Advanced Biocatalytic Production of Chiral Tetrahydroisoquinoline Intermediates for Scalable Pharmaceutical Manufacturing
Introduction to High-Efficiency Biocatalytic Synthesis
The pharmaceutical industry continuously seeks robust methodologies for generating optically pure intermediates, particularly for complex heterocyclic scaffolds like tetrahydroisoquinolines which serve as critical building blocks for oncology and neurodegenerative therapeutics. Patent CN110317849B discloses a groundbreaking method for preparing (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid and its derivatives, utilizing a sophisticated enzymatic dynamic kinetic resolution (DKR) strategy. This technology leverages the stereoselectivity of D-amino acid oxidase (D-AAO) to distinguish between enantiomers, coupled with a chemical reduction step that recycles the undesired isomer. Unlike traditional methods that discard half of the starting material, this approach theoretically allows for 100% conversion of the racemate into the desired (S)-enantiomer. The disclosed process operates under mild aqueous conditions, typically between 30°C and 50°C, and achieves exceptional optical purity with ee values consistently greater than 99%. For global procurement teams and R&D directors, this represents a paradigm shift from resource-intensive chemical synthesis to sustainable, high-efficiency biocatalysis, offering a reliable pathway for securing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active tetrahydroisoquinoline-1-carboxylic acids has relied heavily on classical chemical resolution or asymmetric synthesis, both of which present significant bottlenecks for industrial scale-up. Classical resolution techniques, such as diastereomeric salt formation, are inherently limited by a maximum theoretical yield of 50%, necessitating the disposal or costly recycling of the unwanted (R)-enantiomer. Furthermore, these processes often require multiple recrystallization steps to achieve acceptable enantiomeric excess, leading to substantial solvent consumption and extended production cycles. Asymmetric chemical synthesis, while capable of higher yields, frequently involves expensive chiral ligands, transition metal catalysts, and stringent anhydrous conditions that complicate safety protocols and waste management. The presence of heavy metal residues also imposes rigorous purification requirements to meet regulatory standards for active pharmaceutical ingredients (APIs). These factors collectively drive up the cost of goods sold (COGS) and introduce supply chain vulnerabilities related to raw material availability and environmental compliance.
The Novel Approach
The methodology outlined in CN110317849B overcomes these barriers by integrating biocatalysis with chemical reduction in a one-pot dynamic kinetic resolution system. By employing D-amino acid oxidase, the process selectively oxidizes the (R)-isomer of the racemic substrate into an imino acid intermediate, while the desired (S)-isomer remains untouched in the reaction mixture. Crucially, the addition of a chemical reducing agent, such as sodium cyanoborohydride or sodium borohydride, instantly reduces the unstable imino acid back into the racemic amine form. This creates a cyclic equilibrium where the (R)-isomer is continuously consumed and regenerated until the entire substrate pool is converted into the non-reactive (S)-enantiomer. 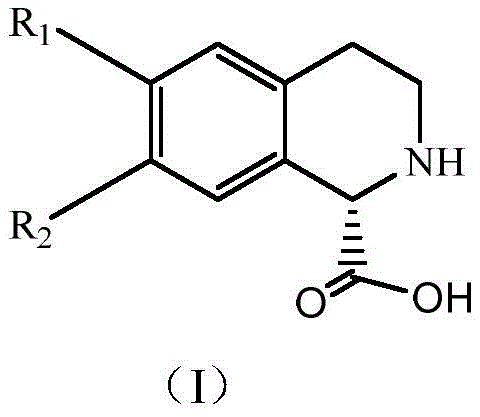 This mechanism effectively bypasses the 50% yield ceiling, enabling experimental yields ranging from 75% to 97.4% as demonstrated in the patent examples. The reaction proceeds in an aqueous phosphate buffer at neutral pH, eliminating the need for hazardous organic solvents and simplifying downstream processing through simple protein precipitation and crystallization.
