Optimizing Ipratropium Bromide Production: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for critical respiratory medications like Ipratropium Bromide. A significant advancement in this domain is detailed in patent CN111269226A, which discloses a novel synthetic method that fundamentally restructures the production workflow. Unlike conventional approaches that rely on tropic acid and suffer from cumulative yield losses, this innovative protocol utilizes 2-phenyl-3-acetoxypropionic acid as the starting material. By integrating acyl chlorination, acylation, and hydrolysis into a unified reaction system, the process eliminates the need for intermediate isolation, thereby drastically reducing material consumption and processing time. This technical evolution represents a paradigm shift for manufacturers aiming to secure a reliable ipratropium bromide supplier capable of delivering consistent quality at scale.
Furthermore, the patent highlights the critical importance of stereochemical control, ensuring that the final product meets stringent regulatory requirements for enantiomeric purity. The method achieves a mass yield exceeding 75%, a substantial improvement over the industry standard of approximately 45%, while maintaining an HPLC purity of greater than 99.9%. For procurement and supply chain leaders, this translates to a more predictable production schedule and reduced waste generation. The ability to conduct key reactions under mild conditions, specifically avoiding high-temperature hazards associated with volatile reagents, further underscores the safety and viability of this route for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ipratropium Bromide has been plagued by inefficiencies inherent in multi-step batch processes that require rigorous purification between each stage. Traditional routes typically commence with tropic acid, necessitating sequential acyl chlorination, acylation, alcoholysis, and methylation reactions. Each transition between these steps traditionally mandates a work-up procedure involving separation, purification, and drying of the intermediate product to prevent carryover impurities. While these measures aim to enhance final purity, they inevitably lead to significant mechanical losses of material, driving the overall yield down to roughly 45%. Moreover, the repeated handling of intermediates increases the risk of contamination and introduces variability in the impurity profile, complicating the validation process for regulatory compliance. The cumulative time and resource expenditure required for these isolated steps render the conventional method economically burdensome and environmentally less sustainable due to higher solvent and energy consumption.
The Novel Approach
In stark contrast, the methodology described in CN111269226A introduces a telescoped strategy that streamlines the initial reaction phases into a continuous flow within a single reactor. By reacting 2-phenyl-3-acetoxypropionic acid directly with oxalyl chloride and subsequently introducing isopropyltropine mesylate without isolating the acid chloride intermediate, the process preserves the integrity of the reactive species. The subsequent in-situ hydrolysis of the acetyl group further simplifies the workflow, removing the need for a separate deprotection step. This consolidation not only accelerates the reaction kinetics but also significantly mitigates the loss of product associated with physical transfers and extractions. The result is a streamlined operation that boosts yield to over 75% while simultaneously simplifying the equipment requirements, making it an ideal candidate for cost reduction in API manufacturing where throughput and efficiency are paramount.
Mechanistic Insights into Telescoped Acylation and Quaternization
The core of this synthetic breakthrough lies in the precise management of reaction thermodynamics and stereochemistry during the formation of the ester linkage and the subsequent quaternary ammonium salt. The initial acyl chlorination using oxalyl chloride generates a highly reactive acid chloride species which immediately undergoes nucleophilic attack by the isopropyltropine mesylate. This tandem reaction is facilitated by the use of dichloromethane as a solvent, which provides an optimal medium for solubilizing both the organic acid and the amine salt. Following the acylation, the introduction of inorganic acid triggers the hydrolysis of the acetoxy group, revealing the essential 3-hydroxy functionality required for biological activity. This one-pot transformation minimizes the exposure of sensitive intermediates to external environments, thereby reducing the formation of degradation by-products that often complicate downstream purification efforts.
Crucially, the structural integrity of the tropane ring and the stereochemistry at the chiral centers must be preserved to ensure therapeutic efficacy. The active pharmaceutical ingredient relies on a specific spatial configuration to effectively block muscarinic receptors in the bronchial smooth muscle. However, synthetic processes can inadvertently generate enantiomers that lack pharmacological activity and are classified as impurities by global pharmacopoeias. 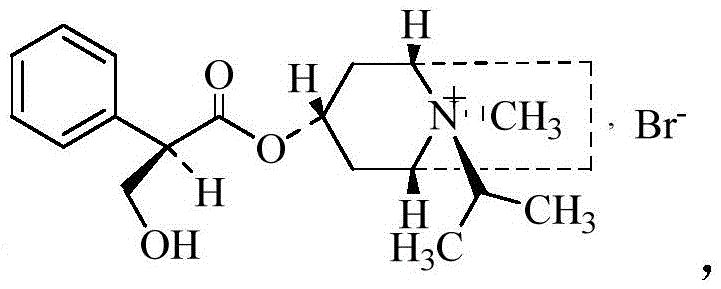 As illustrated in the active structure, the specific arrangement of the phenyl and hydroxy groups is critical. Conversely, the formation of the opposite enantiomer,
As illustrated in the active structure, the specific arrangement of the phenyl and hydroxy groups is critical. Conversely, the formation of the opposite enantiomer, 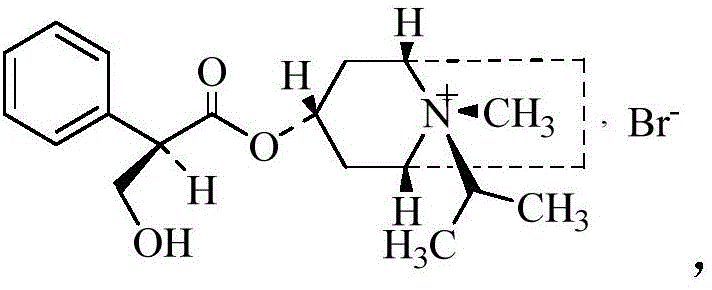 represents a potential liability. The patented process addresses this by optimizing the bromomethylation conditions—specifically maintaining temperatures between 0-5°C and utilizing vacuum pressures ≤-0.04MPa—to kinetically favor the formation of the desired quaternary ammonium salt while suppressing racemization or the formation of the inactive isomer, ensuring the final enantiomeric impurity content remains below 0.1%.
