Advanced Manufacturing of Rosuvastatin Calcium Intermediates via Optimized Grignard and Wittig Routes
Advanced Manufacturing of Rosuvastatin Calcium Intermediates via Optimized Grignard and Wittig Routes
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of high-value statin intermediates, particularly for next-generation HMG-CoA reductase inhibitors like Rosuvastatin Calcium. Patent CN101735272B discloses a groundbreaking seven-step synthetic methodology that transforms simple, commercially available feedstocks into the critical phosphonium salt intermediate required for Rosuvastatin assembly. This novel approach addresses significant bottlenecks found in legacy processes by leveraging a copper-catalyzed Grignard coupling followed by a stereoselective Wittig olefination. The target molecule, Rosuvastatin Calcium, is a potent lipid-lowering agent known for its high efficacy and low toxicity profile, making the reliable supply of its precursors a strategic priority for global generic and innovator drug manufacturers alike.
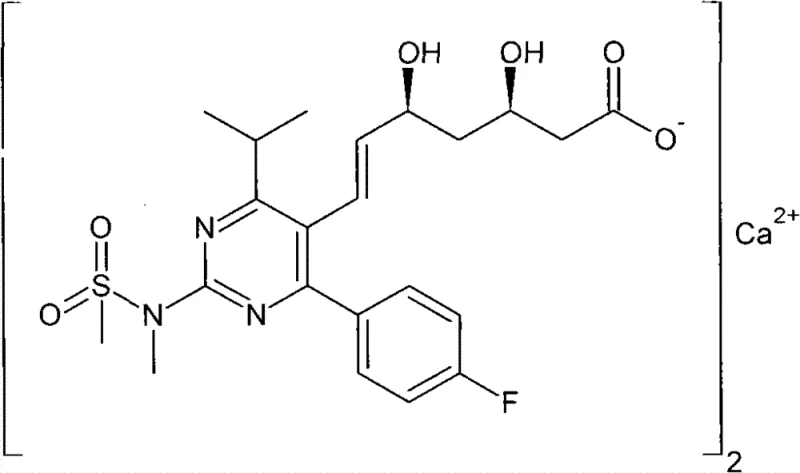
As illustrated in the structural diagram, the complexity of the final active pharmaceutical ingredient demands precise control over stereochemistry and functional group tolerance during the intermediate stages. The disclosed method starts from vinyl chloride and R-epoxy chloropropane, bypassing the need for exotic starting materials. By integrating nucleophilic substitution, alcoholysis, hydroxyl protection, oxidation, acylation, and Wittig reaction steps, the process achieves a stable and high-yielding route. This technical breakthrough not only enhances the purity profile of the resulting intermediate but also aligns with modern green chemistry principles by minimizing environmental pollution and simplifying waste treatment protocols, thereby offering a compelling value proposition for procurement teams focused on sustainable sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key Rosuvastatin intermediates has been plagued by economic and operational inefficiencies inherent in earlier patented technologies. For instance, the route disclosed in PCT application WO/2006/091771 relies heavily on the use of bulky carboxy protective groups to enhance the stability of the ester moiety and improve selectivity during mono-methyl hydrolysis. While chemically effective, this requirement introduces significant cost penalties; if smaller, cheaper groups like methyl are used, the difficulty of selective hydrolysis increases dramatically, often leading to poor yields or complete failure to isolate the desired mono-hydrolysate. Furthermore, alternative literature methods, such as those described in J. Org. Chem. (1994), utilize expensive anhydride compounds as starting materials, which drastically inflates the raw material costs and renders the process economically unviable for large-scale commercial manufacturing.