This mechanism effectively bypasses the 50% yield ceiling, enabling experimental yields ranging from 75% to 97.4% as demonstrated in the patent examples. The reaction proceeds in an aqueous phosphate buffer at neutral pH, eliminating the need for hazardous organic solvents and simplifying downstream processing through simple protein precipitation and crystallization.
Mechanistic Insights into D-AAO Catalyzed Dynamic Kinetic Resolution
The core of this technology lies in the precise stereochemical recognition capabilities of D-amino acid oxidase enzymes derived from sources such as Fusarium solani or Trigonopsis variabilis. These enzymes utilize flavin adenine dinucleotide (FAD) as a cofactor to facilitate the oxidative dehydrogenation of the amino acid substrate. During the catalytic cycle, the enzyme binds specifically to the (R)-configuration of the tetrahydroisoquinoline ring, abstracting a hydride to form the corresponding imino acid while reducing FAD to FADH2. Molecular oxygen then re-oxidizes FADH2 back to FAD, generating hydrogen peroxide as a byproduct. To maintain enzyme stability and prevent oxidative degradation of the product, the system incorporates catalase, which rapidly decomposes hydrogen peroxide into water and oxygen. 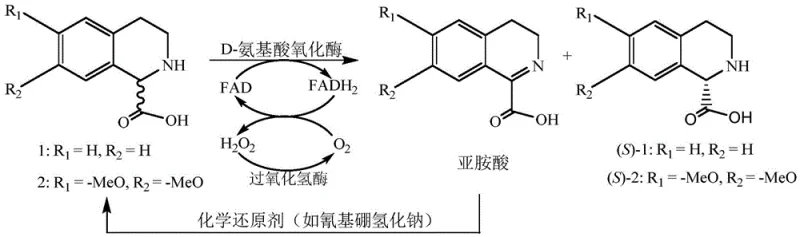 The imino acid intermediate, being highly reactive, is immediately intercepted by the added chemical reducing agent. This reduction step is non-stereoselective, regenerating a racemic mixture of the starting amine. However, because the enzyme continuously removes the (R)-isomer from this regenerated pool, the equilibrium shifts entirely towards the accumulation of the (S)-isomer. This synergistic interplay between enzymatic oxidation and chemical reduction ensures that impurities arising from incomplete resolution are minimized, resulting in a final product with an enantiomeric excess exceeding 99%, which is critical for meeting the stringent purity specifications required for clinical-grade intermediates.
The imino acid intermediate, being highly reactive, is immediately intercepted by the added chemical reducing agent. This reduction step is non-stereoselective, regenerating a racemic mixture of the starting amine. However, because the enzyme continuously removes the (R)-isomer from this regenerated pool, the equilibrium shifts entirely towards the accumulation of the (S)-isomer. This synergistic interplay between enzymatic oxidation and chemical reduction ensures that impurities arising from incomplete resolution are minimized, resulting in a final product with an enantiomeric excess exceeding 99%, which is critical for meeting the stringent purity specifications required for clinical-grade intermediates.
How to Synthesize (S)-1,2,3,4-Tetrahydroisoquinoline-1-carboxylic Acid Efficiently
Implementing this biocatalytic route requires careful optimization of the reaction parameters to balance enzyme activity with chemical reduction rates. The process begins with the preparation of a substrate solution containing the racemic tetrahydroisoquinoline derivative dissolved in a phosphate buffer adjusted to a pH between 7 and 8. To this solution, the biocatalyst—either as a crude enzyme lysate, resting cells of engineered E. coli, or purified enzyme—is added alongside catalase to manage oxidative stress. The reaction is initiated by the addition of the reducing agent, such as sodium cyanoborohydride, in stoichiometric excess relative to the substrate to ensure rapid turnover of the imino acid intermediate. Maintaining the temperature between 30°C and 50°C is essential to maximize enzymatic turnover without denaturing the protein. Detailed standardized synthetic steps follow below.
- Prepare the reaction system by dissolving racemic substrate in phosphate buffer (pH 6-9) and adding D-amino acid oxidase catalyst along with catalase.
