Advanced Donepezil Intermediate Synthesis: A Scalable Route for Global API Manufacturing
Advanced Donepezil Intermediate Synthesis: A Scalable Route for Global API Manufacturing
The global demand for acetylcholinesterase inhibitors, particularly Donepezil (E2020), continues to surge as the primary therapeutic intervention for Alzheimer's disease, necessitating robust and economically viable manufacturing pathways. Patent CN1613848A introduces a transformative synthetic methodology that addresses the critical bottlenecks of yield, safety, and scalability inherent in legacy production techniques. This technical insight report analyzes the proprietary three-step sequence which leverages a unique acid-activation strategy to protonate the pyridine ring prior to hydrogenation. By fundamentally altering the electronic properties of the intermediate, this approach enables catalytic reduction under exceptionally mild conditions, bypassing the harsh reagents and complex purification protocols that have historically plagued industrial scale-up. For R&D directors and supply chain leaders, understanding this mechanistic shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Donepezil have been fraught with significant technical and economic inefficiencies that hinder large-scale commercial viability. Early methodologies reported by major pharmaceutical entities often resulted in total recovery rates of less than 20%, primarily due to the formation of intractable byproducts and the necessity for labor-intensive column chromatography purification. Subsequent iterations attempted to streamline the process but frequently encountered severe limitations; for instance, certain routes required the use of Sodium Hydride (NaH) and highly concentrated sodium hydroxide solutions under strictly anhydrous conditions. These requirements not only escalate facility investment costs due to the need for specialized moisture-free equipment but also introduce substantial safety hazards associated with handling pyrophoric reagents and corrosive bases. Furthermore, the sensitivity of the carbonyl group during catalytic hydrogenation in these traditional pathways often leads to over-reduction to hydroxyl derivatives, drastically compromising the purity profile and necessitating additional downstream processing steps that erode overall profit margins.
The Novel Approach
The innovative strategy detailed in the patent data circumvents these obstacles by introducing a protonation step that fundamentally stabilizes the reaction intermediate before the critical hydrogenation phase. Instead of relying on dangerous bases or extreme anhydrous environments, this method utilizes strong organic acids, such as p-toluenesulfonic acid, to convert the pyridine moiety into a stable pyridinium salt. This structural modification activates the ring system, allowing for selective hydrogenation of the double bond without affecting the sensitive carbonyl functionality or causing unwanted benzyl cleavage. The result is a dramatic improvement in process robustness, where each step can be purified through simple recrystallization rather than complex chromatography. This shift from hazardous, low-yield chemistry to a mild, acid-catalyzed protocol represents a paradigm shift in cost reduction in API manufacturing, offering a pathway that is not only safer for operators but also significantly more amenable to continuous processing and large-volume production.
Mechanistic Insights into Acid-Activated Pyridine Hydrogenation
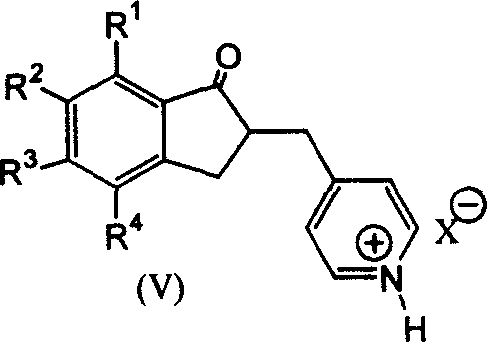
The core chemical innovation lies in the strategic manipulation of the pyridine ring's electronic environment through protonation. In conventional synthesis, the conjugated system of the 2-(4-pyridine methylene)-1-indanone derivative presents a challenge for catalytic hydrogenation because the pyridine nitrogen can coordinate with the metal catalyst, potentially poisoning it or leading to non-selective reduction. By treating the intermediate with a strong acid (HX), the nitrogen atom is fully protonated to form a pyridinium ammonium salt. This positive charge withdraws electron density from the ring, effectively activating it towards nucleophilic attack by hydrogen while simultaneously preventing the coordination issues that plague neutral pyridine substrates. This activation allows the use of standard catalysts like Platinum Dioxide (PtO2) at ambient temperatures and low hydrogen pressures (1-5 atm), conditions that are far gentler than the high-pressure or high-temperature regimes required by older methods. The preservation of the carbonyl group during this reduction is critical, as it maintains the structural integrity required for the final alkylation step, ensuring that the final product meets stringent purity specifications without the need for extensive remediation.
Following the hydrogenation, the resulting piperidine ammonium salt serves as a highly reactive precursor for the final alkylation. The presence of the acid counter-ion facilitates the subsequent nucleophilic substitution reaction with benzyl halides in the presence of mild inorganic bases like potassium carbonate. This sequence minimizes the formation of quaternary ammonium byproducts that typically arise from direct N-benzylation of neutral amines. The mechanistic elegance of this route ensures that impurities are kept to a minimum, with the patent data indicating purities exceeding 97% after simple filtration and drying. For technical teams evaluating process feasibility, this level of control over the impurity profile is paramount, as it directly correlates to the ease of regulatory filing and the reliability of the supply chain. The ability to achieve such high selectivity under mild conditions underscores the superiority of this acid-activation mechanism over traditional base-mediated approaches.
