Advanced Valsartan Manufacturing: Optimized Synthesis for High-Purity API Production
Advanced Valsartan Manufacturing: Optimized Synthesis for High-Purity API Production
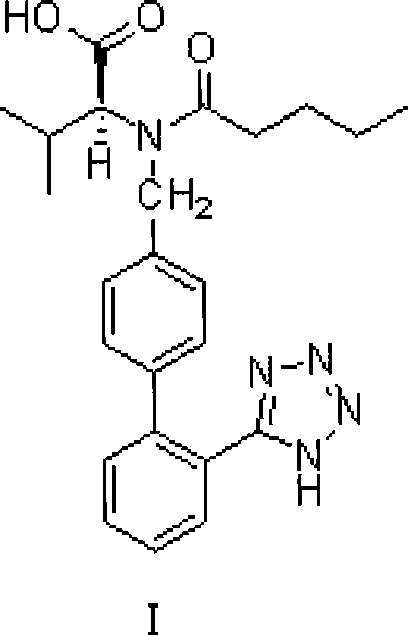
The pharmaceutical industry continuously demands more efficient, safer, and cost-effective routes for the production of essential antihypertensive agents like Valsartan. Patent CN101362728A presents a significant technological advancement in the synthetic methodology of this Angiotensin II receptor antagonist. This innovation specifically targets the optimization of the N-[(2'-cyanobiphenyl-4-yl)methyl]-(L)-valine methyl ester intermediate synthesis and refines the subsequent acylation and tetrazole cyclization steps. By addressing critical bottlenecks such as reaction time, catalyst loading, and hazardous waste management, this method offers a robust framework for commercial-scale manufacturing. The following analysis details how these technical improvements translate into tangible benefits for R&D directors, procurement managers, and supply chain heads seeking reliable API sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Valsartan, as described in earlier patents like US5399578, relied heavily on dimethylformamide (DMF) as the primary solvent for the alkylation of L-valine methyl ester. While effective, DMF presents significant challenges in large-scale operations due to its high boiling point, which complicates solvent recovery and increases energy consumption during distillation. Furthermore, conventional protocols often required excessive amounts of catalysts, such as 4-dimethylaminopyridine (DMAP), ranging from 0.5% to 2%, which not only inflates raw material costs but also necessitates rigorous purification steps to remove residual amine impurities from the final active pharmaceutical ingredient. Additionally, the tetrazole ring formation traditionally suffered from prolonged reaction times, often extending up to 48 hours, which severely limits reactor throughput and overall plant capacity.
The Novel Approach
The methodology disclosed in CN101362728A fundamentally re-engineers these process parameters to enhance efficiency and safety. A pivotal improvement is the substitution of DMF with acetonitrile for the initial alkylation step, conducted at a controlled temperature of 50 to 55°C. This switch facilitates easier solvent removal and recycling, directly impacting the environmental footprint and operational expenditure. Moreover, the novel approach drastically reduces the catalytic loading of DMAP to a range of 0.1% to 0.5% without compromising reaction kinetics, demonstrating a superior understanding of the acylation mechanism. The optimization extends to the cyclization phase, where the reaction time is compressed to approximately 30 hours through precise control of reagent stoichiometry and solvent selection, thereby maximizing asset utilization and reducing the overall manufacturing cycle time significantly.
Mechanistic Insights into Tetrazole Cyclization and Hydrolysis
The formation of the tetrazole ring is the most chemically intricate and safety-critical step in the Valsartan synthesis pathway. In this optimized process, the nitrile group of the biphenyl intermediate undergoes a [2+3] cycloaddition with an azide source. The patent specifies the use of tributyltin chloride and sodium azide in xylene, generating tributyltin azide in situ. This organometallic mediator facilitates the attack of the azide on the nitrile carbon, leading to the formation of the tetrazole ring coordinated with the tin species. The use of xylene as a high-boiling solvent allows the reaction to proceed under reflux conditions, providing the necessary thermal energy to overcome the activation barrier while maintaining a homogeneous reaction mixture. This mechanistic pathway ensures high conversion rates, with HPLC analysis indicating that the starting material content drops below 0.5%, signifying near-complete transformation.
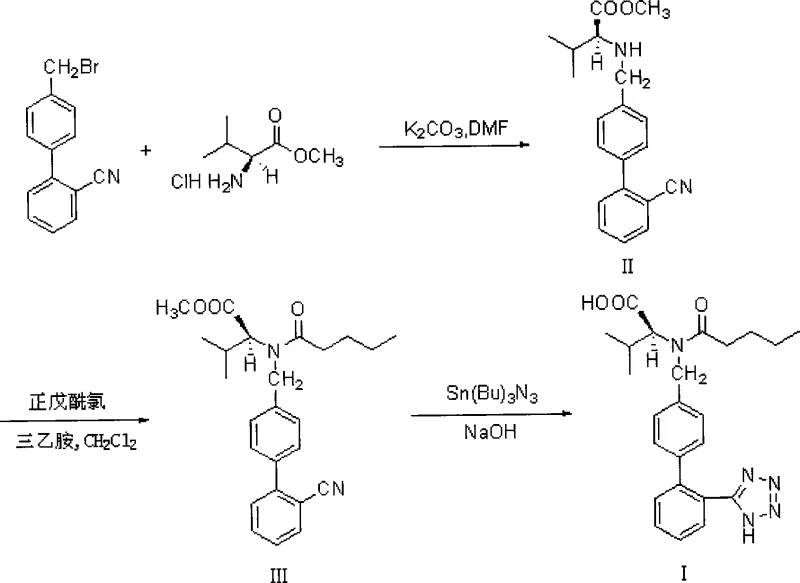
Beyond the cyclization mechanism, the post-reaction workup described in this patent introduces a vital safety intervention that is often overlooked in standard literature. The hydrolysis step utilizes potassium hydroxide to cleave the methyl ester, yielding the potassium salt of Valsartan. However, the presence of excess sodium azide poses a severe explosion hazard upon acidification, as it can generate volatile and toxic hydrazoic acid. To mitigate this, the process mandates the addition of sodium nitrite prior to pH adjustment. The nitrite ion reacts rapidly with the residual azide ions to form nitrogen gas and other harmless species, effectively neutralizing the explosive potential. This chemical quenching mechanism ensures that the subsequent acidification to pH 2-2.5 using hydrochloric acid can be performed safely, protecting both personnel and equipment while ensuring the final product is free from hazardous azide contaminants.
