Revolutionizing Donepezil Manufacturing: A High-Yield Route for Global API Supply Chains
Revolutionizing Donepezil Manufacturing: A High-Yield Route for Global API Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value neurological agents, and the synthesis of Donepezil (E2020) remains a critical focus for Alzheimer's disease treatment markets. Patent CN1280273C introduces a transformative methodology that addresses the longstanding inefficiencies of prior art, offering a novel three-step reaction sequence that dramatically enhances overall yield and operational simplicity. Unlike conventional routes that struggle with complex purification and low conversion rates, this invention leverages a unique acid-catalyzed activation strategy to stabilize reactive intermediates, enabling a total recovery rate that can reach an impressive 82 percent. For global stakeholders, this represents a significant leap forward in process chemistry, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates while mitigating the technical risks associated with traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Donepezil has been plagued by significant technical bottlenecks that hinder cost efficiency and supply stability. Early methodologies reported by major pharmaceutical entities often yielded total recoveries of less than 20 percent, primarily due to the formation of stubborn byproducts that necessitated labor-intensive column chromatographic isolation. Furthermore, improved routes attempting to bypass these issues frequently introduced new hazards, such as the requirement for strictly anhydrous conditions and the use of highly corrosive reagents like sodium hydride. These demanding processing conditions not only escalate facility investment costs but also complicate waste management protocols, creating substantial barriers for manufacturers aiming to achieve green chemistry standards. The cumulative effect of these limitations is a fragile supply chain characterized by high production costs and inconsistent batch quality, which poses a severe risk to procurement managers seeking long-term vendor reliability.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the synthetic logic by introducing a protonation step that activates the pyridine ring prior to hydrogenation. By reacting 4-pyridylaldehyde with an indanone derivative in the presence of a strong acid such as tosic acid, the process generates a stable pyridine ammonium salt intermediate that is uniquely susceptible to selective reduction. This clever chemical maneuver allows the subsequent catalytic hydrogenation to proceed under exceptionally mild conditions, specifically at atmospheric pressure and ambient temperatures, thereby eliminating the need for high-pressure reactors. Moreover, the resulting intermediates can be purified through straightforward recrystallization rather than chromatography, which drastically simplifies the downstream processing workflow. This approach not only boosts the yield of the critical hydrogenation step to over 90 percent but also ensures that the final alkylation proceeds with high fidelity, delivering a commercially viable pathway for high-purity API intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Pyridine Activation
The core scientific breakthrough of this synthesis lies in the mechanistic manipulation of the pyridine ring's electronic properties through protonation. In traditional hydrogenation scenarios, the conjugated system of the pyridine ring often resists reduction or leads to unwanted side reactions such as the reduction of the adjacent carbonyl group. However, by converting the pyridine nitrogen into a quaternary ammonium species via strong acid treatment, the electron density of the ring is sufficiently altered to facilitate rapid and selective hydrogenation using catalysts like Platinum Dioxide. This activation ensures that the double bonds within the heterocyclic ring are reduced to form the piperidine structure without affecting other sensitive functional groups on the indanone scaffold. Such selectivity is paramount for maintaining the structural integrity of the molecule and preventing the formation of hydroxyl-reduced impurities that typically plague less controlled reaction environments.
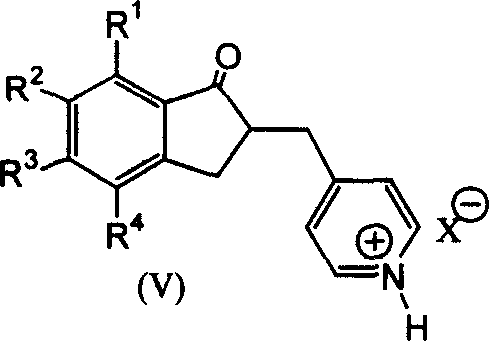
Furthermore, this mechanistic pathway offers superior control over the impurity profile, which is a critical metric for R&D directors overseeing regulatory compliance. The use of specific strong acids like methylsulfonic acid or tosic acid creates a reaction environment where the protonated intermediate remains stable throughout the hydrogenation process, preventing premature deprotection or decomposition. This stability allows for the use of common solvents such as methanol or ethanol, removing the logistical burden of sourcing and handling exotic anhydrous solvents. Consequently, the impurity spectrum is significantly narrowed, reducing the burden on analytical quality control teams and ensuring that the final product meets stringent pharmacopeial standards with minimal additional purification effort. The ability to tune the reaction by selecting specific acid catalysts provides a versatile toolkit for optimizing the process across different manufacturing scales.