How to Synthesize Donepezil Intermediate Efficiently
The implementation of this synthesis route involves a streamlined three-step protocol designed for maximum operational efficiency and minimal waste generation. The process begins with the condensation of 5,6-dimethoxy-1-indanone and 4-pyridinecarboxaldehyde in a solvent such as toluene, catalyzed by p-toluenesulfonic acid under reflux conditions to yield the pyridinium salt intermediate with yields reaching up to 94%. This is followed by the critical hydrogenation step using Platinum Dioxide in methanol at room temperature, which converts the pyridinium salt to the corresponding piperidine derivative with near-quantitative conversion. The final step involves alkylation with a benzyl halide in a polar aprotic solvent like DMF using potassium carbonate as the base. Detailed standardized operating procedures, including specific molar ratios, solvent volumes, and temperature ramps essential for GMP compliance, are outlined in the technical guide below.
- Condense 5,6-dimethoxy-1-indanone with 4-pyridinecarboxaldehyde in the presence of a strong acid like p-toluenesulfonic acid to form the pyridinium salt intermediate.
- Perform catalytic hydrogenation on the pyridinium salt using Platinum Dioxide (PtO2) at mild temperatures (room temperature to 50°C) and low pressure (1-5 atm) to generate the piperidine derivative.
- Alkylate the resulting piperidine salt with a benzyl halide in the presence of a base such as potassium carbonate to yield the final Donepezil target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend beyond mere technical performance. The elimination of hazardous reagents like Sodium Hydride removes a significant bottleneck in raw material sourcing and storage, as it negates the need for specialized hazardous material handling protocols and expensive safety infrastructure. This simplification of the chemical bill of materials directly translates to enhanced supply chain reliability, as the required reagents—strong organic acids, platinum catalysts, and common inorganic bases—are commodity chemicals with stable global availability. Furthermore, the ability to purify intermediates via recrystallization rather than column chromatography drastically reduces solvent consumption and waste disposal costs, aligning with increasingly stringent environmental regulations and sustainability goals. These factors collectively contribute to a more resilient and cost-effective supply chain, reducing the risk of production stoppages due to reagent shortages or safety incidents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial simplification of the operational workflow and the elimination of expensive purification technologies. By avoiding the use of column chromatography, which is notoriously solvent-intensive and difficult to scale, the process significantly lowers the variable costs associated with solvent purchase and recovery. Additionally, the high yield of each individual step, culminating in a total recovery rate of approximately 82%, means that less raw material is wasted per kilogram of final product, optimizing the overall material balance. The use of mild reaction conditions also reduces energy consumption, as there is no need for cryogenic cooling or high-temperature heating, further driving down utility costs. These cumulative efficiencies allow for a competitive pricing structure that is sustainable even in volatile raw material markets.
- Enhanced Supply Chain Reliability: Operational continuity is secured through the robustness of the reaction conditions, which are tolerant to minor variations in temperature and pressure. Unlike processes requiring absolute anhydrous conditions, this method can be executed in standard reactor vessels without the need for rigorous moisture exclusion, reducing the likelihood of batch failures due to environmental factors. The high purity of the intermediates produced (>97%) ensures that downstream processing is predictable and consistent, minimizing the need for rework or rejection of off-spec material. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by global pharmaceutical clients, thereby strengthening the partnership between supplier and manufacturer.
- Scalability and Environmental Compliance: The pathway is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing unit operations that are standard in the fine chemical industry. The reduction in hazardous waste generation, particularly the avoidance of heavy metal contamination often associated with difficult purifications, simplifies the effluent treatment process. The solvents used, such as methanol, ethanol, and ethyl acetate, are readily recyclable, supporting a circular economy approach within the manufacturing plant. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, a factor of increasing importance to end-user pharmaceutical companies seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific challenges identified in the background art and the beneficial effects demonstrated in the patent examples. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this route for their own production needs or for qualifying a new vendor. The answers provided reflect the empirical data and mechanistic logic presented in the intellectual property documentation.
Q: How does the acid-activation method improve yield compared to traditional routes?
A: Traditional routes often suffer from low yields (below 30%) due to difficult purification and side reactions during hydrogenation. The acid-activation method protonates the pyridine ring, facilitating selective hydrogenation under mild conditions, achieving total yields up to 82%.
Q: Does this synthesis route require hazardous reagents like Sodium Hydride?
A: No. Unlike previous optimized methods that require dangerous Sodium Hydride (NaH) and strictly anhydrous conditions, this novel route utilizes stable strong acids and standard catalytic hydrogenation, significantly improving operational safety.
Q: What represents the critical quality control parameter for the intermediate?
A: Purity is paramount. The process allows for recrystallization purification rather than column chromatography, consistently delivering intermediates with purity greater than 97%, which is essential for downstream API compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications like Donepezil depends on a partnership grounded in technical excellence and unwavering reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity Donepezil intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in acid-catalyzed transformations and catalytic hydrogenation allows us to optimize this specific patent route for maximum throughput and minimal environmental impact, providing our clients with a secure and sustainable source of critical pharmaceutical building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest degree of confidence and technical support. Let us be your partner in navigating the complexities of modern API manufacturing.
