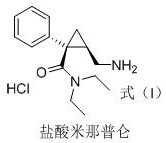
Advanced Synthesis of Milnacipran Hydrochloride Intermediate for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex chiral intermediates, particularly for antidepressant agents like Levomilnacipran Hydrochloride. Patent CN113493395A introduces a transformative preparation method that addresses critical bottlenecks in the manufacturing of this key SNRI inhibitor intermediate. The core innovation lies in a metal-free amination strategy that converts a chloro-substituted cyclopropane derivative directly into the corresponding aminomethyl intermediate using ammonia gas in a methanol solution. This approach not only simplifies the operational workflow but also significantly enhances the purity profile of the final active pharmaceutical ingredient (API) precursor. For R&D directors and technical procurement leaders, understanding the nuances of this patent is essential for evaluating supply chain resilience and cost efficiency. The method circumvents the limitations of traditional resolution techniques, which inherently cap yields at 50%, and avoids the safety hazards associated with azide chemistry. By leveraging this technology, manufacturers can achieve a more sustainable and economically viable production model for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levomilnacipran has relied heavily on chiral resolution or asymmetric synthesis routes that introduce significant operational and safety liabilities. One prevalent method involves the use of sodium azide to introduce the nitrogen functionality, a reagent known for its high explosive potential and stringent handling requirements. This reliance on hazardous materials creates substantial barriers to commercial scale-up, necessitating specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, alternative routes utilizing aluminum chloride for ring-opening reactions generate large volumes of aluminum-containing wastewater, posing severe environmental compliance challenges and increasing waste treatment costs. The presence of residual metals also complicates downstream purification, often requiring additional chromatography or crystallization steps to meet regulatory standards. These factors collectively result in a fragmented supply chain with higher production costs and increased risk of batch failure, making conventional methods less attractive for long-term commercial partnerships.
The Novel Approach
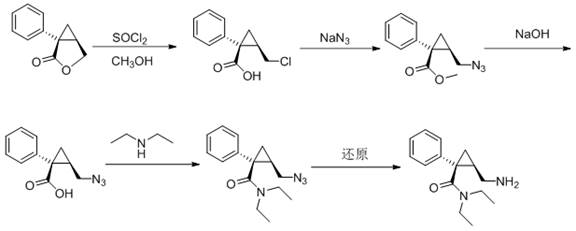
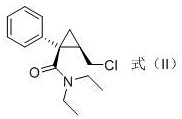
In stark contrast, the methodology disclosed in CN113493395A offers a streamlined pathway that eliminates these critical pain points through a direct nucleophilic substitution mechanism. The process utilizes a chloro-intermediate, specifically (1S,2R)-N,N-diethyl-2-chloromethyl-1-phenylcyclopropanecarboxamide, as the starting substrate. By introducing ammonia gas directly into a methanol solution containing this substrate, the reaction achieves efficient amination under mild thermal conditions, typically between 50°C and 100°C. This metal-free protocol ensures that the final product is devoid of heavy metal contaminants, thereby simplifying the quality control process and reducing the burden on analytical laboratories. The use of common solvents like methanol and isopropyl acetate further enhances the economic feasibility of the route, as these materials are readily available and cost-effective on a global scale. For procurement managers, this translates to a more stable supply chain with reduced dependency on specialized or hazardous reagents, ultimately leading to substantial cost savings in API manufacturing.
Mechanistic Insights into Metal-Free Ammonolysis
The core chemical transformation in this patented process is a nucleophilic substitution where the chlorine atom on the methyl side chain is displaced by an amino group derived from ammonia. This reaction proceeds through a standard SN2 mechanism, facilitated by the polar protic environment of the methanol solvent. The choice of methanol is critical, as it solubilizes both the organic substrate and the ammonia gas, creating a homogeneous reaction medium that maximizes collision frequency and reaction kinetics. Operating at temperatures around 80°C provides sufficient activation energy to drive the reaction to completion within 3 to 8 hours, ensuring high conversion rates without degrading the sensitive cyclopropane ring structure. The stereochemistry at the chiral centers is preserved throughout this transformation, which is vital for maintaining the biological activity of the final Levomilnacipran molecule. This mechanistic simplicity allows for precise control over impurity profiles, as side reactions are minimized by the absence of aggressive Lewis acids or oxidizing agents. For technical teams, this predictability is invaluable for establishing robust control strategies and defining critical process parameters (CPPs) during technology transfer.
Impurity control is another significant advantage of this metal-free approach, particularly regarding the elimination of inorganic residues. In traditional routes involving aluminum chloride or thionyl chloride, the removal of inorganic salts often requires extensive aqueous workups, which can lead to product loss and emulsion formation. The new method avoids these issues by generating ammonium chloride as the primary byproduct, which can be easily managed during the subsequent salification step. The final salification is performed using hydrogen chloride gas in isopropyl acetate, a solvent system that promotes the crystallization of the hydrochloride salt with high purity. This integrated approach minimizes the number of unit operations required, reducing the overall processing time and energy consumption. From a regulatory perspective, the simplified impurity spectrum facilitates the identification and quantification of potential genotoxic impurities, ensuring compliance with ICH M7 guidelines. This level of chemical elegance directly supports the goal of producing high-purity pharmaceutical intermediates suitable for direct use in final drug formulation.