- Introduce an imino acid reducing agent such as sodium cyanoborohydride to the mixture to facilitate the in-situ reduction of the oxidized imine intermediate back to the racemate.
- Maintain the reaction at 30-50°C under aerobic conditions for 20-48 hours, then adjust pH to precipitate proteins and isolate the high-purity (S)-enantiomer via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic DKR process offers transformative benefits regarding cost structure and operational reliability. The shift from multi-step chemical synthesis to a streamlined biocatalytic process drastically simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor and equipment costs. By eliminating the need for expensive chiral ligands and transition metal catalysts, the raw material costs are significantly lowered, and the risk of supply disruption due to scarce metal availability is mitigated. Furthermore, the aqueous nature of the reaction reduces the dependency on volatile organic compounds (VOCs), aligning production with increasingly strict environmental regulations and reducing waste disposal expenses. The high yield and selectivity of the process mean that less starting material is required to produce the same amount of API intermediate, directly improving the overall material efficiency and reducing the carbon footprint of the supply chain.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet residual metal limits in pharmaceutical products. Additionally, the ability to achieve near-quantitative yields from racemic starting materials, which are generally cheaper and more readily available than single-enantiomer precursors, leads to substantial cost savings in raw material procurement. The simplified workup procedure, involving protein denaturation and crystallization rather than complex chromatographic separations, further reduces processing time and solvent consumption, contributing to a lower overall cost of production.
- Enhanced Supply Chain Reliability: Biocatalytic processes are inherently more robust against fluctuations in raw material quality compared to sensitive chemical syntheses. The enzymes used, such as FsDAAO and TvDAAO, can be produced via fermentation at a large scale, ensuring a consistent and renewable supply of the catalyst. This biological manufacturing capability decouples the production of the intermediate from the volatility of petrochemical feedstocks. Moreover, the mild reaction conditions reduce the risk of thermal runaways or hazardous incidents, ensuring continuous operation and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is designed for scalability, having been demonstrated effectively from gram-scale screening to potential multi-ton production. The use of water as the primary solvent and the generation of benign byproducts like nitrogen, water, and carbon dioxide simplify effluent treatment and reduce the environmental impact. This green chemistry profile facilitates easier regulatory approval and permits for manufacturing facilities, accelerating the time-to-market for new drug candidates relying on these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented biocatalytic technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: What represents the primary advantage of using D-amino acid oxidase for this synthesis compared to chemical resolution?
A: The primary advantage is the ability to achieve yields significantly higher than the theoretical 50% limit of classical resolution. By coupling enzymatic oxidation with chemical reduction, the process dynamically converts the unwanted (R)-isomer back into the substrate pool, allowing for yields up to 97.4% with ee values exceeding 99%.
Q: Which specific enzymes are identified as effective catalysts in this patented method?
A: The patent identifies D-amino acid oxidases derived from Fusarium solani (FsDAAO), Fusarium graminearum (FgDAAO), Fusarium poae (FpDAAO), and Trigonopsis variabilis (TvDAAO) as highly effective catalysts for stereoselectively oxidizing the (R)-isomer while leaving the desired (S)-isomer intact.
Q: How does the process manage the hydrogen peroxide byproduct generated during oxidation?
A: The process incorporates catalase, specifically bovine liver catalase, into the reaction system. This enzyme rapidly decomposes the hydrogen peroxide byproduct into water and oxygen, preventing oxidative damage to the biocatalyst and ensuring reaction stability over extended periods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1,2,3,4-Tetrahydroisoquinoline-1-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of production. Our facility is equipped to handle complex biocatalytic processes, including the enzymatic dynamic kinetic resolution described in CN110317849B, providing our partners with a secure and compliant source of supply.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for tetrahydroisoquinoline derivatives. By leveraging our expertise in process development and scale-up, we can help you realize the full commercial potential of this technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your project timelines and regulatory filings.