represents a potential liability. The patented process addresses this by optimizing the bromomethylation conditions—specifically maintaining temperatures between 0-5°C and utilizing vacuum pressures ≤-0.04MPa—to kinetically favor the formation of the desired quaternary ammonium salt while suppressing racemization or the formation of the inactive isomer, ensuring the final enantiomeric impurity content remains below 0.1%.
How to Synthesize Ipratropium Bromide Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of reagents and the control of reaction parameters to maximize the benefits of the telescoped design. The process begins with the dissolution of 2-phenyl-3-acetoxypropionic acid in dichloromethane, followed by the controlled addition of oxalyl chloride to generate the acyl chloride in situ. Once this conversion is complete, the isopropyltropine mesylate solution is introduced to effect the coupling. The subsequent hydrolysis step is performed directly in the reaction mixture using a mineral acid, after which the product is extracted into an organic phase. The final quaternization with methyl bromide is conducted under strictly controlled low-temperature and vacuum conditions to ensure safety and selectivity. For a detailed breakdown of the specific molar ratios, addition rates, and temperature profiles required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Perform acyl chlorination of 2-phenyl-3-acetoxypropionic acid with oxalyl chloride in dichloromethane, followed by direct acylation with isopropyltropine mesylate.
- Conduct in-situ hydrolysis of the acetyl group using inorganic acid within the same reaction system to generate the free hydroxyl intermediate.
- Extract the hydrolyzed product, adjust pH to 9.5-10.5, and perform quaternization with methyl bromide under vacuum at 0-5°C to obtain the final bromide salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for supply chain stability and cost management. The primary driver of value is the substantial increase in mass yield, which moves from the historical baseline of roughly 45% to over 75%. This near-doubling of efficiency means that for every kilogram of starting material purchased, the output of saleable API is significantly higher, directly impacting the cost of goods sold. Furthermore, the elimination of intermediate isolation steps reduces the demand for auxiliary materials such as filtration media, drying agents, and additional solvents, leading to a leaner manufacturing footprint. The simplified workflow also shortens the overall cycle time per batch, allowing facilities to increase throughput without requiring capital investment in new reactor capacity, thereby enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The economic benefits are driven by the telescoped nature of the reaction sequence, which removes the need for multiple work-up and purification stages. By avoiding the isolation of the acid chloride and the acylated intermediate, the process saves significant amounts of solvent and energy that would otherwise be consumed in evaporation and drying operations. Additionally, the use of dichloromethane, which is easily recovered and recycled, further lowers the variable costs associated with solvent procurement and waste disposal. This efficiency gain allows for a more competitive pricing structure for the final API, providing a strategic advantage in cost-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of this method contributes to a more reliable supply of high-purity pharmaceutical intermediates. Traditional methods with multiple isolation points are prone to bottlenecks; if one purification step fails or yields low recovery, the entire batch is compromised. By consolidating the critical steps into a single vessel, the risk of batch failure due to handling errors is minimized. Moreover, the starting material, 2-phenyl-3-acetoxypropionic acid, is readily available, reducing the risk of raw material shortages. This stability ensures that production schedules can be maintained consistently, safeguarding against disruptions that could impact downstream drug formulation and patient access.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating under mild conditions that do not require extreme heating or high-pressure equipment beyond standard industrial capabilities. The low-temperature requirement for the final methylation step enhances operational safety by mitigating the risks associated with volatile methyl bromide. From an environmental standpoint, the reduction in solvent usage and the ability to recycle dichloromethane align with green chemistry principles, reducing the facility's environmental footprint. This compliance with stricter environmental regulations future-proofs the manufacturing process against evolving sustainability mandates, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's feasibility. Understanding these details is essential for technical teams assessing the transferability of the process to their own manufacturing sites and for procurement officers evaluating the long-term viability of the supply source.
Q: How does this new synthesis route improve yield compared to traditional methods?
A: Traditional methods using tropic acid often yield around 45% due to multiple isolation steps. This patented route achieves over 75% yield by telescoping acyl chlorination, acylation, and hydrolysis into a single pot, minimizing material loss.
Q: What is the control strategy for enantiomeric impurities in this process?
A: The process utilizes precise temperature control during the bromomethylation step (0-5°C) and specific extraction protocols to ensure the enantiomeric impurity content remains below 0.1% by HPLC, meeting strict pharmacopoeia standards.
Q: Is the solvent system suitable for large-scale industrial production?
A: Yes, the process primarily uses dichloromethane, which is easily evaporated and recovered. The low-temperature requirements for the final quaternization step also enhance operational safety and scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ipratropium Bromide Supplier
The technical advancements outlined in patent CN111269226A demonstrate a clear pathway to producing Ipratropium Bromide with superior efficiency and purity. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this optimized route to life. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the highest international standards. We understand the critical nature of respiratory APIs and are committed to delivering a product that not only meets but exceeds the expectations of global regulatory bodies, ensuring safety and efficacy for the end patient.
We invite you to collaborate with us to leverage this technology for your supply chain needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this high-yield route can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a stable, high-quality supply of Ipratropium Bromide, backed by our commitment to innovation and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
