Scalable Biocatalytic Production of High-Purity (S)-1,2,3,4-Tetrahydroisoquinoline-1-Carboxylic Acid Intermediates
Introduction to Advanced Biocatalytic Chiral Resolution
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant innovations in biocatalysis, as exemplified by the groundbreaking technology disclosed in Chinese Patent CN110835639B. This patent details a sophisticated method for preparing (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid and its derivatives, which serve as critical building blocks for developing potent Keap1-Nrf2 protein-protein interaction inhibitors used in treating cancer and neurodegenerative diseases. Unlike traditional synthetic routes that rely on harsh chemical reagents and offer limited stereocontrol, this novel approach leverages a multi-enzyme coupling system to achieve dynamic kinetic resolution with exceptional efficiency. The technology represents a paradigm shift in how complex heterocyclic amino acids are manufactured, offering a pathway to >99% enantiomeric excess while maintaining mild reaction parameters that are conducive to large-scale industrial application. For global procurement teams and R&D directors, understanding the mechanistic advantages of this biocatalytic route is essential for securing a reliable supply chain of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid has been plagued by significant technical and economic hurdles inherent to chemical chiral synthesis. Traditional methods often necessitate the use of expensive chiral starting materials or involve multi-step sequences utilizing hazardous reagents such as ozone and sodium borohydride, followed by oxidation with TEMPO and acidic deprotection. These chemical processes are not only operationally complex but also suffer from low overall yields and poor atom economy, generating substantial waste streams that complicate environmental compliance. Furthermore, achieving high optical purity through chemical means frequently requires repeated recrystallization or chromatographic separation, which drastically increases production costs and extends lead times. The reliance on transition metal catalysts in some prior art methods also introduces the risk of heavy metal contamination, necessitating rigorous and costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients. Consequently, these limitations render conventional chemical synthesis less viable for the cost-effective, large-scale manufacturing required by the modern pharmaceutical market.
The Novel Approach
In stark contrast to the deficiencies of chemical synthesis, the biocatalytic method disclosed in the patent utilizes a highly specific enzymatic cascade to convert racemic substrates directly into the desired S-enantiomer with theoretical 100% yield potential. This innovative strategy employs a D-amino acid oxidase to selectively oxidize the unwanted R-isomer into an imino acid intermediate, while leaving the target S-isomer untouched in the reaction mixture. Crucially, the process integrates a pipecolic acid reductase that subsequently reduces the imino acid back into the S-configuration, effectively recycling the R-isomer and driving the equilibrium towards the product. 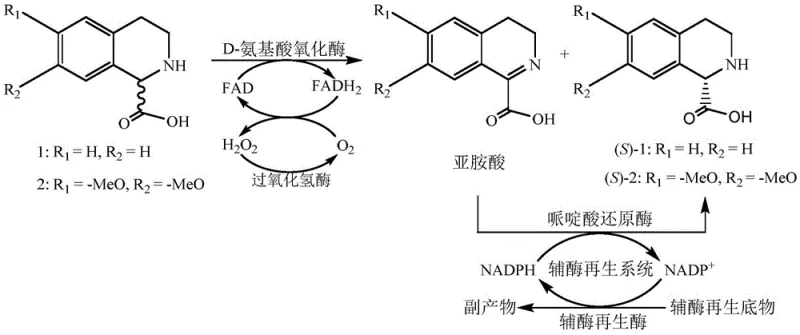 As illustrated in the reaction mechanism, the inclusion of catalase is a vital safety feature that decomposes the hydrogen peroxide byproduct into water and oxygen, preventing oxidative damage to the enzymes and eliminating the need for hazardous peroxide quenching agents. This tandem enzymatic system operates under mild physiological conditions, typically between 30°C and 40°C at a neutral pH, thereby significantly reducing energy consumption and equipment corrosion risks compared to the extreme conditions of chemical synthesis.
As illustrated in the reaction mechanism, the inclusion of catalase is a vital safety feature that decomposes the hydrogen peroxide byproduct into water and oxygen, preventing oxidative damage to the enzymes and eliminating the need for hazardous peroxide quenching agents. This tandem enzymatic system operates under mild physiological conditions, typically between 30°C and 40°C at a neutral pH, thereby significantly reducing energy consumption and equipment corrosion risks compared to the extreme conditions of chemical synthesis.
