarily composed of organic salts and phthalhydrazide, which are easier to treat and dispose of compared to heavy metal residues or toxic azide wastes. This environmental compatibility facilitates faster regulatory approvals and permits for new manufacturing lines, accelerating the time-to-market for generic versions of the drug and supporting sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the specific reaction conditions and impurity profiles associated with this patent. These insights are derived directly from the experimental data provided in the disclosure, offering a transparent view of the process capabilities and limitations. Understanding these technical nuances is essential for R&D directors planning to integrate this route into their existing manufacturing portfolios.
Q: Why is the phthalimide route safer than traditional azide methods?
A: Traditional routes often utilize sodium azide, which poses significant explosion risks and requires specialized handling equipment. The novel method replaces hazardous azides with stable potassium phthalimide, drastically improving industrial safety profiles.
Q: What are the yield advantages of this new synthetic pathway?
A: Experimental data within the patent indicates high efficiency, with the formation of Intermediate III achieving yields up to 89.5% and the final hydrazinolysis step reaching 85.3%, demonstrating superior material throughput compared to older low-yield methods.
Q: Does this process require expensive transition metal catalysts?
A: No, unlike prior art that relies on costly lithium complexes or palladium on carbon (Pd/C), this optimized route utilizes common organic bases and reagents, eliminating the need for expensive metal removal steps and reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milnacipran Intermediate Supplier
The technological advancements presented in patent CN112759542B highlight the immense potential for optimizing the production of Milnacipran Hydrochloride, yet translating this potential into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex psychiatric drug intermediates. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this phthalimide route, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical testing protocols. We understand that consistency is key in the pharmaceutical industry, and our dedicated process development team works tirelessly to refine reaction parameters, ensuring maximum yield and minimal impurity carryover for your final API.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative synthesis for their supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this azide-free methodology for your specific production volumes. We encourage you to reach out today to obtain specific COA data from our pilot runs and discuss comprehensive route feasibility assessments tailored to your project timelines. Let us help you secure a reliable, cost-effective, and safe supply of high-quality Milnacipran intermediates that will drive your product's success in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
