Advanced Manufacturing of Ipratropium Bromide: A High-Yield, Green Methylation Strategy for Global Pharma Supply Chains
The pharmaceutical landscape for respiratory treatments continues to demand robust, scalable, and environmentally conscious manufacturing processes for critical active ingredients. Patent CN111978316B introduces a transformative synthesis method for Ipratropium Bromide, a potent anticholinergic drug widely used in managing Chronic Obstructive Pulmonary Disease (COPD) and asthma. This technology addresses long-standing inefficiencies in the quaternization of tropine derivatives by implementing a sophisticated temperature-gradient stirring protocol and a staged reagent addition strategy. Unlike traditional batch processes that struggle with incomplete conversion and hazardous waste generation, this novel approach leverages excessive methyl bromide coupled with a closed-loop recovery system. For global supply chain leaders, this represents a pivotal shift towards sustainable API intermediate manufacturing, ensuring that the production of this life-saving bronchodilator is not only economically viable but also aligns with stringent environmental regulations regarding ozone-depleting substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ipratropium Bromide has been plagued by suboptimal reaction kinetics and significant material wastage. Conventional protocols typically involve dissolving the tropine-N-isopropyl nortropine ester intermediate in anhydrous chloroform and introducing methyl bromide in a single bulk addition at low temperatures, often around 0°C. This brute-force approach frequently results in a reaction yield capped at approximately 80%, leaving a substantial portion of valuable starting material unconverted. Furthermore, the highly volatile and toxic nature of methyl bromide makes its containment difficult in open or semi-open systems, leading to direct discharge into waste streams. This not only poses severe safety risks to operators due to toxicity but also creates a heavy environmental burden, as the unrecovered reagent contributes to ozone layer depletion. The inability to recycle this expensive reagent drives up the variable cost of goods sold (COGS) and complicates waste treatment logistics for manufacturers.
The Novel Approach
The methodology outlined in the patent fundamentally reengineers the methylation step to maximize atomic economy and operational safety. By shifting from a single-dose addition to a staged introduction of methyl bromide—performed at least twice with specific time intervals—the process maintains an optimal concentration gradient that drives the equilibrium towards the product. Crucially, the reaction does not remain static at low temperatures; instead, it employs a progressive heating profile, moving from -5°C up to 50°C across three distinct stirring phases. This thermal ramping ensures that the activation energy barriers for the quaternization are overcome systematically, facilitating near-complete conversion of the intermediate. The result is a dramatic improvement in yield, consistently reaching levels between 95.9% and 98.8%, while simultaneously enabling the efficient distillation and recovery of unreacted methyl bromide for reuse in future batches.
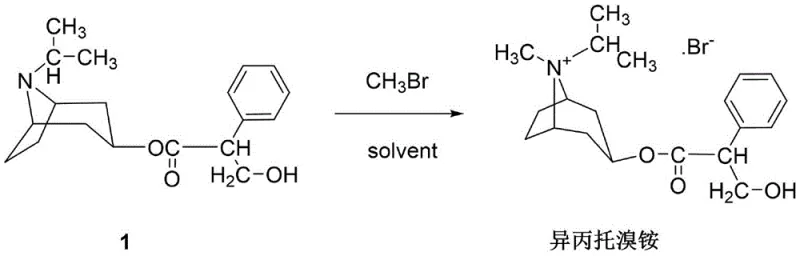
Mechanistic Insights into Temperature-Gradient Quaternization
The core chemical transformation involves the nucleophilic attack of the tertiary amine nitrogen on the methyl carbon of methyl bromide, resulting in the formation of a quaternary ammonium salt. In standard conditions, steric hindrance around the nitrogen atom in the tropane ring system can slow down this alkylation, especially as the product precipitates or viscosity changes. The patented process mitigates this by utilizing a significant molar excess of methyl bromide (ratio of 7 to 9.5:1), effectively flooding the reaction environment to drive the kinetics forward according to the law of mass action. However, simply adding excess reagent is insufficient without precise thermal management. The initial prolonged stirring at -5°C to 0°C for 15 to 17 hours allows for the controlled formation of the salt without triggering rapid exothermic spikes that could degrade the sensitive ester linkage. Subsequent heating to ambient and then elevated temperatures (40-50°C) serves to solubilize any occluded reactants and ensures the final push to completion, effectively eliminating residual starting materials that would otherwise appear as impurities in the final assay.
Impurity control is another critical dimension where this mechanistic approach excels. In respiratory APIs, the profile of related substances is strictly regulated due to the direct delivery to the lungs. The gradual temperature increase prevents the formation of thermal degradation byproducts that might occur if the reaction were heated too aggressively at the start. Moreover, the recovery step acts as a purification mechanism in itself; by distilling off the volatile methyl bromide, the system removes potential halogenated impurities that could form under harsher conditions. The final recrystallization from a methanol-acetone mixture further refines the crystal lattice, excluding structurally similar analogs. This rigorous control over the reaction trajectory ensures that the content of related substances remains well below the limits specified by the European Pharmacopoeia, delivering a high-purity API intermediate that simplifies downstream formulation processing for pharmaceutical partners.
