Advanced Biocatalytic Synthesis of High-Purity (S)-Tetrahydroisoquinoline Derivatives for Global Pharma Supply Chains
Introduction to Patent CN110835639A and Technological Breakthrough
The pharmaceutical industry constantly seeks robust methods for producing chiral intermediates with exceptional optical purity, and patent CN110835639A presents a significant advancement in this domain by disclosing a novel biocatalytic method for preparing (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid and its derivatives. This technology addresses the critical need for high-efficiency stereoselective synthesis, utilizing a sophisticated dual-enzyme system that combines oxidative dehydrogenase with pipecolic acid reductase to achieve dynamic kinetic resolution. Unlike traditional approaches that often struggle with limited conversion or moderate enantiomeric excess, this invention leverages the specific catalytic properties of D-amino acid oxidase sourced from organisms such as Fusarium solani or Trigonopsis variabilis to selectively transform the unwanted (R)-isomer while preserving the desired (S)-configuration. The integration of a cofactor regeneration system further enhances the economic viability of the process, making it a compelling solution for the scalable manufacturing of high-value pharmaceutical intermediates used in the treatment of conditions ranging from cancer to neurodegenerative diseases.
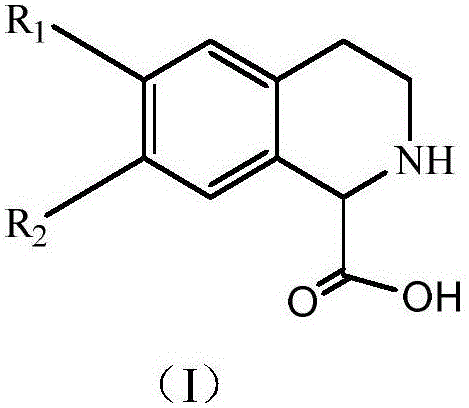
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid has relied heavily on chemical chiral synthesis or less efficient biocatalytic resolutions, both of which present substantial drawbacks for industrial application. Chemical routes, such as those described by Kurata et al., often involve multi-step sequences utilizing hazardous reagents like ozone for ozonolysis and strong oxidants like TEMPO, followed by acidic deprotection steps that generate significant waste and pose safety risks. These synthetic pathways frequently suffer from low overall yields and require rigorous purification to remove trace metal catalysts and organic impurities, which complicates the supply chain and increases the cost of goods. Furthermore, earlier biocatalytic attempts, such as the dynamic kinetic enzymatic hydrolysis reported by Paál et al., while milder, often resulted in products with enantiomeric excess values around 93%, which may not meet the stringent purity specifications required for modern active pharmaceutical ingredients without additional recrystallization steps that reduce overall throughput.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110835639A introduces a streamlined one-pot biocatalytic cascade that dramatically improves both efficiency and stereocontrol. By employing a specific D-amino acid oxidase to selectively oxidize the (R)-enantiomer of the racemic substrate into an imino acid intermediate, the process effectively channels the unwanted isomer into a pathway that ultimately yields the desired product. This intermediate is then immediately reduced by pipecolic acid reductase in the presence of a hydride-donating coenzyme, ensuring that the theoretical yield can approach 100% rather than being capped at 50% as in classical kinetic resolutions. The reaction conditions are notably mild, typically operating between 30°C and 40°C at a neutral to slightly alkaline pH, which minimizes energy consumption and equipment corrosion. This novel approach not only simplifies the downstream processing by eliminating the need for complex chromatographic separations but also ensures a consistent supply of high-purity material suitable for direct use in sensitive drug synthesis applications.
Mechanistic Insights into Dual-Enzyme Dynamic Kinetic Resolution
The core of this technological innovation lies in the precise orchestration of two distinct enzymatic activities working in tandem to drive the reaction equilibrium towards the formation of the (S)-enantiomer. The process initiates with the D-amino acid oxidase, which exhibits strict stereoselectivity for the (R)-isomer of the tetrahydroisoquinoline substrate, catalyzing its oxidative dehydrogenation to form an imino acid derivative while generating hydrogen peroxide as a byproduct. To prevent enzyme inactivation by the accumulated peroxide, catalase is included in the system to decompose it into water and oxygen, thereby maintaining a stable reaction environment. Subsequently, the generated imino acid serves as the substrate for the pipecolic acid reductase, which utilizes reduced nicotinamide adenine dinucleotide phosphate (NADPH) to asymmetrically reduce the imine bond, locking the stereochemistry into the (S)-configuration with exceptional fidelity. This coupling ensures that as the (R)-isomer is consumed, it is continuously converted into the (S)-product, effectively overcoming the thermodynamic limitations of simple resolution.
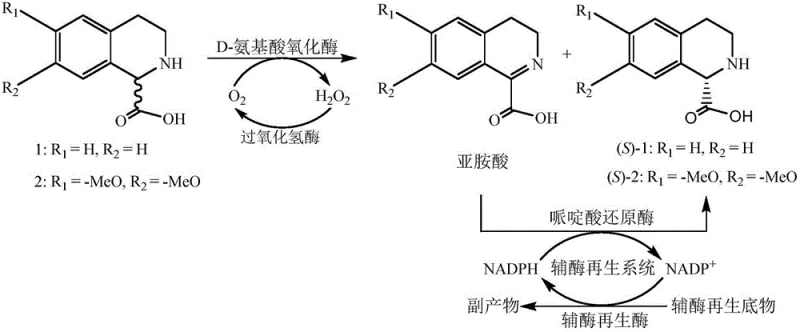
Furthermore, the sustainability of this mechanism is bolstered by an integrated cofactor regeneration system that prevents the stoichiometric consumption of expensive NADPH. By incorporating enzymes such as glucose dehydrogenase or alcohol dehydrogenase along with their respective substrates like glucose or isopropanol, the oxidized cofactor (NADP+) produced during the reduction step is rapidly recycled back to its active reduced form. This closed-loop system allows for the use of catalytic amounts of the cofactor, significantly reducing the raw material costs associated with the biocatalytic process. The impurity profile is also tightly controlled, as the high specificity of the enzymes minimizes the formation of side products commonly seen in chemical synthesis, such as over-oxidized species or regio-isomers. The result is a reaction mixture where the primary component is the target (S)-isomer with an ee value consistently exceeding 99%, simplifying the final isolation steps to basic pH adjustment and crystallization.
