Advanced Chiral Resolution of 5'-Methoxylaudanosine for Commercial Scale Manufacturing
Advanced Chiral Resolution of 5'-Methoxylaudanosine for Commercial Scale Manufacturing
The pharmaceutical industry's relentless pursuit of safer neuromuscular blocking agents has placed a premium on the stereochemical purity of key intermediates. Patent CN111004179B introduces a groundbreaking resolution method for 5'-methoxylaudanosine, a critical chiral building block in the synthesis of Mickuammonium chloride. This novel approach addresses the longstanding challenges of low yield and insufficient optical purity that have plagued previous manufacturing routes. By leveraging a sophisticated mixed-solvent recrystallization technique, the process ensures that the final active pharmaceutical ingredient meets the rigorous safety standards required to prevent cardiovascular toxicity. For R&D directors and procurement specialists, this technology represents a significant leap forward in process reliability and product quality control.
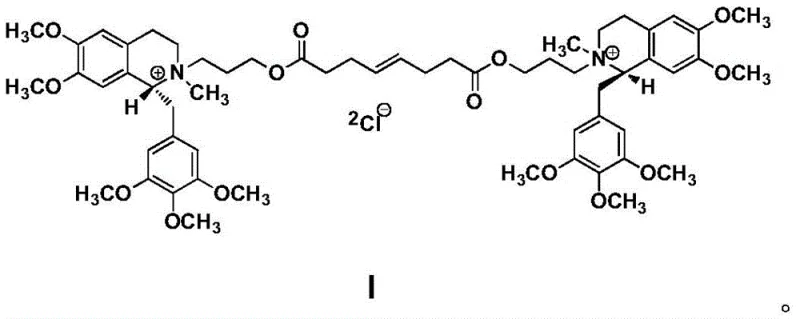
Mickuammonium chloride, depicted in the structural formula above, is a novel nerve muscle blocker characterized by high selectivity, rapid onset, and short duration of action without depolarization. Its clinical safety profile is heavily dependent on the absolute configuration of its chiral centers. Specifically, the molecule possesses two chiral carbon atoms and two quaternary ammonium nitrogen atoms. When these chiral carbons are in the R-configuration, the drug exhibits its therapeutic effects with minimal side effects. However, the presence of the S-configuration isomers can lead to adverse reactions such as histamine release and cardiovascular injury. Consequently, the synthesis of the precursor, R-5'-methoxylaudanosine, with ultra-high optical purity is not merely a quality metric but a critical safety imperative for the entire supply chain.
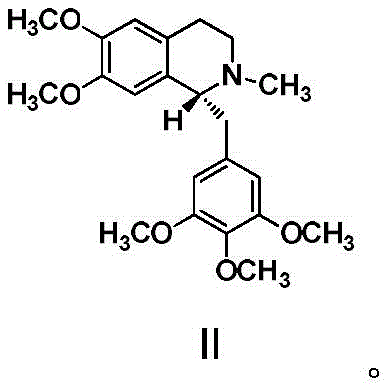
The structural integrity of R-5'-methoxylaudanosine, shown above, serves as the foundation for the stereochemistry of the final API. Existing synthetic routes have historically faced significant bottlenecks. Conventional resolution methods, such as those utilizing L/D-dibenzoyl tartaric acid reported in older literature like US4761418, often suffered from suboptimal yields that inflated production costs. Alternatively, direct chiral synthesis methods starting from 6,7-dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline frequently resulted in products with high impurity loads and enantiomeric excess values capped at roughly 95%. These limitations necessitated complex downstream purification steps, increasing both the environmental footprint and the lead time for reliable pharmaceutical intermediate supplier operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing pathways for this key intermediate were fraught with inefficiencies that impacted both cost reduction in pharmaceutical intermediates manufacturing and overall supply stability. The earlier resolution techniques often failed to adequately separate the diastereomeric salts, leading to a crude product that required multiple, yield-eroding recrystallization cycles. Furthermore, the chiral synthesis alternatives, while avoiding resolution steps, introduced complex catalytic systems that were difficult to scale and often left behind trace metal impurities. The inability to consistently achieve optical purity levels above 99% meant that manufacturers had to contend with variable batch quality, posing a risk to the regulatory approval of the final neuromuscular blocker. These process instabilities created a fragile supply chain vulnerable to disruptions and quality failures.
The Novel Approach
The methodology disclosed in patent CN111004179B fundamentally reengineers the crystallization thermodynamics to overcome these historical barriers. By employing a specific mixed solvent system comprising a first solvent (methanol or isopropanol) and a second solvent (methyl tert-butyl ether or isopropyl ether), the process creates a highly selective environment for crystal growth. The innovation lies not just in the solvent choice but in the precise temperature control protocol, maintaining recrystallization temperatures between 0°C and 30°C, with specific holding periods at 5°C and 15°C. This refined approach allows for the effective separation of the target R-5'-methoxylaudanosine dibenzoyl tartrate salt from impurities and the unwanted S-enantiomer. The result is a robust process capable of delivering yields exceeding 75% with chemical purity greater than 99.5% and optical purity surpassing 99.9%, setting a new benchmark for high-purity pharmaceutical intermediates.
Mechanistic Insights into Mixed-Solvent Recrystallization Resolution
The core of this technological advancement lies in the nuanced manipulation of solubility parameters through binary solvent systems. In the resolution of 5'-methoxylaudanosine, the formation of diastereomeric salts with D-(+)-dibenzoyltartaric acid monohydrate creates two distinct species with different physical properties. The novel process exploits the differential solubility of these salts in a mixture of polar protic solvents (like methanol) and less polar ethers (like MTBE). At elevated temperatures, the solvent mixture is tuned to dissolve the crude salt completely. Upon controlled cooling to 5°C, the solubility of the desired R-salt drops precipitously, inducing nucleation and crystal growth. Crucially, the specific ratio of solvents ensures that the S-salt and other organic impurities remain in the mother liquor, preventing their co-crystallization. This thermodynamic selectivity is the primary driver for the exceptional optical purity observed in the final product.