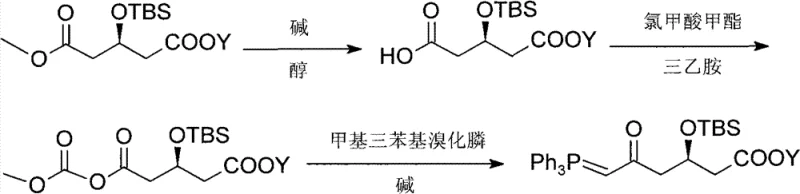
These conventional limitations create a fragile supply chain where minor fluctuations in the price of specialized reagents can disrupt production schedules. The reliance on complex protecting group strategies also adds unnecessary synthetic steps, increasing the overall cycle time and the potential for impurity generation. In an industrial setting, the disposal of waste streams associated with these bulky protecting groups poses additional environmental compliance challenges. Consequently, there is a pressing need for a streamlined synthesis that eliminates these costly dependencies while maintaining the rigorous stereochemical integrity required for downstream API production, a gap that the current invention successfully fills through its innovative use of common petrochemical derivatives.
The Novel Approach
The methodology presented in CN101735272B represents a paradigm shift by utilizing vinyl chloride and R-epoxy chloropropane as the foundational building blocks. This seven-step sequence begins with the formation of a vinyl chloride Grignard reagent, which undergoes a copper-catalyzed coupling with the epoxide to establish the carbon backbone with high regioselectivity. Unlike the prior art, this route does not depend on sterically demanding protecting groups to drive selectivity; instead, it leverages the intrinsic reactivity of the intermediates and precise temperature control. The subsequent transformation involves a nucleophilic substitution with sodium cyanide, followed by alcoholysis under dry hydrogen chloride gas, which efficiently installs the ester functionality without the need for expensive activation agents.
Furthermore, the oxidation step employs acidified potassium permanganate under mild conditions (0-10°C), a reagent that is both cost-effective and easy to handle on a multi-ton scale. The final stages involve acylation with methyl chloroformate and a Wittig reaction using methyltriphenylphosphonium bromide and n-butyllithium. This sequence ensures that the critical double bond geometry is established with high fidelity. The entire process is characterized by mild reaction conditions, typically ranging from -70°C to ambient temperature, which reduces energy consumption and equipment stress. By avoiding the pitfalls of bulky protecting groups and expensive anhydrides, this novel approach offers a direct, linear path to the target phosphonium salt, significantly enhancing the overall process mass intensity and economic feasibility for industrial partners.
Mechanistic Insights into Cu-Catalyzed Grignard Coupling and Wittig Olefination
The cornerstone of this synthetic strategy is the initial copper-catalyzed Grignard reaction, which sets the stereochemical foundation for the entire molecule. In this step, vinyl magnesium chloride reacts with R-epoxy chloropropane in the presence of cuprous chloride (CuCl). The copper catalyst plays a pivotal role in directing the nucleophilic attack of the Grignard reagent to the less hindered carbon of the epoxide ring, ensuring the formation of the desired (2R)-1-chloro-2-hydroxyl-4-amylene with high regioselectivity. The patent specifies a molar ratio of CuCl to epoxide between 0.10 and 0.15:1, optimized to balance catalytic activity with cost. Maintaining the reaction temperature between -35°C and -25°C is critical to suppress side reactions such as homocoupling of the Grignard reagent or elimination pathways that could compromise the yield and optical purity of the chiral alcohol intermediate.
Following the backbone construction, the installation of the exocyclic double bond via the Wittig reaction is another mechanistic highlight. The process generates the phosphorus ylide in situ by treating methyltriphenylphosphonium bromide with n-butyllithium at cryogenic temperatures (-60°C to -55°C). This low-temperature generation of the ylide is essential to prevent its decomposition and to ensure high reactivity towards the ketone intermediate formed after the oxidation and acylation steps. The subsequent addition of the acylated intermediate leads to the formation of the alkene with specific E/Z geometry, driven by the nature of the semi-stabilized ylide. The rigorous control of pH during the workup, adjusting to 6-7 with citric acid, ensures the clean separation of the triphenylphosphine oxide byproduct, which is crucial for achieving the high purity levels (>97%) reported in the embodiments.
