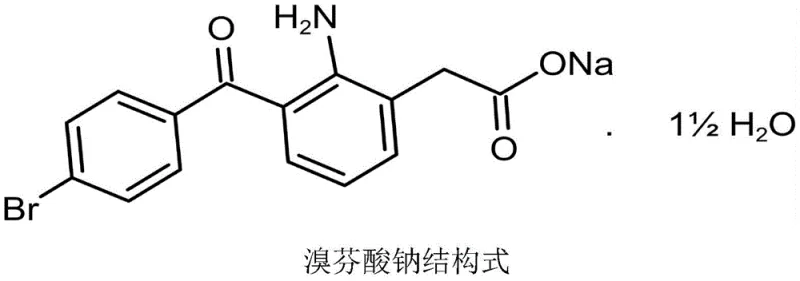
Advanced Bromfenac Sodium Synthesis: Eliminating Indole Impurities for Superior API Quality
Advanced Bromfenac Sodium Synthesis: Eliminating Indole Impurities for Superior API Quality
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with operational safety, particularly for potent non-steroidal anti-inflammatory drugs (NSAIDs) like bromfenac sodium. A groundbreaking approach detailed in patent CN113698308A introduces a transformative synthesis strategy that fundamentally shifts away from traditional indole-based scaffolds. This novel methodology utilizes o-aminophenylacetic acid as the primary starting material, orchestrating a sequence of acylation, sulfonation, and selective Friedel-Crafts reactions to construct the target molecule with exceptional precision. By circumventing the formation of the indole ring entirely, this process addresses critical quality attributes that have long plagued existing manufacturing routes, specifically the presence of persistent indole-related impurities and pH instability in the final active pharmaceutical ingredient (API). For global supply chain leaders and R&D directors, this represents a significant leap forward in process chemistry, offering a pathway to ultra-high purity intermediates that meet the stringent demands of ophthalmic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of bromfenac sodium has relied heavily on routes originating from indole or indoline derivatives, as disclosed in various prior art patents such as CN106397235A and EP0221753. These conventional pathways typically involve the construction of the indole core followed by complex functionalization steps including halogenation and oxidation. However, these methods suffer from inherent structural liabilities; the indole scaffold is prone to generating specific byproducts that retain the indole ring structure. These indole-containing impurities possess physicochemical properties remarkably similar to the target molecule, rendering their removal via standard crystallization or chromatography extremely difficult and costly. Furthermore, many of these legacy processes rely on hazardous reagents such as boron trichloride (BCl3) and N-bromosuccinimide (NBS), which pose severe safety risks regarding toxicity, corrosivity, and potential explosion hazards during scale-up. Additionally, the reliance on phosphoric acid for hydrolysis in older methods frequently results in the entrapment of phosphate salts, leading to final products that fail alkalinity tests with pH values exceeding the acceptable range of 8.5 to 9.5.
The Novel Approach
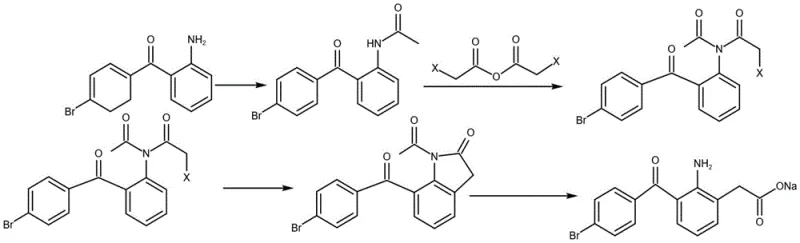
In stark contrast, the innovative synthesis described in patent CN113698308A adopts a linear, non-cyclizing strategy that begins with the commercially abundant o-aminophenylacetic acid. This route strategically employs a sulfonation step to direct subsequent substitution, effectively blocking unwanted reaction sites and ensuring regioselectivity without the need for complex protecting group strategies typical of indole chemistry. The process replaces high-risk halogenating agents with safer alternatives and utilizes hydrochloric acid for the final deprotection, which volatilizes easily and does not leave behind non-volatile acid salts that compromise pH stability. By decoupling the synthesis from the indole nucleus, the method inherently prevents the formation of the most troublesome class of impurities. This results in a crude product with significantly higher purity, simplifying downstream purification and reducing the overall environmental footprint by eliminating the need for aggressive heavy metal oxidants like manganese dioxide often found in older protocols.
Mechanistic Insights into Regioselective Sulfonation and Hydrolysis
The core chemical ingenuity of this new process lies in the precise manipulation of electronic effects on the benzene ring to achieve perfect regiocontrol. The initial acylation of o-aminophenylacetic acid protects the amine group, preventing it from interfering with subsequent electrophilic aromatic substitutions. The critical step involves the sulfonation of this protected intermediate using concentrated sulfuric acid. Mechanistically, the sulfonic acid group acts as a powerful electron-withdrawing directing group that occupies the para-position relative to the acetamido group. This occupation is strategic; it blocks the position that might otherwise lead to polysubstitution or incorrect isomer formation during the subsequent Friedel-Crafts acylation. When p-bromobenzoyl chloride is introduced in the presence of aluminum trichloride, the acylation occurs exclusively at the desired ortho-position relative to the acetamido group, driven by the steric and electronic landscape created by the sulfonate blocker. This level of control is difficult to achieve in indole-based systems where the heterocyclic nitrogen can complicate Lewis acid coordination.