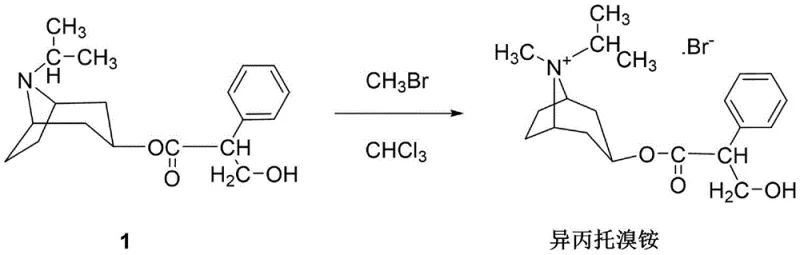
How to Synthesize Ipratropium Bromide Efficiently
Implementing this advanced synthesis route requires precise adherence to the defined operational parameters to replicate the high yields and purity demonstrated in the patent examples. The process begins with the dissolution of the tropine ester precursor in chloroform, followed by a carefully controlled cooling phase. The critical innovation lies in the dosing strategy of the methylating agent and the subsequent thermal profile, which must be managed to balance reaction rate with safety. Operators must utilize equipment capable of handling cryogenic temperatures and pressurized gas addition safely. The following guide outlines the standardized workflow derived from the patent data, serving as a foundational reference for process engineers looking to adopt this superior manufacturing technique for commercial scale-up of complex pharmaceutical intermediates.
- Dissolve tropine-N-isopropyl nortropine ester in chloroform and cool the mixture to between -5°C and 0°C.
- Introduce excessive methyl bromide in at least two stages while maintaining the low temperature, followed by a multi-stage stirring process ranging from -5°C up to 50°C.
- Recover unreacted methyl bromide via distillation, filter the product, wash with chloroform, and recrystallize to obtain high-purity Ipratropium Bromide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates directly into enhanced operational resilience and cost efficiency. The primary economic driver is the drastic reduction in raw material consumption achieved through the closed-loop recovery of methyl bromide. In traditional processes, the loss of this reagent represents a sunk cost that accumulates with every batch; here, the ability to recover and reuse over 90% of the input significantly lowers the recurring material expense. Furthermore, the substantial increase in yield from roughly 80% to nearly 99% means that less starting material is required to produce the same amount of finished API, effectively amplifying the throughput of existing reactor capacity without the need for capital-intensive expansion. This efficiency gain is compounded by the reduction in waste disposal costs, as the process generates significantly less hazardous liquid waste requiring specialized treatment.
- Cost Reduction in Manufacturing: The implementation of a methyl bromide recovery system fundamentally alters the cost structure of Ipratropium Bromide production. By capturing and recycling the excessive reagent rather than venting or neutralizing it, manufacturers eliminate a major variable cost component. Additionally, the higher conversion rate reduces the burden on downstream purification steps, as there is less unreacted intermediate to separate, leading to lower solvent usage and energy consumption during crystallization. These cumulative efficiencies result in a leaner manufacturing process that offers substantial cost savings in pharmaceutical intermediate manufacturing, making the final product more competitive in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of raw material markets and regulatory restrictions on hazardous chemicals. This process mitigates those risks by reducing the net consumption of methyl bromide per kilogram of product, thereby decreasing the frequency of hazardous material shipments and storage requirements. The robustness of the reaction conditions, which tolerate slight variations while maintaining high purity, ensures consistent batch-to-batch quality. This reliability is crucial for reliable API intermediate supplier relationships, as it minimizes the risk of production delays caused by failed batches or out-of-specification results, ensuring a steady flow of material to downstream formulation sites.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, particularly concerning ozone-depleting substances, the ability to demonstrate a green manufacturing process is a significant competitive advantage. This method transforms a potentially polluting reaction into a sustainable operation by preventing the release of methyl bromide into the atmosphere. The mild reaction conditions and the use of common solvents like chloroform facilitate easy scale-up from pilot plants to multi-ton commercial production. The simplified work-up procedure, involving straightforward filtration and washing, reduces the complexity of the plant operations, allowing for faster turnaround times and reducing lead time for high-purity pharmaceutical intermediates, which is essential for meeting urgent market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route. Understanding these nuances is essential for assessing the fit of this process within your existing manufacturing infrastructure and quality assurance frameworks.
Q: How does the staged addition of methyl bromide improve yield compared to conventional methods?
A: Conventional methods often add methyl bromide in a single portion, leading to incomplete conversion and yields around 80%. The patented staged addition (2-4 times) combined with a temperature gradient ensures thorough reaction kinetics, pushing yields above 95%.
Q: Is the methyl bromide recovery process effective for large-scale production?
A: Yes, the process includes a dedicated distillation step at 40-50°C that allows for the recovery of over 92% of the excessive methyl bromide. This recovered reagent can be reused in subsequent batches without affecting product quality, significantly reducing raw material costs.
Q: What purity levels can be achieved with this synthesis method?
A: By optimizing the stirring time and temperature profiles, specifically holding at -5°C to 0°C for 15-17 hours, the method minimizes side reactions. This results in a final product purity exceeding 99.9%, meeting strict European Pharmacopoeia standards for related substances.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ipratropium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis methods requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of Ipratropium Bromide meets the highest international pharmacopoeial standards. Our commitment to quality extends beyond mere compliance; we actively optimize every unit operation to maximize yield and minimize environmental impact, aligning our manufacturing philosophy with the sustainability goals of our global partners.
We invite pharmaceutical companies and contract manufacturers to explore how this optimized synthesis route can enhance their supply chain efficiency and product quality. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can secure your supply of high-quality respiratory API intermediates while driving down your overall production costs.