Mechanistic Insights into Multi-Enzyme Coupled Dynamic Kinetic Resolution
The core of this technological breakthrough lies in the precise orchestration of a multi-enzyme system that facilitates a dynamic kinetic resolution (DKR) of the racemic tetrahydroisoquinoline carboxylic acid. The process initiates with the D-amino acid oxidase (DAAO), derived from sources such as Fusarium solani or Trigonopsis variabilis, which exhibits strict stereoselectivity for the R-configured substrate. Upon binding, the DAAO catalyzes the oxidative dehydrogenation of the R-isomer, utilizing Flavin Adenine Dinucleotide (FAD) as a cofactor to generate the corresponding imino acid and reduced FADH2. 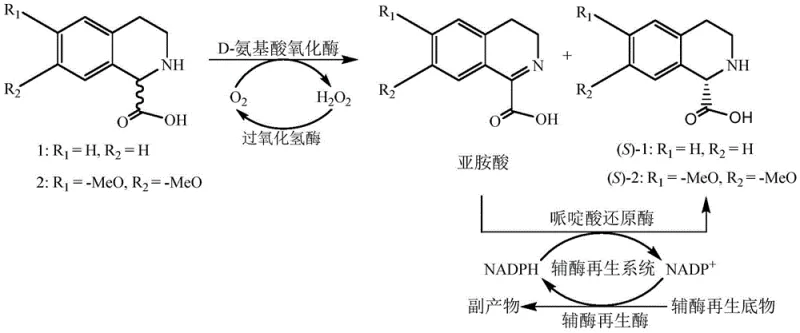 The reduced flavin is then re-oxidized by molecular oxygen, producing hydrogen peroxide, which is immediately scavenged by the added catalase to maintain enzyme stability. Following this oxidation step, the transient imino acid intermediate is subjected to asymmetric reduction by a pipecolic acid reductase, such as those derived from Pseudomonas putida. This reductase utilizes NADPH as a hydride donor to stereospecifically reduce the imine double bond, regenerating the tetrahydroisoquinoline ring structure exclusively in the S-configuration. To ensure economic viability, the system incorporates a coenzyme regeneration module, typically involving glucose dehydrogenase or alcohol dehydrogenase, which recycles the oxidized NADP+ back to NADPH using inexpensive substrates like glucose or isopropanol. This closed-loop cofactor system minimizes the requirement for expensive nicotinamide cofactors, making the process economically feasible for industrial scale-up.
The reduced flavin is then re-oxidized by molecular oxygen, producing hydrogen peroxide, which is immediately scavenged by the added catalase to maintain enzyme stability. Following this oxidation step, the transient imino acid intermediate is subjected to asymmetric reduction by a pipecolic acid reductase, such as those derived from Pseudomonas putida. This reductase utilizes NADPH as a hydride donor to stereospecifically reduce the imine double bond, regenerating the tetrahydroisoquinoline ring structure exclusively in the S-configuration. To ensure economic viability, the system incorporates a coenzyme regeneration module, typically involving glucose dehydrogenase or alcohol dehydrogenase, which recycles the oxidized NADP+ back to NADPH using inexpensive substrates like glucose or isopropanol. This closed-loop cofactor system minimizes the requirement for expensive nicotinamide cofactors, making the process economically feasible for industrial scale-up.
From an impurity control perspective, this biocatalytic route offers superior selectivity that inherently minimizes the formation of side products common in chemical synthesis. The high specificity of the enzymes ensures that functional groups on the aromatic ring, such as the methoxy groups in 6,7-dimethoxy derivatives, remain intact without undergoing unwanted oxidation or substitution reactions. The patent data indicates that the ee value of the resulting S-isomer consistently exceeds 99%, effectively eliminating the need for complex chiral separation techniques post-reaction. Furthermore, the use of engineered E. coli strains expressing the recombinant enzymes allows for the production of the biocatalysts via fermentation, ensuring batch-to-batch consistency and scalability. The reaction conditions are carefully optimized to balance enzyme activity and stability, with pH levels maintained between 7 and 8.5 using phosphate buffers to prevent enzyme denaturation. This robust control over the reaction environment results in a clean product profile, simplifying downstream processing and reducing the burden on quality control laboratories to detect and quantify trace impurities.
How to Synthesize (S)-1,2,3,4-Tetrahydroisoquinoline-1-Carboxylic Acid Efficiently
The implementation of this biocatalytic synthesis route requires a systematic approach to enzyme preparation and reaction engineering to maximize yield and productivity. The process begins with the construction of genetically engineered bacterial strains, specifically E. coli BL21(DE3), transformed with expression vectors containing the genes for D-amino acid oxidase, pipecolic acid reductase, and the coenzyme-regenerating enzyme. These strains are cultivated in liquid media supplemented with appropriate antibiotics and induced with IPTG to express the recombinant proteins, which are then harvested and lysed to obtain crude enzyme solutions or used directly as resting cells.
