Advanced Synthesis of Milnacipran Hydrochloride Intermediates for Commercial Scale Production
Introduction to Next-Generation Milnacipran Synthesis
The pharmaceutical landscape for serotonin-norepinephrine reuptake inhibitors (SNRIs) demands robust, scalable, and economically viable synthetic routes for active pharmaceutical ingredients (APIs). Patent CN103613513A introduces a groundbreaking methodology for the preparation of Milnacipran hydrochloride, a critical medication for major depressive disorder and fibromyalgia. This intellectual property outlines a sophisticated five-step reaction sequence that transforms readily available lactone precursors into a novel intermediate, designated as Compound V, which is subsequently reduced and salified to yield the final therapeutic agent. The technical breakthrough lies in the strategic manipulation of the cyclopropane ring system, ensuring that every reaction step operates under mild conditions while maintaining exceptional stereochemical integrity. By prioritizing high yield and purity at each stage, this process addresses the longstanding challenges of impurity profiles that often plague traditional antidepressant manufacturing. For global supply chain stakeholders, this represents a pivotal shift towards more reliable pharmaceutical intermediates supplier networks capable of delivering consistent quality.
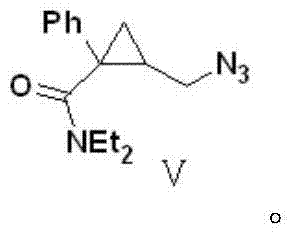
The core innovation focuses on the generation of Compound V, a structurally complex amide that serves as the direct precursor to the final drug substance. The patent details a pathway that circumvents the harsh thermal conditions and low-efficiency transformations characteristic of legacy methods. Instead, it leverages a sequence involving ring-opening esterification, nucleophilic azide substitution, hydrolysis, and amide coupling. This approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process by avoiding excessively high temperatures. As we delve deeper into the mechanistic nuances, it becomes evident that this route offers a superior alternative for the commercial scale-up of complex pharmaceutical intermediates, aligning perfectly with modern Good Manufacturing Practice (GMP) standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Milnacipran hydrochloride have been fraught with significant technical and economic inefficiencies that hinder large-scale production. For instance, the synthetic route disclosed in Patent EP0200638 relies on reaction conditions that necessitate elevated temperatures, which invariably lead to thermal degradation of sensitive intermediates and a consequent plummet in overall yield. This thermal instability creates a bottleneck in production capacity, forcing manufacturers to accept suboptimal recovery rates that drive up the cost per kilogram. Furthermore, alternative methods such as those described in Patent CN101107228 introduce additional complexities by utilizing sodium methylate in the initial steps. This reagent choice often results in the formation of cyclopropane ring-opened methyl ester byproducts, which are difficult to separate and negatively impact the purity of the final API. Additionally, the reliance on phthalimide potassium salts for amino group introduction in these older routes adds unnecessary synthetic steps and expensive reagent costs, complicating the supply chain logistics.
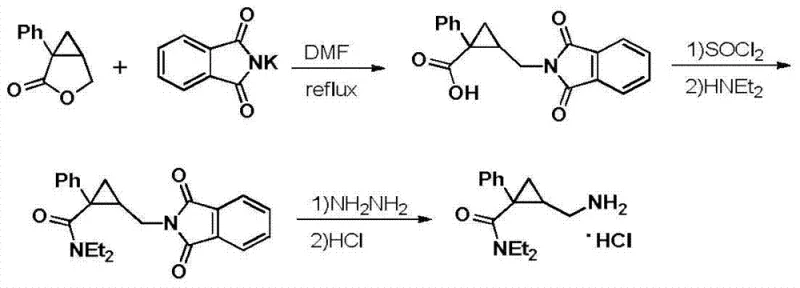
The Novel Approach
In stark contrast to these legacy methodologies, the process defined in CN103613513A offers a streamlined and chemically elegant solution that resolves these historical pain points. The new route initiates with a controlled ring-opening reaction using thionyl chloride in an alcoholic solvent, a transformation that proceeds efficiently at temperatures ranging from -10°C to 80°C, thereby preserving the structural integrity of the cyclopropane motif. By replacing the problematic phthalimide protection strategy with a direct azide substitution followed by catalytic reduction, the synthesis eliminates multiple steps and reduces the accumulation of toxic heavy metal impurities often associated with phthalimide removal. The operational simplicity is further enhanced by the use of common, industrially available solvents such as dichloromethane, toluene, and dimethylformamide. This strategic redesign of the synthetic tree ensures that the production of high-purity pharmaceutical intermediates is not only technically feasible but also economically superior, offering a clear pathway for cost reduction in API manufacturing without compromising on quality or safety standards.
