Advanced Crystallization Technology for High-Purity Losartan Manufacturing and Commercial Scale-Up
Introduction to Advanced Losartan Purification Technologies
The pharmaceutical industry continuously demands higher purity standards for Active Pharmaceutical Ingredients (APIs) to ensure patient safety and regulatory compliance. Patent CN105198863B introduces a groundbreaking method for preparing high-purity Losartan, a critical angiotensin II receptor antagonist used globally for hypertension treatment. Unlike conventional approaches that rely on complex salt formation and dissociation cycles, this innovation utilizes a sophisticated recrystallization technique to directly purify the Losartan free acid. By carefully manipulating solvent systems comprising tetrahydrofuran, acetone, or methanol, potentially mixed with water, the process achieves exceptional purity levels exceeding 99.5%. This technical breakthrough addresses the persistent challenge of removing polymeric impurities that typically plague the synthesis of biphenyl-based intermediates. For a reliable pharmaceutical intermediate supplier, mastering such purification logic is essential to delivering materials that meet the stringent requirements of global regulatory bodies.
The significance of this patent lies in its ability to bypass the traditional, loss-heavy conversion to Losartan Potassium and back. Historically, achieving purity above 99% required converting the crude free acid into a potassium salt, purifying it, and then regenerating the acid, a sequence that introduced unnecessary operational complexity and yield loss. The disclosed method simplifies this landscape by optimizing the thermodynamic conditions of crystallization. By heating the crude material to temperatures between 20-80°C and subsequently cooling to 0-5°C, the process leverages solubility differentials to exclude impurities from the crystal lattice. This approach not only streamlines the manufacturing workflow but also significantly enhances the overall economic efficiency of producing high-quality antihypertensive agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for Losartan have long been hindered by the difficulty of separating structurally similar impurities and polymeric byproducts formed during the initial coupling reactions. Standard protocols often necessitate the formation of Losartan Potassium as an intermediate purification step, relying on the differential solubility of the salt form to remove contaminants. However, this salt-formation route is inherently inefficient, involving multiple reaction steps including neutralization, precipitation, and subsequent acidification to recover the free acid. Each additional unit operation introduces potential points of failure, increases solvent consumption, and inevitably leads to substantial cumulative yield losses. Furthermore, the handling of strong bases and acids required for salt conversion poses additional safety and environmental challenges, complicating waste management and increasing the overall cost burden for manufacturers striving to remain competitive in the generic drug market.
The Novel Approach
The novel approach detailed in the patent revolutionizes this workflow by demonstrating that high-purity Losartan can be obtained directly from the crude free acid through precise solvent engineering. By selecting specific organic solvents like tetrahydrofuran or acetone and controlling the water content, the process creates an environment where the target molecule crystallizes with high selectivity. The method allows for the direct isolation of Losartan with single impurity levels reduced to below 0.2%, effectively matching or exceeding the quality of material produced via the more arduous salt-conversion routes. This direct crystallization strategy eliminates the need for pH swings and salt handling, thereby reducing the number of processing steps and minimizing the residence time of the product in solution, which further mitigates the risk of degradation or polymerization. For procurement teams, this translates to a more robust and predictable supply chain for high-purity Losartan.
Mechanistic Insights into Solvent-Mediated Recrystallization
The core mechanism driving this purification success is the careful manipulation of solubility profiles and supersaturation kinetics. In the first stage, the crude Losartan is dissolved in a solvent system at elevated temperatures ranging from 40-70°C. At this thermal energy level, both the target API and the associated impurities, including oligomers and unreacted starting materials, are fully solubilized. The choice of solvent is critical; polar aprotic solvents like THF or ketones like acetone interact favorably with the imidazole and tetrazole moieties of the Losartan structure, ensuring complete dissolution. As the system is cooled to 0-5°C, the solubility of Losartan drops precipitously, inducing nucleation and crystal growth. The rate of cooling and the presence of anti-solvents like water are tuned to favor the formation of a pure crystal lattice, effectively rejecting impurity molecules that do not fit sterically or electronically into the growing crystal structure.