How to Synthesize Levomilnacipran Intermediate Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent quality to maximize yield and purity. The process begins with the preparation of the chloro-intermediate, which is then dissolved in methanol at a specific molar ratio to ensure optimal solubility and reaction kinetics. Ammonia gas is then introduced into the system under controlled pressure and temperature, driving the amination reaction to completion. Following the reaction, the mixture is concentrated and treated to isolate the free amine, which is subsequently converted to the hydrochloride salt. The detailed standardized synthesis steps, including specific stoichiometry, addition rates, and workup procedures, are critical for reproducibility and are outlined in the technical guide below.
- Dissolve the chloro-intermediate (Formula II) in methanol at a molar ratio of 1: 5 and heat to 80°C.
- Introduce ammonia gas into the methanol solution to perform nucleophilic substitution, yielding the amino-intermediate (Formula III).
- Salify the resulting amino-intermediate with hydrogen chloride gas in isopropyl acetate to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of hazardous azides and heavy metal catalysts fundamentally alters the risk profile of the manufacturing process, leading to lower insurance premiums and reduced regulatory overhead. The use of commodity solvents and reagents ensures that the supply chain is resilient to market fluctuations, as these materials are sourced from a broad base of global suppliers. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of multinational pharmaceutical clients. Furthermore, the simplified purification process reduces the consumption of auxiliary materials and energy, contributing to a lower carbon footprint and aligning with corporate sustainability goals. These factors collectively position this technology as a strategic asset for companies seeking to optimize their cost structure while maintaining the highest standards of quality and safety.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents like sodium azide and aluminum chloride significantly lowers the raw material costs associated with production. Additionally, the simplified workup procedure reduces the need for specialized waste treatment facilities, leading to substantial operational savings. The high yield and purity of the process minimize material loss, ensuring that every kilogram of starting material is efficiently converted into valuable product. These efficiencies translate directly into a more competitive pricing structure for the final intermediate, providing a clear advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: By relying on readily available solvents like methanol and isopropyl acetate, the manufacturing process is less vulnerable to supply disruptions caused by geopolitical events or raw material shortages. The mild reaction conditions also reduce the risk of equipment failure or batch loss due to thermal runaway, ensuring consistent output. This reliability allows supply chain planners to forecast production with greater accuracy, reducing the need for safety stock and optimizing inventory levels. For global buyers, this means a dependable source of high-quality intermediates that can support long-term product launches without interruption.
- Scalability and Environmental Compliance: The metal-free nature of the reaction simplifies the environmental permitting process, as there is no need for complex heavy metal discharge controls. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, minimizing the risk of compliance violations. The process is inherently scalable, allowing for seamless transition from pilot plant to commercial production without significant re-engineering. This scalability ensures that supply can be rapidly ramped up to meet market demand, providing a strategic buffer against unexpected spikes in consumption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for stakeholders evaluating this route for potential adoption. Understanding these nuances is key to making informed decisions about technology transfer and supply chain integration.
Q: How does this new method improve safety compared to azide routes?
A: The patented method eliminates the use of sodium azide, a highly explosive reagent required in conventional asymmetric synthesis routes. By utilizing ammonia gas for amination, the process removes significant safety risks associated with handling energetic materials, making it far more suitable for industrial scale-up and reducing insurance and containment costs.
Q: What are the purity advantages of avoiding aluminum catalysts?
A: Traditional routes often employ aluminum chloride (AlCl3), which generates substantial aluminum-containing wastewater and complicates purification due to metal residue. This metal-free approach ensures the final product is free from heavy metal contamination, simplifying the purification workflow and ensuring compliance with stringent ICH Q3D guidelines for elemental impurities in pharmaceuticals.
Q: Is this process scalable for commercial production?
A: Yes, the reaction conditions are mild (50-100°C) and utilize common solvents like methanol and isopropyl acetate. The absence of cryogenic conditions or hazardous azides allows for straightforward engineering control, facilitating reliable commercial scale-up from pilot batches to multi-ton annual production without complex safety infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milnacipran Hydrochloride Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our CDMO expertise allows us to adapt complex pathways like the one described in CN113493395A for industrial application, ensuring that technical potential is realized as commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications. Our rigorous QC labs are equipped to handle the detailed impurity profiling required for modern API intermediates, ensuring that every batch meets the exacting standards of global regulatory agencies. By partnering with us, you gain access to a team that understands both the chemistry and the commercial imperatives of pharmaceutical manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your project. Our team is ready to provide specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a reliable supply of high-purity intermediates that drive your product success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