How to Synthesize Donepezil Efficiently
The implementation of this synthesis route involves a logical progression of three distinct chemical transformations that are optimized for industrial throughput. The process begins with the condensation of the indanone precursor with the pyridine aldehyde, followed by the critical activation and hydrogenation sequence, and concludes with the final alkylation to install the benzyl moiety. Each step has been refined to maximize atom economy and minimize waste generation, making it an ideal candidate for facilities aiming to reduce their environmental footprint. Detailed standard operating procedures regarding specific molar ratios, temperature gradients, and workup protocols are essential for replicating the high yields reported in the patent literature. For technical teams preparing for technology transfer, understanding the nuances of the catalyst loading and acid selection is vital to achieving the reported 82 percent total recovery.
- Condense 5,6-dimethoxy-1-indanone with 4-pyridinecarboxaldehyde using a strong acid catalyst like tosic acid to form the pyridine methylene intermediate.
- Perform catalytic hydrogenation on the protonated pyridine intermediate using Platinum Dioxide (PtO2) under mild atmospheric pressure to selectively reduce the pyridine ring.
- Execute the final alkylation reaction using a suitable base like potassium carbonate to attach the benzyl group, yielding the target Donepezil compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of procurement managers and supply chain heads. By eliminating the need for column chromatography and replacing it with crystallization, the process significantly reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste disposal. The shift away from hazardous reagents like sodium hydride also lowers the insurance and safety compliance costs associated with manufacturing, while the ability to run reactions at atmospheric pressure reduces the capital expenditure required for specialized high-pressure equipment. These factors combine to create a leaner, more agile production model that can respond more effectively to market demand fluctuations without compromising on margin or quality.
- Cost Reduction in Manufacturing: The elimination of expensive purification techniques and hazardous reagents translates directly into a lower cost of goods sold. By avoiding the use of sodium hydride and anhydrous conditions, manufacturers can utilize standard reactor vessels and common solvents, which drastically simplifies the operational budget. Additionally, the high yield of each step means that less starting material is wasted, further enhancing the economic efficiency of the entire production campaign. This logical deduction of cost benefits suggests a highly competitive pricing structure for the final API intermediate compared to legacy methods.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more consistent supply of high-purity intermediates, mitigating the risk of production delays caused by difficult purifications or low yields. Since the process relies on readily available catalysts like Platinum Dioxide and common acids, the supply chain is less vulnerable to shortages of exotic or highly regulated chemicals. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the strict delivery timelines required by downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this process inherently scalable from pilot plant to full commercial production. The reduction in three-waste discharge, particularly the avoidance of heavy metal contamination from complex chromatography and the use of less toxic solvents, aligns perfectly with modern environmental regulations. This compliance advantage not only future-proofs the manufacturing site against tightening ecological laws but also enhances the brand reputation of the supplier as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks.
Q: How does this new synthesis route improve upon traditional Eisai methods?
A: Traditional methods often suffer from low total yields (below 30%) and require harsh anhydrous conditions or expensive column chromatography. This patented route achieves up to 82% total yield by utilizing acid-catalyzed activation which allows for mild hydrogenation conditions and simple recrystallization purification.
Q: What are the key advantages for large-scale procurement of Donepezil intermediates?
A: The process eliminates the need for hazardous reagents like sodium hydride (NaH) and avoids complex purification steps. This significantly reduces production costs, minimizes three-waste discharge, and simplifies equipment requirements, ensuring a more stable and cost-effective supply chain for bulk buyers.
Q: Is the catalytic hydrogenation step safe for industrial scale-up?
A: Yes, the hydrogenation step operates effectively at atmospheric pressure (1 atm) and moderate temperatures (room temperature to 50°C). This contrasts sharply with high-pressure hydrogenation methods, drastically lowering safety risks and capital investment for reactor infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN1280273C for securing a competitive edge in the neurological drug market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield chemistry are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Donepezil intermediate meets the exacting standards required for global regulatory submissions. We are committed to translating complex patent innovations into reliable, commercial-grade supply solutions.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both your production costs and supply security.