Mechanistic Insights into the Five-Step Cascade Synthesis
The heart of this technological advancement lies in the precise orchestration of five distinct chemical transformations that convert the starting lactone (Compound I) into the advanced intermediate (Compound V). The first step involves the reaction of Compound I with thionyl chloride in methanol or ethanol, facilitating a ring-opening esterification that generates the chloro-ester derivative, Compound II. This step is critical as it installs the necessary leaving group for subsequent nucleophilic attack while establishing the ester functionality required for later hydrolysis. Following this, Compound II undergoes a nucleophilic substitution with sodium azide in an inert solvent like DMF or toluene at approximately 80°C. This azidation step is highly selective, displacing the chloride atom to form the azido-ester (Compound III) with excellent regioselectivity, ensuring that the cyclopropane ring remains intact despite the reactive conditions. The third phase involves the hydrolysis of the ester moiety in Compound III using an inorganic base such as sodium hydroxide. This saponification reaction cleanly converts the ester into the corresponding carboxylic acid (Compound IV), preparing the molecule for the crucial amide bond formation.
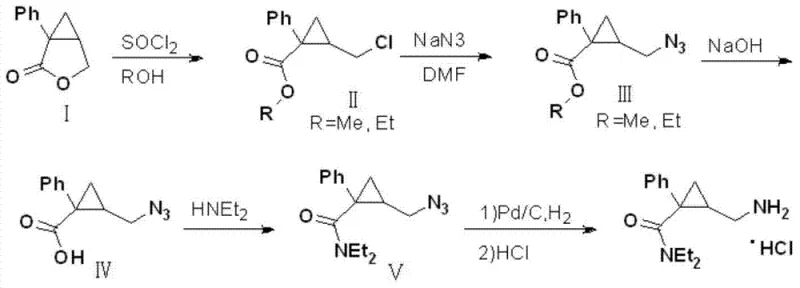
The fourth step represents a pivotal coupling reaction where Compound IV reacts with diethylamine in the presence of a coupling auxiliary like DCC (dicyclohexylcarbodiimide) or via a mixed anhydride method. This amidation constructs the diethylamide side chain, yielding the key intermediate Compound V, which possesses the complete carbon skeleton and functional group arrangement of the target drug. Finally, the azide group in Compound V is subjected to catalytic hydrogenation using Pd/C under hydrogen pressure. This reduction simultaneously converts the azide to a primary amine and allows for immediate salt formation with hydrochloric acid to precipitate the final Milnacipran hydrochloride product. Throughout this cascade, the control of impurities is paramount; the mild conditions prevent racemization and ring-opening side reactions, ensuring that the impurity profile remains well within regulatory limits. This mechanistic robustness provides R&D directors with the confidence that the process can be validated and transferred to commercial manufacturing sites with minimal risk of failure.
How to Synthesize Milnacipran Hydrochloride Intermediate Efficiently
Executing this synthesis requires strict adherence to the optimized parameters outlined in the patent to maximize yield and minimize waste. The process begins with the careful addition of thionyl chloride to the lactone substrate, followed by a controlled workup to isolate the chloro-ester. Subsequent azidation must be monitored closely to ensure complete conversion before proceeding to the hydrolysis step, where pH control is essential for isolating the free acid. The amidation step utilizes standard peptide coupling techniques, and the final reduction requires careful handling of hydrogen gas and catalyst filtration. For a comprehensive understanding of the specific stoichiometric ratios, solvent volumes, and isolation procedures, please refer to the detailed standardized synthesis guide below.
