Scalable Production of High-Purity Ipratropium Bromide via Optimized One-Pot Synthesis
Scalable Production of High-Purity Ipratropium Bromide via Optimized One-Pot Synthesis
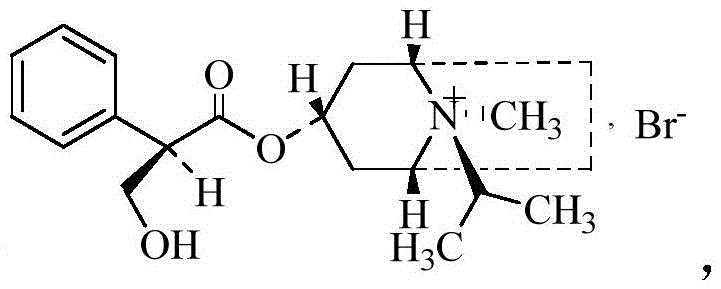
The pharmaceutical landscape for respiratory therapeutics continues to demand robust, scalable, and highly pure active pharmaceutical ingredients (APIs), with Ipratropium Bromide standing as a cornerstone treatment for chronic obstructive pulmonary disease (COPD) and asthma. Recent advancements in process chemistry, specifically detailed in patent CN111269226B, have introduced a transformative synthesis route that addresses long-standing inefficiencies in manufacturing this critical quaternary ammonium compound. This innovative methodology shifts away from fragmented, multi-step isolation protocols towards a streamlined, one-pot strategy that integrates acyl chlorination, esterification, and hydrolysis within a single reaction vessel. By fundamentally re-engineering the synthetic pathway, manufacturers can now achieve superior control over stereochemistry and impurity profiles while drastically reducing operational complexity. The following technical analysis dissects this breakthrough, highlighting its implications for R&D feasibility, cost structures, and supply chain resilience in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Ipratropium Bromide has been plagued by inherent inefficiencies associated with the traditional tropine acid route. Conventional processes typically necessitate a sequential series of discrete reactions, including acyl chlorination, acylation, alcoholysis, and methylation, each requiring rigorous intermediate purification and separation. This fragmented approach creates significant bottlenecks; every isolation step introduces opportunities for material loss, leading to cumulative yields that often stagnate around 45%. Furthermore, the extensive handling of intermediates increases the risk of cross-contamination and introduces variability in the final impurity profile, particularly regarding enantiomeric excess. The reliance on multiple purification cycles not only inflates the consumption of solvents and reagents but also extends the overall production cycle time, thereby increasing capital expenditure on equipment occupancy and labor. Consequently, the traditional workflow struggles to meet the dual demands of modern pharmaceutical manufacturing: high throughput and stringent quality consistency.
The Novel Approach
In stark contrast, the novel synthesis method disclosed in patent CN111269226B orchestrates a highly efficient convergence of reaction steps that eliminates the need for intermediate isolation. By reacting 2-phenyl-3-acetoxypropionic acid directly with oxalyl chloride and subsequently introducing isopropyl tropine mesylate in the same system, the process creates a seamless flow from acyl chloride formation to esterification. Crucially, the subsequent hydrolysis is performed in situ by adding inorganic acid to the residual reaction liquid, bypassing the need to isolate the unstable ester intermediate. This telescoping of reactions not only preserves the integrity of the chiral centers but also minimizes exposure to environmental contaminants. The result is a dramatic enhancement in process mass intensity, where the elimination of purification steps between the acyl chlorination and hydrolysis phases leads to a substantial reduction in waste generation and solvent usage. This integrated approach represents a paradigm shift from batch-and-hold processing to continuous-flow-like efficiency within a batch reactor setup.
Mechanistic Insights into One-Pot Esterification and Hydrolysis
The core of this technological advancement lies in the precise manipulation of reaction kinetics and thermodynamics during the acyl chlorination and esterification phases. The use of oxalyl chloride as the chlorinating agent is particularly strategic; it reacts with 2-phenyl-3-acetoxypropionic acid to form the reactive acyl chloride intermediate while generating gaseous byproducts (CO and CO2) that drive the equilibrium forward and simplify the reaction mixture. The addition of dimethylformamide (DMF) acts as a nucleophilic catalyst, forming a Vilsmeier-Haack type intermediate that significantly accelerates the acylation rate without requiring excessive thermal energy. Following this, the introduction of isopropyl tropine mesylate allows for immediate nucleophilic attack on the acyl chloride, forming the ester linkage essential for the drug's pharmacological activity. The subsequent hydrolysis step, triggered by the addition of hydrochloric acid, selectively removes the acetyl protecting group under mild conditions (20-30°C), ensuring that the sensitive tropane ring structure remains intact while exposing the hydroxyl group required for the final quaternization.