Furthermore, the temperature cycling protocol—cooling to 5°C, holding, and then warming to 15°C—plays a vital role in Ostwald ripening and defect correction within the crystal lattice. This step allows smaller, less perfect crystals to dissolve and redeposit onto larger, more stable crystals, effectively purifying the solid phase further. This mechanism ensures that the chemical purity remains consistently above 99.5%, eliminating the need for aggressive chromatographic purification steps that are costly and difficult to scale. For R&D teams, understanding this mechanism is key to transferring the process from the lab to commercial scale-up of complex pharmaceutical intermediates, as it relies on standard unit operations rather than exotic chemistry.
How to Synthesize R-5'-Methoxylaudanosine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this critical intermediate with industrial viability. The process begins with the resolution of racemic 5'-methoxylaudanosine using L-(-)-dibenzoyltartaric acid to remove the S-isomer, followed by the conversion of the remaining filtrate into the R-salt using D-(+)-dibenzoyltartaric acid. The pivotal step is the recrystallization of this crude R-salt in the optimized mixed solvent system. Detailed operational parameters, including specific solvent volumes relative to the crude mass and precise stirring times, are essential for reproducibility. The following guide summarizes the standardized synthesis steps derived from the patent examples to ensure consistent high-quality output.
- Dissolve racemic 5'-methoxylaudanosine in methanol and react with L-(-)-dibenzoyltartaric acid to form the S-salt, which is removed by filtration.
- Concentrate the filtrate, alkalize, and extract the free base. React the residue with D-(+)-dibenzoyltartaric acid to form the crude R-salt.
- Recrystallize the crude R-salt in a mixed solvent system (e.g., Methanol/MTBE) by heating to clarity, cooling to 5°C, and tempering at 15°C to maximize optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution method offers tangible strategic benefits beyond mere technical specifications. The shift from low-yield conventional methods to this high-efficiency process directly translates to substantial cost savings in raw material consumption. By achieving yields over 75% in the resolution step, the overall throughput of the manufacturing line is significantly increased without the need for additional reactor capacity. Moreover, the elimination of complex chiral catalysts and the reliance on commercially available resolving agents like dibenzoyltartaric acid simplifies the sourcing strategy, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of supply chain bottlenecks associated with specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the purification workflow. Traditional methods often required multiple recrystallizations or preparative chromatography to reach acceptable purity levels, both of which are resource-intensive. By achieving >99.9% optical purity in a single optimized recrystallization step, the process eliminates the need for expensive downstream processing. Additionally, the use of common solvents like methanol and methyl tert-butyl ether, which are easily recovered and recycled, further lowers the operational expenditure compared to processes requiring exotic or hazardous solvents. This efficiency ensures a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for life-saving medications like neuromuscular blockers. This method enhances reliability by utilizing robust, scalable chemistry that is less sensitive to minor fluctuations in reaction conditions. The starting materials, including 5'-methoxylaudanosine and dibenzoyltartaric acid derivatives, are readily available from multiple global suppliers, preventing single-source dependency. The high consistency of the output, with chemical purity consistently above 99.5%, reduces the rate of batch rejections and reworks. This predictability allows supply chain planners to maintain leaner inventory levels while ensuring uninterrupted production schedules for the finished drug product.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed for seamless transition from pilot plant to multi-ton production. The absence of transition metal catalysts removes the burden of extensive heavy metal clearance testing and validation, streamlining the regulatory filing process. The solvent system employed is compatible with standard industrial waste treatment protocols, facilitating easier compliance with increasingly stringent environmental regulations. The ability to scale this resolution method without compromising yield or purity makes it an ideal candidate for long-term commercial manufacturing, supporting the growing global demand for safe and effective anesthetic agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing synthesis pipelines.
Q: What is the critical advantage of the mixed solvent system in this resolution process?
A: The use of a mixed solvent system, specifically combining a polar protic solvent like methanol with a less polar ether like methyl tert-butyl ether, creates a precise solubility window. This allows for the selective crystallization of the target R-enantiomer salt while keeping impurities and the undesired S-enantiomer in solution, driving optical purity above 99.9%.
Q: Why is controlling the chiral configuration of 5'-methoxylaudanosine vital for the final API?
A: The final neuromuscular blocker, Mickuammonium chloride, contains four chiral centers. If the precursor 5'-methoxylaudanosine possesses the incorrect S-configuration at its chiral carbons, the resulting API can cause severe cardiovascular side effects and histamine release. Therefore, achieving >99.9% optical purity in the intermediate is a non-negotiable safety requirement.
Q: How does this method compare to previous chiral synthesis routes?
A: Previous asymmetric synthesis routes often struggled with impurity profiles and capped out at approximately 95% enantiomeric excess (ee). This resolution method, optimized with specific temperature gradients (5°C to 15°C) and solvent ratios, consistently achieves yields over 75% with chemical purity exceeding 99.5% and optical purity surpassing 99.9%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5'-Methoxylaudanosine Supplier
The resolution method detailed in patent CN111004179B underscores the critical importance of precise stereochemical control in modern drug synthesis. NINGBO INNO PHARMCHEM stands ready to leverage this advanced technology to support your development and commercialization goals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5'-methoxylaudanosine delivered meets the highest standards of chemical and optical purity required for GMP manufacturing.
We invite you to collaborate with us to optimize your supply chain for neuromuscular blocker production. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient resolution process can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to deliver high-quality intermediates that ensure the safety and efficacy of your final pharmaceutical products.
