Scalable Amidine Production via Benzoyl Chloride Mediation for Global Agrochemical Supply Chains
Scalable Amidine Production via Benzoyl Chloride Mediation for Global Agrochemical Supply Chains
The global demand for high-performance agrochemicals and pharmaceutical intermediates continues to drive innovation in organic synthesis, particularly for nitrogen-containing heterocycles. Patent CN102993109A introduces a transformative preparation method for amidine compounds that addresses long-standing inefficiencies in traditional manufacturing protocols. This technology leverages benzoyl chloride as a key mediator to facilitate the reaction between aminotriazole and acetonitrile under moderate thermal conditions, specifically ranging from 75 to 85 degrees Celsius. By shifting away from hazardous gaseous reagents and extreme pressure conditions, this novel approach offers a safer, more controllable pathway for producing critical intermediates used in miticides like Amitraz and insecticides like Acetamiprid. For R&D directors and procurement specialists, understanding the mechanistic advantages and scalability of this route is essential for optimizing supply chain resilience and reducing overall production costs in the competitive fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amidine compounds has relied heavily on the classical Pinner reaction or aluminum-mediated pathways, both of which present significant logistical and safety challenges for large-scale manufacturing. The Pinner reaction typically requires the saturation of nitrile solutions with dry hydrogen chloride gas followed by ammonolysis, a process that demands specialized corrosion-resistant equipment and rigorous safety protocols to handle toxic gases. 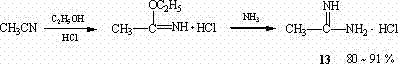 Furthermore, alternative methods utilizing alkyl aluminum reagents, while effective for specific substrates, involve moisture-sensitive organometallic chemistry that complicates reactor operations and increases raw material costs due to the need for strictly anhydrous conditions. These conventional routes often suffer from prolonged reaction times, sometimes extending up to 40 hours under high pressure, which severely limits throughput and increases energy consumption per kilogram of product. The generation of substantial acidic waste streams and the difficulty in removing metal residues from the final product further complicate downstream purification, creating bottlenecks that impact both environmental compliance and final product quality specifications.
Furthermore, alternative methods utilizing alkyl aluminum reagents, while effective for specific substrates, involve moisture-sensitive organometallic chemistry that complicates reactor operations and increases raw material costs due to the need for strictly anhydrous conditions. These conventional routes often suffer from prolonged reaction times, sometimes extending up to 40 hours under high pressure, which severely limits throughput and increases energy consumption per kilogram of product. The generation of substantial acidic waste streams and the difficulty in removing metal residues from the final product further complicate downstream purification, creating bottlenecks that impact both environmental compliance and final product quality specifications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102993109A utilizes benzoyl chloride to activate the nitrile component in a liquid-phase reaction that proceeds efficiently at atmospheric pressure. This method eliminates the need for handling dangerous dry HCl gas or expensive, pyrophoric aluminum reagents, thereby simplifying the reactor setup and reducing capital expenditure on specialized containment systems. The reaction operates within a moderate temperature window of 75 to 85 degrees Celsius, which is easily achievable with standard heating jackets and does not require cryogenic cooling or high-pressure autoclaves. By using acetonitrile as both a reactant and solvent, the process streamlines the material balance and reduces the volume of auxiliary solvents required, leading to a more concentrated and efficient reaction mixture. This operational simplicity translates directly into reduced cycle times, with optimal conversion achieved in approximately 4 hours, allowing for faster batch turnover and improved asset utilization in multipurpose chemical plants.
Mechanistic Insights into Benzoyl Chloride Mediated Amidination
The core innovation of this synthesis lies in the activation mechanism facilitated by benzoyl chloride, which likely functions by generating a reactive imidoyl chloride intermediate in situ. When benzoyl chloride interacts with the nitrile group of acetonitrile in the presence of the amine nucleophile (aminotriazole), it enhances the electrophilicity of the nitrile carbon, making it more susceptible to nucleophilic attack. 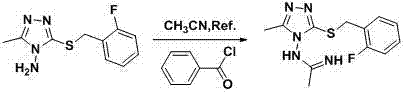 This activation lowers the energy barrier for the addition reaction, allowing the transformation to proceed rapidly at 78 degrees Celsius without the need for strong Lewis acid catalysts that are difficult to remove. The stoichiometry is carefully balanced, with a molar ratio of aminotriazole to benzoyl chloride to acetonitrile optimized at 1:1.0~1.5:50~100, ensuring that the activating agent is present in sufficient quantity to drive the reaction to completion while minimizing excess reagent waste. The mechanism avoids the formation of stable salt complexes that often trap products in traditional acid-catalyzed routes, thereby facilitating easier isolation of the free base amidine product during workup.