How to Synthesize Valsartan Efficiently
The synthesis of Valsartan via this optimized route requires precise adherence to reaction conditions to maximize yield and purity. The process begins with the alkylation of L-valine methyl ester hydrochloride using 4-bromomethyl-2'-cyanobiphenyl in an acetonitrile-potassium carbonate system. Following isolation, the intermediate undergoes N-acylation with valeryl chloride using minimal DMAP catalysis. The resulting ester is then subjected to tetrazole formation using tributyltin chloride and sodium azide in xylene, followed by hydrolysis and a specialized safety workup. For a detailed breakdown of the specific molar ratios, temperature profiles, and isolation techniques required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- React 4-bromomethyl-2'-cyanobiphenyl with L-valine methyl ester hydrochloride in acetonitrile with potassium carbonate at 50-55°C to form the alkylated intermediate.
- Perform N-acylation using valeryl chloride and reduced DMAP catalyst (0.1-0.5%) in the presence of solid carbonate acid binders.
- Execute tetrazole ring formation via reflux with tributyltin chloride and sodium azide in xylene for approximately 30 hours.
- Conduct hydrolysis using potassium hydroxide followed by a critical safety workup involving sodium nitrite to quench excess azides before acidification.
- Purify the crude product through a secondary crystallization process using ethyl acetate to achieve HPLC purity greater than 99.7%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical refinements in this patent translate directly into enhanced supply security and cost competitiveness. The shift away from problematic solvents like DMF towards more manageable systems like acetonitrile and xylene simplifies the logistics of raw material sourcing and waste disposal. By reducing the dependency on high volumes of difficult-to-remove solvents, the manufacturing process becomes more agile, allowing for faster batch turnover and reduced inventory holding times. Furthermore, the implementation of a robust secondary crystallization protocol ensures that the final product consistently meets high-purity specifications, minimizing the risk of batch rejection and the associated financial losses. These process stabilizations are critical for maintaining a continuous supply of high-quality API to downstream formulation partners.
- Cost Reduction in Manufacturing: The strategic reduction of catalyst usage, specifically lowering DMAP loading from typical industry levels to a fraction of that amount, results in substantial raw material savings over large production campaigns. Additionally, the shortening of the tetrazole cyclization reaction time from nearly two days to roughly 30 hours significantly decreases energy consumption related to heating and stirring. This acceleration of the reaction kinetics allows manufacturing facilities to increase their annual production capacity without requiring capital investment in new reactors, effectively lowering the fixed cost per kilogram of the produced API.
- Enhanced Supply Chain Reliability: The adoption of safer workup procedures, particularly the nitrite quenching of azides, reduces the likelihood of safety incidents that could lead to unplanned plant shutdowns. A safer process is inherently a more reliable one, ensuring that production schedules are met without interruption. Moreover, the use of common, commercially available solvents and reagents mitigates the risk of supply disruptions associated with specialty chemicals, providing a more resilient supply chain capable of withstanding market fluctuations and ensuring consistent delivery to global clients.
- Scalability and Environmental Compliance: The optimized process is designed with scalability in mind, utilizing standard unit operations such as filtration, extraction, and crystallization that are easily transferred from pilot to commercial scale. The improved purity profile achieved through secondary crystallization reduces the burden on downstream purification steps and minimizes the generation of complex waste streams. This alignment with green chemistry principles not only aids in meeting stringent environmental regulations but also lowers the costs associated with waste treatment and disposal, contributing to a more sustainable and economically viable manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Valsartan. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the specific advantages of this methodology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production or for procurement specialists assessing the quality implications of the manufacturing process.
Q: How does the new synthesis method improve safety regarding azide handling?
A: The patent introduces a specific post-treatment protocol where sodium nitrite is added to the reaction mixture prior to acidification. This step chemically quenches any unreacted sodium azide, preventing the formation of explosive hydrazoic acid during the acidic workup phase, thereby significantly enhancing operational safety.
Q: What are the key solvent optimizations in this Valsartan process?
A: The process replaces DMF with acetonitrile for the initial alkylation step, which simplifies solvent recovery and reduces toxicity concerns. Furthermore, xylene is utilized as the solvent for the high-temperature tetrazole cyclization, providing better thermal stability and facilitating the removal of water generated during the reaction.
Q: How is high purity (>99.7%) achieved in the final API?
A: High purity is secured through a rigorous secondary crystallization protocol. Unlike single crystallization methods that may yield unstable quality, this method employs two sequential crystallizations in ethyl acetate, effectively removing trace impurities and isomeric byproducts to meet stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated optimizations described in patent CN101362728A are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Valsartan meets or exceeds the >99.7% HPLC purity benchmark. Our commitment to quality assurance ensures that the complex stereochemistry and impurity profiles of this molecule are tightly controlled throughout the entire manufacturing lifecycle.
We invite global pharmaceutical partners to collaborate with us to leverage these process efficiencies for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and supply security for your Valsartan requirements.
