Revolutionizing Bromfenac Sodium Production: A Technical Analysis of Patent CN111196770A for Global Supply Chains
Revolutionizing Bromfenac Sodium Production: A Technical Analysis of Patent CN111196770A for Global Supply Chains
The pharmaceutical landscape for non-steroidal anti-inflammatory drugs (NSAIDs) is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A pivotal development in this sector is detailed in Chinese patent CN111196770A, which discloses a remarkably simple preparation method for Bromfenac Sodium, a potent cyclooxygenase inhibitor widely used in ophthalmic solutions for treating acute pain and inflammation. This patent represents a significant departure from legacy synthetic pathways, offering a streamlined two-step protocol that addresses long-standing inefficiencies in yield, purity, and operational complexity. For R&D directors and procurement strategists alike, understanding the nuances of this technology is critical for securing a competitive edge in the global supply of high-purity pharmaceutical intermediates.
Bromfenac Sodium, chemically known as 2-amino-3-(4-bromobenzoyl) sodium phenylacetate, has historically been challenging to synthesize with high atom economy. The traditional reliance on multi-step sequences involving expensive starting materials and harsh reaction conditions has created bottlenecks in production capacity and inflated costs. The methodology outlined in CN111196770A circumvents these obstacles by utilizing readily available commodity chemicals—specifically p-bromophenylacetonitrile and 2-nitrophenylacetic acid—as the foundational building blocks. This strategic shift not only simplifies the synthetic logic but also fundamentally alters the economic profile of the final active pharmaceutical ingredient (API).
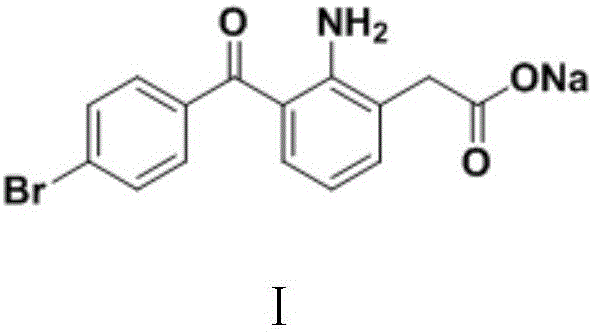
The structural integrity and therapeutic efficacy of Bromfenac Sodium depend heavily on the precise arrangement of its functional groups, particularly the amino and carboxylate positions relative to the bromobenzoyl core. As illustrated in the molecular structure, the target compound requires a specific substitution pattern that is difficult to achieve without generating significant impurities. The patent's approach ensures that the critical amino group is generated in situ during the final reduction step, thereby minimizing the need for protective group chemistry that often plagues alternative synthetic routes. This directness is a hallmark of modern process chemistry, aiming to reduce the total number of unit operations while maximizing the overall yield of the reliable pharmaceutical intermediates supplier network.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial synthesis of Bromfenac Sodium was dominated by four primary routes, each fraught with significant technical and economic drawbacks that hindered scalable production. The first conventional method, often cited in early literature, relies on a Friedel-Crafts acylation of 2-amino-4'-bromobenzophenone. This pathway is notoriously inefficient, requiring cryogenic reaction temperatures as low as -70°C to control selectivity, which imposes a massive energy burden on manufacturing facilities. Furthermore, the use of stoichiometric amounts of aluminum trichloride generates substantial quantities of acidic wastewater, creating severe environmental compliance challenges and increasing the cost of waste treatment significantly.
Another prevalent route involves the use of p-acetamidobenzene sulfonic acid as a starting material, where the sulfonic acid group acts as a temporary blocking agent. While this method achieves the desired substitution pattern, it suffers from poor atom economy because the sulfonic acid and acetyl protecting groups must be installed and subsequently removed, adding two unnecessary steps to the process. Additionally, this route also relies heavily on Friedel-Crafts chemistry, perpetuating the issues of high catalyst loading and corrosive waste generation. A third approach utilizes indole as a raw material, which, despite having high single-step yields, is prohibitively expensive for large-scale applications, rendering the final product cost-prohibitive for generic drug manufacturers seeking cost reduction in API manufacturing.