- Construct a reaction system containing racemic substrate, D-amino acid oxidase, pipecolic acid reductase, catalase, and a coenzyme regeneration system.
- Maintain the reaction at 30-40°C and pH 7-8.5 under aerobic conditions to facilitate oxidative dehydrogenation of the R-isomer.
- Allow the pipecolic acid reductase to asymmetrically reduce the generated imino acid intermediate into the target S-isomer with >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology translates into tangible strategic advantages regarding cost structure and supply reliability. The elimination of expensive chiral resolving agents and transition metal catalysts fundamentally alters the cost basis of production, removing the volatility associated with precious metal markets and specialized chiral reagents. By utilizing renewable biological catalysts produced via fermentation, manufacturers can achieve substantial cost savings in raw material procurement while simultaneously reducing the environmental footprint of the manufacturing process. The mild reaction conditions further contribute to cost reduction in pharmaceutical intermediate manufacturing by lowering energy requirements for heating and cooling, as well as reducing maintenance costs associated with corrosion-resistant reactor vessels. Additionally, the high conversion rates and excellent stereoselectivity minimize material loss, ensuring that the maximum amount of starting substrate is converted into saleable product, thereby improving overall process mass intensity.
- Cost Reduction in Manufacturing: The biocatalytic process eliminates the need for hazardous chemical oxidants and complex protection-deprotection sequences, leading to a drastically simplified workflow that reduces labor and utility costs. By avoiding the use of heavy metal catalysts, the downstream purification process is streamlined, removing the expensive and time-consuming steps required for metal scavenging and residual metal testing. The integration of an in-situ coenzyme regeneration system ensures that costly cofactors like NADPH are used in catalytic rather than stoichiometric amounts, significantly lowering the variable cost per kilogram of product. Furthermore, the high yield and selectivity reduce the volume of waste solvent and byproducts that require disposal, resulting in lower waste treatment fees and improved compliance with environmental regulations.
- Enhanced Supply Chain Reliability: Reliance on fermentation-derived enzymes provides a more stable and scalable supply of catalysts compared to the extraction of natural products or the multi-step synthesis of chemical ligands. The ability to produce the necessary biocatalysts in-house or through established fermentation partners mitigates the risk of supply disruptions caused by geopolitical issues or raw material shortages affecting chemical reagents. The robustness of the engineered enzymes allows for consistent performance across large batches, ensuring predictable production schedules and reliable delivery timelines for downstream API manufacturers. This stability is crucial for long-term supply agreements, as it guarantees that the quality and quantity of the chiral intermediate can be maintained even as demand scales up for clinical or commercial phases.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction system aligns perfectly with green chemistry principles, significantly reducing the consumption of organic solvents typically required in chemical synthesis. This shift not only lowers the fire and explosion hazards associated with volatile organic compounds but also simplifies the regulatory approval process for new drug applications by presenting a cleaner manufacturing profile. The process is inherently scalable from liter-scale laboratory flasks to cubic-meter industrial fermenters without significant re-optimization, facilitating rapid technology transfer from R&D to commercial production. Moreover, the generation of benign byproducts such as water and oxygen, rather than toxic heavy metal sludge or halogenated waste, ensures that the manufacturing facility can meet increasingly stringent global environmental standards with minimal capital investment in end-of-pipe treatment technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis platform. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's feasibility.
Q: What is the stereoselectivity of the biocatalytic method compared to chemical synthesis?
A: The biocatalytic method described in patent CN110835639B achieves an enantiomeric excess (ee) value of greater than 99%, significantly outperforming traditional chemical methods which often struggle to exceed 90% ee without complex purification steps.
Q: How does the process handle the hazardous byproducts of oxidation?
A: The process incorporates catalase into the multi-enzyme system, which efficiently decomposes hydrogen peroxide generated during the D-amino acid oxidase catalyzed step into water and oxygen, ensuring operational safety and preventing enzyme inactivation.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the method utilizes mild reaction conditions (30-40°C, neutral pH) and engineered E. coli strains expressing the necessary enzymes, making it highly suitable for large-scale fermentation and downstream processing in commercial pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1,2,3,4-Tetrahydroisoquinoline-1-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic routes described in patent CN110835639B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the >99% ee values and low impurity profiles guaranteed by this advanced enzymatic methodology. We understand that transitioning to a biocatalytic process requires a partner with deep technical expertise in enzyme engineering and fermentation optimization, capabilities that are central to our service offering.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching from chemical to enzymatic synthesis for your specific project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you secure a sustainable, cost-effective, and high-quality supply of (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid derivatives to accelerate your path to market.