Furthermore, the hydrolysis mechanism in this novel route is optimized for purity and compliance. In traditional methods using phosphoric acid, the resulting phosphate anions form stable ion pairs with the amine cation of the drug substance, which are thermodynamically stable and resist removal during washing. The new method employs hydrochloric acid for the hydrolysis of the intermediate III (3-(4-bromobenzoyl)-2-acetamido-5-sulfophenylacetic acid). The chloride ion is a weaker coordinating anion in this context and the resulting ammonium chloride salts are highly water-soluble. During the subsequent basification and crystallization steps, these salts are efficiently partitioned into the aqueous phase and discarded. This mechanistic distinction ensures that the final bromfenac sodium crystal lattice is free from occluded acid salts, directly addressing the critical quality attribute of pH stability which is vital for ophthalmic tolerance and shelf-life.
How to Synthesize Bromfenac Sodium Efficiently
The operational execution of this synthesis is designed for reproducibility and safety, utilizing standard unit operations found in most multipurpose pharmaceutical plants. The process flows logically from protection to functionalization and finally to deprotection and salt formation. Detailed below is the high-level workflow that R&D teams can adapt for pilot plant trials, focusing on the critical control points identified in the patent data.
- Acylation of o-aminophenylacetic acid with acetic anhydride to form o-acetaminophenylacetic acid.
- Sulfonation of the intermediate using sulfuric acid to introduce a sulfonic acid group at the 5-position.
- Friedel-Crafts acylation with p-bromobenzoyl chloride and aluminum trichloride to attach the bromobenzoyl moiety.
- Acid hydrolysis using hydrochloric acid to remove the acetyl and sulfonyl groups, yielding bromfenac.
- Neutralization with sodium hydrox followed by crystallization to obtain high-purity bromfenac sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from specialized, high-cost heterocyclic starting materials like indole or indoline to commodity chemicals like o-aminophenylacetic acid significantly de-risks the supply base. Commodity acids and anhydrides are produced at massive scales globally, ensuring price stability and continuity of supply even during market fluctuations. Moreover, the elimination of hazardous reagents such as boron trichloride and N-bromosuccinimide reduces the regulatory burden associated with handling, storage, and waste disposal of toxic substances. This simplification of the EHS (Environment, Health, and Safety) profile translates directly into lower operational overheads and reduced insurance liabilities for manufacturing partners.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by yield optimization and simplified purification. By avoiding the formation of hard-to-remove indole impurities, the number of recrystallization cycles required to meet specification is drastically reduced. Each skipped purification step saves solvent, energy, and labor costs. Additionally, the use of hydrochloric acid instead of phosphoric acid eliminates the need for extensive washing protocols designed to remove phosphate residues, further shortening cycle times. The overall atom economy is improved by the direct nature of the substitution reactions, minimizing waste generation and maximizing the output per batch of raw materials consumed.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions. The process operates at moderate temperatures and pressures, reducing the risk of batch failures due to thermal runaways or equipment limitations. The raw materials are widely available from multiple global suppliers, preventing single-source bottlenecks. This diversity in sourcing options allows procurement teams to negotiate better terms and ensures that production schedules are not disrupted by raw material shortages. The consistency of the intermediate quality also means that downstream formulation teams receive a more uniform API, reducing variability in the final drug product.
- Scalability and Environmental Compliance: From a sustainability perspective, this route aligns with green chemistry principles by reducing the use of heavy metals and toxic halogenating agents. The waste stream is simpler to treat, primarily consisting of aqueous salts and organic solvents that can be recovered and recycled. The scalability is proven by the straightforward nature of the unit operations—filtration, distillation, and crystallization—which are easily transferred from laboratory glassware to industrial reactors without complex engineering modifications. This ease of scale-up ensures that commercial volumes can be reached rapidly to meet market demand for ophthalmic NSAIDs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality implications of this new synthesis technology. These answers are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on why this method represents a superior choice for modern API manufacturing.
Q: How does this new synthesis method improve impurity profiles compared to indole-based routes?
A: Traditional indole-based routes often generate structurally similar indole ring-containing impurities that are difficult to separate. This novel method starts from o-aminophenylacetic acid, completely bypassing the indole scaffold formation, thereby eliminating these specific genotoxic or hard-to-remove impurities and achieving purity levels exceeding 99.9%.
Q: Why is hydrochloric acid preferred over phosphoric acid in the hydrolysis step?
A: The use of phosphoric acid in prior art methods leads to the formation of phosphate acid salts which are difficult to remove completely, causing the final product's pH to exceed pharmacopeial standards (pH 8.5-9.5). Using hydrochloric acid ensures volatile byproducts and soluble salts that are easily washed away, guaranteeing strict pH compliance.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available raw materials like o-aminophenylacetic acid and avoids high-risk reagents such as boron trichloride or N-bromosuccinimide (NBS). The reaction conditions are mild and controllable, making it highly scalable for industrial production with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromfenac Sodium Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your final ophthalmic formulation depends entirely on the quality of the active ingredient. Our technical team has extensively analyzed the synthesis route described in patent CN113698308A and validated its potential for producing ultra-high purity bromfenac sodium. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facility is equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch meets stringent purity specifications and complies with international pharmacopeial standards for pH and related substances.
We invite you to collaborate with us to leverage this advanced chemistry for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how this novel synthesis can enhance your product's quality profile while optimizing your manufacturing costs.