- Ring Opening: React the starting lactone (Compound I) with thionyl chloride in alcohol (methanol or ethanol) at temperatures between -10°C and 80°C to form the chloro-ester (Compound II).
- Azidation: Treat Compound II with sodium azide in an inert solvent like DMF or toluene at approximately 80°C to substitute the chloride with an azide group, yielding Compound III.
- Hydrolysis & Amidation: Hydrolyze the ester using an inorganic base to get the acid (Compound IV), then couple with diethylamine using DCC to form the key intermediate Compound V.
- Final Reduction: Reduce the azide group in Compound V using Pd/C and hydrogen gas, followed by salt formation with HCl to obtain Milnacipran Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound benefits for procurement managers and supply chain heads tasked with optimizing the sourcing of antidepressant APIs. The elimination of expensive and hazardous reagents like phthalimide potassium salts directly translates to a significant reduction in raw material costs, allowing for more competitive pricing structures in the final drug product. Furthermore, the simplified workflow reduces the number of unit operations required, which in turn lowers energy consumption and labor costs associated with batch processing. The high yields reported at each step mean that less starting material is wasted, enhancing the overall atom economy of the process and contributing to substantial cost savings in pharmaceutical intermediates manufacturing. These efficiencies create a buffer against market volatility in raw material prices, ensuring more stable long-term supply contracts.
- Cost Reduction in Manufacturing: The process achieves cost optimization by removing the need for costly protecting group strategies and reducing the number of purification steps. By utilizing common industrial solvents and reagents, the method avoids the premium pricing associated with specialized catalysts or exotic chemicals. The high conversion rates minimize the volume of waste solvent that needs to be treated or disposed of, further driving down operational expenditures. This lean manufacturing approach ensures that the cost of goods sold is minimized, providing a strong margin advantage for generic drug manufacturers.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as thionyl chloride, sodium azide, and diethylamine mitigates the risk of supply disruptions that often occur with niche reagents. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without requiring highly specialized equipment. This flexibility allows for dual-sourcing strategies and geographic diversification of production, significantly reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream formulation plants.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this route inherently safer and easier to scale from pilot plant to multi-ton production. The avoidance of extreme thermal conditions reduces the risk of runaway reactions, simplifying the engineering controls required for large-scale reactors. Additionally, the cleaner reaction profile results in fewer byproducts, easing the burden on wastewater treatment facilities and helping manufacturers meet stringent environmental regulations. This alignment with green chemistry principles enhances the corporate sustainability profile of the supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the new route. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and process validation.
Q: What are the critical advantages of this new synthesis route over EP0200638?
A: Unlike the prior art EP0200638 which suffers from high reaction temperatures and low overall yields, this novel method operates under mild conditions (-10°C to 80°C) and utilizes a highly efficient azide substitution strategy, resulting in significantly higher purity and yield for the final API.
Q: How does this process improve cost efficiency for procurement teams?
A: The process eliminates the need for expensive phthalimide potassium salts used in older routes (like CN101107228) and avoids complex purification steps associated with methyl ester byproducts. The use of common reagents like thionyl chloride and sodium azide, combined with high step-yields, drastically reduces the cost of goods sold (COGS).
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production. The reactions utilize standard solvents (DCM, Toluene, DMF) and avoid extreme pressures or cryogenic temperatures, making the commercial scale-up of complex pharmaceutical intermediates feasible and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milnacipran Hydrochloride Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex antidepressant intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Milnacipran intermediate meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this patented process, offering a seamless transition from development to commercial supply.
We invite you to collaborate with us to leverage this advanced synthesis technology for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