The Novel Approach
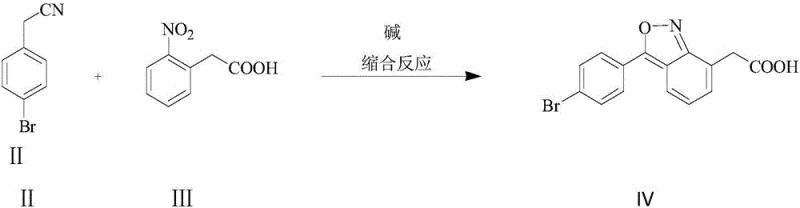
In stark contrast to these legacy methods, the novel approach described in CN111196770A introduces a convergent synthesis that drastically reduces the step count and operational complexity. By employing a base-catalyzed condensation reaction between p-bromophenylacetonitrile and 2-nitrophenylacetic acid, the process constructs the core carbon skeleton in a single pot without the need for cryogenic cooling or exotic reagents. This condensation forms a benzo[c]isoxazole intermediate, which serves as a masked precursor to the final amino-acid functionality. The subsequent step involves a straightforward reduction and salt formation, completing the synthesis in just two distinct stages. This simplification translates directly into reduced capital expenditure for reactor time and lower operational overheads.
The economic implications of this novel approach are profound for supply chain managers. By eliminating the need for expensive starting materials like indole or indoline, and avoiding the use of hazardous oxidants such as active manganese dioxide, the process inherently lowers the bill of materials. Moreover, the reaction conditions are mild, typically operating between 40°C and 100°C, which allows for the use of standard glass-lined or stainless steel reactors without specialized cooling capabilities. This accessibility makes the technology ideal for the commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations without being constrained by specialized infrastructure.
Mechanistic Insights into Base-Catalyzed Condensation and Ring Opening
The success of this synthetic route hinges on a sophisticated understanding of nucleophilic aromatic substitution and heterocyclic ring dynamics. In the first step, the reaction is initiated by the deprotonation of p-bromophenylacetonitrile by a strong base, such as sodium methoxide or potassium hydroxide, to generate a resonance-stabilized carbanion. This nucleophile then attacks the electron-deficient carbon atom at the ortho-position of the nitro group in 2-nitrophenylacetic acid. This specific attack triggers a cascade of intramolecular cyclization events, ultimately yielding the 7-(4-bromophenyl)-3-carboxymethylbenzo[c]isoxazole intermediate. The elegance of this mechanism lies in its specificity; the nitro group acts as a powerful electron-withdrawing director, ensuring that the condensation occurs exclusively at the desired position, thereby minimizing regio-isomeric impurities.
A critical aspect of this mechanism, often overlooked in less optimized processes, is the control of reactant concentration. The patent explicitly mandates the dropwise addition of the nitrile component into the reaction mixture containing the base and the nitro-acid. This technique maintains a low instantaneous concentration of the nitrile carbanion, effectively suppressing the competing self-condensation reaction where two nitrile molecules might react with each other. By kinetically favoring the cross-condensation with the nitro-acid, the process achieves exceptional selectivity. This mechanistic control is vital for producing high-purity OLED material or pharmaceutical grades where trace impurities can compromise downstream crystallization or biological activity.
The second phase of the synthesis involves the reductive cleavage of the isoxazole ring, a transformation that simultaneously unveils the free amino group and preserves the carboxylic acid functionality. Whether achieved through catalytic hydrogenation using palladium on carbon or Raney nickel, or via metal-acid reduction with iron powder, this step is highly chemoselective. The reduction targets the weak N-O bond of the isoxazole ring while leaving the robust carbon-carbon backbone and the aryl bromide intact. Following reduction, the immediate alkalization with sodium hydroxide converts the free acid into the stable sodium salt. This tandem reduction-salt formation sequence ensures that the final product precipitates cleanly from the reaction medium, facilitating easy isolation and drying without the need for complex chromatographic purification.