Stereochemical control is paramount in the synthesis of Ipratropium Bromide, as the biological activity is strictly dependent on the specific (1R,3R,5S,8R) configuration. The novel process mitigates the formation of inactive enantiomers through strict temperature regulation and the avoidance of harsh basic conditions that could induce racemization. By maintaining the bromomethylation reaction at low temperatures (0-5°C) and under controlled vacuum conditions (≤-0.04 MPa), the method suppresses side reactions that typically lead to epimerization at the chiral centers. The extraction protocol, utilizing dichloromethane at a specific pH range of 9.5-10.5, further refines the stereochemical purity by selectively partitioning the desired isomer into the organic phase while leaving polar impurities and potential enantiomeric contaminants in the aqueous layer. This meticulous control results in a final product with an enantiomer content of less than 0.1%, far exceeding the stringent requirements of international pharmacopoeias and ensuring consistent therapeutic efficacy.
How to Synthesize Ipratropium Bromide Efficiently
Implementing this advanced synthesis route requires adherence to specific operational parameters to maximize yield and safety. The process begins with the dissolution of 2-phenyl-3-acetoxypropionic acid in dichloromethane, followed by the controlled addition of oxalyl chloride to initiate acyl chlorination. Once the acyl chloride is formed, isopropyl tropine mesylate is introduced to effect esterification, after which the solvent is removed and hydrolysis is induced with inorganic acid. The resulting hydrolysate undergoes a specialized extraction sequence to isolate the free base intermediate, which is then subjected to bromomethylation with methyl bromide under vacuum. For a comprehensive breakdown of the standardized operating procedures, including exact molar ratios, addition rates, and safety protocols, please refer to the detailed guide below.
- Perform acyl chlorination of 2-phenyl-3-acetoxypropionic acid with oxalyl chloride in dichloromethane, followed by reaction with isopropyl tropine mesylate and subsequent hydrolysis.
- Extract the hydrolysate using dichloromethane, adjust the aqueous phase pH to 9.5-10.5, and re-extract to isolate the high-purity organic intermediate.
- Conduct bromomethylation with methyl bromide under vacuum (≤-0.04 MPa) at 0-5°C, followed by drying to obtain the final Ipratropium Bromide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers profound advantages for procurement managers and supply chain directors seeking to optimize their API sourcing strategies. The most immediate impact is observed in the drastic improvement of overall process yield, which jumps from the industry standard of approximately 45% to over 75%. This near-doubling of efficiency translates directly into a significant reduction in the cost of goods sold (COGS), as less raw material is required to produce the same quantity of finished API. Furthermore, the elimination of intermediate purification steps reduces the consumption of auxiliary chemicals and solvents, lowering both material costs and the expenses associated with waste disposal and environmental compliance. The simplified workflow also shortens the manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The consolidation of multiple reaction steps into a single pot significantly lowers operational expenditures by reducing energy consumption and labor hours. By avoiding the repeated heating, cooling, and filtration cycles associated with intermediate isolation, the facility can achieve substantial utility savings. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the need for expensive chromatographic purification or recrystallization processes. This streamlined approach ensures that the cost reduction in API manufacturing is driven by fundamental process intensification rather than mere economies of scale, providing a sustainable competitive advantage.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the number of potential failure points. Traditional methods with multiple isolation steps are prone to yield losses at each stage, creating variability in output volumes that can disrupt downstream formulation schedules. In contrast, the high-yield, one-pot nature of this new method ensures consistent batch sizes and predictable delivery timelines. Moreover, the use of common, readily available reagents like oxalyl chloride and dichloromethane mitigates the risk of raw material shortages, ensuring that production can be maintained even during periods of global supply chain stress.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations that can be easily adapted from pilot scale to commercial production of 100 MT or more. The ability to recover and recycle dichloromethane efficiently aligns with green chemistry principles, reducing the environmental footprint of the manufacturing operation. By operating at lower temperatures and avoiding high-pressure conditions during the critical methylation step, the process also enhances plant safety, lowering insurance premiums and regulatory scrutiny. This combination of scalability and safety makes the technology an ideal candidate for long-term strategic partnerships focused on sustainable API production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing supplier capabilities.
Q: How does the new synthesis method improve yield compared to traditional processes?
A: The novel method achieves a mass yield of over 75% by eliminating intermediate purification steps and utilizing a one-pot reaction system, whereas traditional methods typically yield around 45% due to material loss during multiple isolation stages.
Q: What is the enantiomeric purity of the Ipratropium Bromide produced?
A: The process ensures strict stereochemical control, resulting in an HPLC enantiomer content of less than 0.1%, meeting rigorous pharmacopoeia standards for this anticholinergic agent.
Q: Is the bromomethylation step safe for large-scale manufacturing?
A: Yes, the reaction is conducted at low temperatures (0-5°C) and controlled vacuum conditions (≤-0.04 MPa), significantly reducing the safety risks associated with volatile methyl bromide in closed high-temperature environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ipratropium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN111269226B are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, employing state-of-the-art analytical techniques to verify that every batch of Ipratropium Bromide meets the highest global standards for enantiomeric purity and residual solvent content. Our commitment to quality assurance ensures that your supply of this critical respiratory API remains uninterrupted and compliant with all regulatory requirements.
We invite you to engage with our technical procurement team to explore how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impacts of switching to this high-efficiency process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a reliable, cost-effective, and high-quality supply of Ipratropium Bromide that supports your mission to deliver life-saving therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