This activation lowers the energy barrier for the addition reaction, allowing the transformation to proceed rapidly at 78 degrees Celsius without the need for strong Lewis acid catalysts that are difficult to remove. The stoichiometry is carefully balanced, with a molar ratio of aminotriazole to benzoyl chloride to acetonitrile optimized at 1:1.0~1.5:50~100, ensuring that the activating agent is present in sufficient quantity to drive the reaction to completion while minimizing excess reagent waste. The mechanism avoids the formation of stable salt complexes that often trap products in traditional acid-catalyzed routes, thereby facilitating easier isolation of the free base amidine product during workup.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions associated with harsh acidic or basic conditions. Traditional ammonolysis often leads to over-alkylation or hydrolysis of the nitrile to the corresponding amide or carboxylic acid if water control is not perfect. The benzoyl chloride mediated pathway appears to be highly selective for the amidine functionality, as evidenced by the clean reaction profiles reported in the patent embodiments. The use of acetonitrile as the solvent also ensures high solubility of the polar intermediates, preventing precipitation that could lead to incomplete conversion or localized hot spots. Post-reaction processing involves simple concentration followed by standard purification techniques such as column chromatography using ethyl acetate and petroleum ether, or recrystallization, which effectively removes the benzoic acid byproduct and any unreacted starting materials, yielding a high-purity final product suitable for sensitive pharmaceutical or agrochemical applications.
How to Synthesize Amidine Compounds Efficiently
Implementing this synthesis route in a pilot or production facility requires strict adherence to the optimized parameters regarding temperature and reagent addition rates to maximize yield and safety. The process begins by charging the reactor with the solid aminotriazole and the liquid acetonitrile solvent, establishing a homogeneous suspension before the introduction of the activating agent. Benzoyl chloride is then added dropwise to control the exotherm and ensure uniform mixing, preventing local concentrations that could lead to degradation. Once the addition is complete, the mixture is heated to the target range of 75 to 85 degrees Celsius and maintained for a duration of 3 to 8 hours, with 4 hours identified as the optimal timeframe for balancing conversion and throughput.
- Charge a reaction vessel with aminotriazole and acetonitrile solvent under stirring conditions.
- Slowly drip benzoyl chloride into the mixture while maintaining temperature control.
- Heat the reaction mixture to 75-85°C for 3-8 hours, then concentrate and purify via chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this benzoyl chloride mediated synthesis offers tangible benefits in terms of cost structure and operational reliability. The elimination of high-pressure reactors and corrosion-resistant linings significantly reduces the capital intensity required for production, allowing manufacturers to utilize existing standard glass-lined or stainless steel equipment. This compatibility with standard infrastructure accelerates the timeline for technology transfer and scale-up, reducing the risk of delays associated with custom equipment fabrication. Furthermore, the reagents involved—benzoyl chloride and acetonitrile—are commodity chemicals available from multiple global suppliers, ensuring a robust and competitive supply base that mitigates the risk of single-source dependency.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the simplification of the operational workflow and the reduction in utility consumption. By avoiding the need for cryogenic cooling or high-pressure compression of gases, the energy footprint per kilogram of product is drastically lowered. Additionally, the shorter reaction time of approximately 4 hours compared to multi-day traditional methods increases the annual capacity of existing production assets without requiring physical expansion. The avoidance of expensive organometallic catalysts like alkyl aluminum chlorides further reduces the raw material cost bill, while the simplified workup procedure minimizes solvent usage and waste disposal fees, contributing to a leaner overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain continuity is strengthened by the use of stable, shelf-stable liquid reagents that do not require special storage conditions such as inert atmosphere or freezing. Unlike moisture-sensitive aluminum reagents that degrade upon exposure to air, benzoyl chloride can be handled with standard industrial hygiene protocols, reducing the likelihood of batch failures due to reagent decomposition. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in ambient temperature or humidity, ensuring consistent batch-to-batch quality and reliable delivery schedules to downstream customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of hazardous gas evolution during the main reaction phase. The primary byproduct, benzoic acid, is a solid that can be easily separated and potentially recycled or sold as a co-product, aligning with green chemistry principles of atom economy. The reduction in acidic wastewater generation compared to the Pinner reaction simplifies effluent treatment requirements, lowering the burden on environmental management systems and ensuring compliance with increasingly stringent regulatory standards for chemical manufacturing facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amidine synthesis technology, based on the data provided in the patent documentation. These insights are designed to clarify the operational parameters and potential integration points for partners looking to adopt this method for their own production lines.
Q: What are the primary advantages of using benzoyl chloride over traditional Pinner reaction conditions?
A: The benzoyl chloride method avoids the use of hazardous dry HCl gas and high-pressure ammonia required in traditional Pinner reactions, significantly improving operational safety and reducing equipment corrosion risks.
Q: What is the typical yield and purity profile for this amidine synthesis route?
A: Embodiments in the patent demonstrate yields around 65% with simple purification steps like column chromatography or recrystallization, indicating a robust process suitable for industrial scaling.
Q: Can this method be applied to the synthesis of specific agrochemical active ingredients?
A: Yes, amidine structures are key intermediates for major agrochemicals like Amitraz and Acetamiprid, making this synthetic route highly relevant for pesticide manufacturing supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader value chain of agrochemical and pharmaceutical development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of amidine intermediate meets the exacting standards required for downstream active ingredient synthesis, providing our partners with the confidence needed to accelerate their own product development timelines.
We invite global partners to engage with our technical procurement team to discuss how this novel benzoyl chloride mediated route can be integrated into your supply strategy. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your long-term growth and competitiveness in the global market.