How to Synthesize Bromfenac Sodium Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel, typically a four-neck flask equipped with efficient stirring and temperature control, reflecting the standards required for reducing lead time for high-purity pharmaceutical intermediates. The choice of solvent is flexible, encompassing alcohols like methanol and ethanol, or ethers like tetrahydrofuran, allowing manufacturers to optimize based on local availability and recycling capabilities. The base selection is equally critical, with sodium methoxide and potassium hydroxide demonstrating superior performance in generating the necessary nucleophile without inducing excessive side reactions.
- Perform a condensation reaction between p-bromophenylacetonitrile and 2-nitrophenylacetic acid in an alcoholic solvent under alkaline conditions, utilizing a dropwise addition strategy to maximize selectivity.
- Isolate the intermediate 7-(4-bromophenyl)-3-carboxymethylbenzo[c]isoxazole through acidification, filtration, and drying.
- Subject the intermediate to catalytic hydrogenation or metal-acid reduction to open the isoxazole ring, followed by alkalization with sodium hydroxide to form the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By shifting away from specialized, high-cost precursors like indoline or protected benzophenones to commodity chemicals like p-bromophenylacetonitrile, companies can significantly mitigate supply risk. These commodity feedstocks are produced by a wide range of global chemical manufacturers, ensuring a robust and competitive supply base that prevents single-source bottlenecks and stabilizes pricing volatility over the long term.
- Cost Reduction in Manufacturing: The elimination of cryogenic processing and the reduction of reaction steps from four or five down to two results in substantial operational savings. Energy costs associated with maintaining -70°C environments are completely eradicated, and the reduced cycle time allows for higher throughput per reactor volume. Furthermore, the avoidance of expensive catalysts like boron trichloride and the minimization of aluminum chloride usage lower the direct material costs, while the simplified workup procedures reduce labor and utility consumption associated with solvent recovery and waste neutralization.
- Enhanced Supply Chain Reliability: The robustness of this chemistry enhances supply continuity by reducing the likelihood of batch failures due to sensitive reaction conditions. Traditional routes involving moisture-sensitive Lewis acids or unstable oxidants are prone to variability, whereas this base-catalyzed condensation is tolerant and forgiving. The ability to recycle the cyanide byproduct back into the raw material supply chain further insulates the manufacturer from external raw material price shocks, creating a more circular and resilient production ecosystem that guarantees consistent delivery schedules to downstream API formulators.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The generation of wastewater is minimized, and the specific capture of hydrogen cyanide byproduct as sodium cyanide for reuse aligns with green chemistry principles. This reduces the regulatory burden associated with hazardous waste disposal and lowers the carbon footprint of the manufacturing process. Such environmental stewardship is increasingly a prerequisite for qualifying as a vendor for major multinational pharmaceutical corporations, ensuring long-term contract viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms historical precedents in terms of yield, purity, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does the new condensation route improve upon traditional Friedel-Crafts methods?
A: Unlike traditional routes requiring cryogenic temperatures (-70°C) and excessive Lewis acids like aluminum trichloride, this novel method operates at mild temperatures (40-100°C) with significantly reduced wastewater generation and superior atom economy.
Q: What are the critical control points for ensuring high purity in this synthesis?
A: The key to high purity lies in the dropwise addition of p-bromophenylacetonitrile during the condensation step. This maintains a low concentration of the nitrile, preventing self-condensation side reactions and ensuring the carbanion selectively attacks the ortho-nitro position.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to the use of commodity raw materials, the elimination of hazardous oxidants like manganese dioxide, and the ability to recycle cyanide byproducts, making it economically and environmentally viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromfenac Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN111196770A can be fully realized in an industrial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Bromfenac Sodium meets the exacting standards required for ophthalmic and systemic applications.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this streamlined route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless integration of this high-performance intermediate into your manufacturing pipeline.