How to Synthesize Rosuvastatin Calcium Intermediate Efficiently
The synthesis of this critical pharmaceutical intermediate requires precise adherence to the seven-step protocol outlined in the patent to ensure consistent quality and yield. The process begins with the preparation of the Grignard reagent in THF, initiated by iodine and 1,2-dibromoethane, followed by the controlled addition of the epoxide. Subsequent steps involve careful management of exothermic reactions, particularly during the cyanide substitution and the permanganate oxidation. Operators must monitor reaction progress via TLC or HPLC to determine exact endpoints, ensuring that intermediates like the protected alcohol (Compound III) are fully converted before proceeding to oxidation. The final crystallization step, utilizing a hexane/ether mixture with seeding, is vital for isolating the product as a high-purity white solid suitable for downstream coupling.
- Prepare vinyl chloride Grignard reagent and react with R-epoxy chloropropane under CuCl catalysis to form (2R)-1-chloro-2-hydroxyl-4-amylene.
- Perform nucleophilic substitution with sodium cyanide, followed by alcoholysis with dry HCl gas to form the ester intermediate.
- Protect the hydroxyl group, oxidize the terminal alkene to a carboxylic acid using KMnO4, acylate with methyl chloroformate, and finalize with a Wittig reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the drastic simplification of the raw material portfolio; by shifting from specialized, high-cost anhydrides or bulky protected esters to commodity chemicals like vinyl chloride and R-epoxy chloropropane, the supply chain becomes significantly more resilient. These feedstocks are produced on a massive global scale, ensuring continuous availability and shielding the manufacturing process from the volatility often associated with niche fine chemical suppliers. This transition effectively de-risks the procurement strategy, allowing for long-term contracting and better inventory planning without the fear of single-source bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group manipulations and the use of low-cost oxidants like potassium permanganate directly translate to substantial reductions in the Cost of Goods Sold (COGS). By removing the need for bulky carboxy protective groups that require additional synthetic steps for installation and removal, the overall material throughput is improved, and solvent consumption is reduced. Furthermore, the high yields reported in the embodiments (often exceeding 80% for key steps like the Grignard coupling and cyanide substitution) minimize the loss of valuable chiral intermediates, ensuring that the input costs are maximized in the final output. This efficiency creates a leaner manufacturing model that is highly competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable production schedule. The tolerance of the process to standard industrial solvents like THF, dichloromethane, and acetone means that facilities do not require specialized equipment or exotic solvent recovery systems. The stability of the intermediates, particularly the silyl-protected species, allows for flexible batch scheduling and potential storage between steps if necessary, providing logistical flexibility. This reliability is crucial for meeting the stringent delivery timelines of downstream API manufacturers, reducing the risk of stockouts that can halt entire production lines for finished dosage forms.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is designed for scalability with minimal ecological footprint. The "three wastes" (waste water, waste gas, waste residue) are described as easy to handle, which simplifies the permitting process for new manufacturing lines and reduces the operational burden on waste treatment facilities. The use of aqueous workups and standard extraction protocols avoids the generation of persistent organic pollutants often associated with more complex organometallic cascades. This alignment with green chemistry principles not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturer, a key metric for modern pharmaceutical supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional routes in terms of yield, purity, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: What are the key advantages of this synthesis method over prior art like WO/2006/091771?
A: Unlike prior art which requires bulky carboxy protective groups to ensure stability and selectivity, this method utilizes common, inexpensive raw materials like vinyl chloride and achieves high yields under mild conditions, significantly reducing preparation costs and simplifying industrial scale-up.
Q: How is stereochemical purity maintained during the synthesis?
A: The process initiates with chiral R-epoxy chloropropane, and subsequent steps including the copper-catalyzed Grignard reaction and careful temperature control during oxidation (-35°C to 10°C ranges) preserve the chiral center, resulting in intermediates with enantiomeric excess (ee) values exceeding 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of cheap and easily accessible raw materials, stable process conditions, and easy disposal of three wastes, making it highly suitable for large-scale industrialization compared to methods using expensive anhydride compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex cardiovascular drugs hinges on the availability of high-quality, cost-effective intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our facility is equipped to handle the specific reagents and conditions required for this seven-step synthesis, including cryogenic reactions and sensitive organometallic transformations.
We invite global partners to collaborate with us to optimize their supply chains for Rosuvastatin Calcium production. By leveraging our expertise in process chemistry and our commitment to operational excellence, we can help you achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the cardiovascular therapeutic sector.