How to Synthesize (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid Efficiently
Implementing this biocatalytic route requires careful optimization of the multi-enzyme system to ensure maximum activity and stability throughout the reaction duration. The process begins with the preparation of a reaction buffer, typically phosphate-based, adjusted to a pH range of 7.0 to 8.5, which is optimal for the concurrent activity of both the oxidase and the reductase. The racemic substrate is introduced at concentrations ranging from 1 g/L to 20 g/L, depending on the specific solubility and enzyme tolerance, alongside the requisite enzyme preparations which can be used as crude lysates, purified enzymes, or immobilized forms for enhanced reusability. Critical to the success of the operation is the maintenance of aerobic conditions to support the oxidase activity, coupled with the continuous monitoring of pH to counteract any acidification caused by the reaction byproducts. Detailed standardized synthesis steps see the guide below.
- Construct a reaction system containing racemic substrate, D-amino acid oxidase, pipecolic acid reductase, catalase, and a cofactor regeneration system (e.g., glucose dehydrogenase).
- Maintain the reaction at 30-40°C and pH 7-8.5 to facilitate the oxidative dehydrogenation of the (R)-isomer and subsequent reduction to the (S)-isomer.
- Isolate the product by adjusting pH to 5.0-6.0, removing proteins via heat denaturation, and crystallizing the final high-purity (S)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers transformative benefits that extend beyond mere technical performance, directly impacting the bottom line through operational efficiencies. The elimination of harsh chemical reagents and transition metals from the synthesis route removes the necessity for expensive and time-consuming heavy metal scavenging processes, which are often regulatory bottlenecks in API manufacturing. This simplification of the purification train leads to a drastic reduction in solvent usage and waste disposal costs, aligning perfectly with green chemistry initiatives and environmental compliance standards that are increasingly scrutinized by global regulators. Moreover, the mild reaction conditions reduce the energy load on manufacturing facilities, as there is no need for extreme heating or cooling, thereby lowering the utility costs associated with production.
- Cost Reduction in Manufacturing: The implementation of this enzymatic cascade significantly lowers the cost of goods sold by maximizing atom economy and minimizing raw material waste. Since the dynamic kinetic resolution theoretically converts 100% of the starting racemate into the desired product, there is no loss of valuable material to the unwanted enantiomer, which is a common inefficiency in traditional resolution methods. Additionally, the use of whole-cell biocatalysts or crude enzyme preparations avoids the high capital expenditure associated with purifying enzymes to homogeneity, further driving down the input costs for the manufacturing process while maintaining high product quality.
- Enhanced Supply Chain Reliability: Relying on biocatalytic processes enhances supply chain resilience by reducing dependence on volatile petrochemical feedstocks and specialized chemical reagents that may be subject to geopolitical supply disruptions. The enzymes used in this process can be produced via fermentation using renewable carbon sources, creating a more sustainable and predictable supply base. The robustness of the engineered strains, such as E. coli BL21 expressing the specific oxidase and reductase genes, ensures consistent batch-to-batch performance, reducing the risk of production delays caused by failed chemical runs or inconsistent raw material quality.
- Scalability and Environmental Compliance: This technology is inherently scalable, having been demonstrated to function effectively across a wide range of substrate concentrations without significant loss in efficiency, facilitating a smooth transition from laboratory benchtop to commercial-scale reactors. The aqueous nature of the reaction medium and the biodegradability of the biological components simplify wastewater treatment protocols, ensuring that the manufacturing facility remains compliant with strict environmental discharge regulations. This ease of scale-up and regulatory alignment makes the process an ideal candidate for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the operational parameters and performance metrics of this biocatalytic system. The following insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development planning. Understanding these details is crucial for integrating this method into existing manufacturing workflows and assessing its compatibility with current infrastructure.
Q: What is the primary advantage of this biocatalytic method over chemical synthesis?
A: The biocatalytic method described in patent CN110835639A achieves significantly higher enantiomeric excess (ee > 99%) under mild conditions, avoiding the harsh reagents and complex purification steps associated with traditional chemical chiral synthesis.
Q: Which enzymes are critical for the stereoselectivity in this process?
A: The process relies on the synergistic action of D-amino acid oxidase (DAAO) for selective oxidation of the (R)-isomer and pipecolic acid reductase for the asymmetric reduction of the resulting imino acid intermediate to the desired (S)-configuration.
Q: How is the cofactor cost managed in this large-scale enzymatic reaction?
A: The protocol incorporates an efficient cofactor regeneration system, typically using glucose dehydrogenase or alcohol dehydrogenase, which continuously recycles NADP+/NADPH, thereby minimizing the requirement for expensive exogenous cofactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a reliable supply of high-quality chiral intermediates for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high enantiomeric excess values achievable through the biocatalytic routes described in patent CN110835639A. We are committed to delivering solutions that not only meet but exceed the demanding requirements of the global pharmaceutical market.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this enzymatic process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, cost-effective, and high-purity pharmaceutical intermediates.