Impurity control is further enhanced by the specific interaction between the solvent matrix and the polymeric byproducts often generated during the synthesis of the biphenyl-imidazole scaffold. These polymeric species tend to remain in the mother liquor due to their differing polarity and molecular weight compared to the monomeric Losartan. The patent data indicates that by adjusting the volume ratio of solvent to crude product (preferably 3.0-5.0 ml/g) and controlling the water addition (0.2-0.5 ml/g), the process maximizes the rejection of these high-molecular-weight contaminants. This mechanistic understanding allows for the consistent production of material with purity reaching 99.6%, ensuring that the final API meets the rigorous specifications required for formulation into oral tablets. Such precise control over the solid-state properties is vital for ensuring consistent bioavailability and stability in the final drug product.
How to Synthesize High-Purity Losartan Efficiently
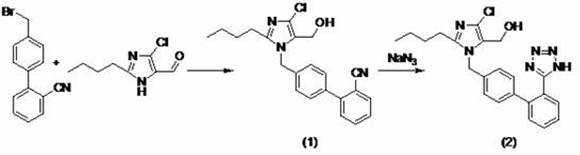
The synthesis of high-purity Losartan via this patented recrystallization method offers a streamlined pathway for manufacturers seeking to optimize their production lines. The process begins with the preparation of the crude Losartan, typically derived from the condensation of biphenyl bromide and imidazole aldehyde followed by azide substitution, as illustrated in the reaction schemes. Once the crude material is obtained, the purification protocol involves dissolving the solid in a heated organic solvent, maintaining the temperature to ensure homogeneity, and then executing a controlled cooling ramp to induce crystallization. This operational simplicity belies the sophisticated thermodynamic principles at play, which allow for the efficient separation of the target molecule from complex impurity profiles. Detailed standardized synthetic steps for implementing this purification protocol are provided in the guide below.
- Dissolve the Losartan crude product in a selected organic solvent such as tetrahydrofuran, acetone, or methanol, potentially mixed with water, and heat the mixture to a temperature range of 20-80°C with continuous stirring to ensure complete solubilization.
- Initiate the crystallization phase by cooling the system to 0-5°C, optionally adding water or evaporating a portion of the solvent prior to cooling to induce supersaturation and precipitate the purified product.
- Separate the high-purity Losartan crystals through filtration, wash if necessary, and dry the final product to obtain material with purity exceeding 99.5% and single impurity levels below 0.2%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this direct purification technology represents a significant strategic advantage in the sourcing of pharmaceutical intermediates. By eliminating the intermediate salt-formation step, manufacturers can drastically reduce the number of unit operations required, leading to shorter production cycles and lower capital expenditure on equipment. The simplified workflow reduces the dependency on multiple reagents, such as strong bases and mineral acids, which not only lowers raw material costs but also simplifies the logistics of hazardous chemical handling. Furthermore, the high yields reported in the patent examples suggest that less starting material is wasted, improving the overall atom economy of the process. This efficiency gain is crucial for maintaining competitive pricing in the generic pharmaceutical market while ensuring a steady supply of high-quality intermediates.
- Cost Reduction in Manufacturing: The elimination of the salt conversion cycle removes entire stages of processing, including neutralization, filtration of salts, and re-acidification. This reduction in process complexity directly correlates to lower utility consumption, reduced labor hours, and decreased solvent usage. Without the need to purchase and handle additional reagents for salt formation and dissociation, the variable cost per kilogram of the final product is significantly optimized. Additionally, the high recovery yields minimize the financial impact of material loss, ensuring that the maximum amount of valuable intermediate is converted into saleable product, thereby enhancing the overall profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: A simpler process with fewer steps inherently carries lower operational risk, leading to more reliable delivery schedules for downstream API manufacturers. The robustness of the recrystallization method means that batch-to-batch variability is minimized, reducing the likelihood of failed quality control tests that could delay shipments. By sourcing from a supplier utilizing this efficient technology, procurement teams can mitigate the risk of supply disruptions caused by complex processing bottlenecks. The ability to rapidly turn around batches due to the shortened cycle time ensures that inventory levels can be maintained more effectively, supporting just-in-time manufacturing strategies for major pharmaceutical clients.
- Scalability and Environmental Compliance: The use of common organic solvents like acetone and methanol, which are widely available and easily recycled, facilitates seamless scale-up from pilot to commercial production volumes. The process avoids the generation of large volumes of inorganic salt waste that typically result from acid-base neutralization steps, thereby reducing the environmental footprint and lowering waste disposal costs. This alignment with green chemistry principles not only aids in meeting increasingly strict environmental regulations but also enhances the sustainability profile of the supply chain. The ease of scaling this technology ensures that supply can be rapidly expanded to meet surging market demand without the need for extensive new infrastructure investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and supply of high-purity Losartan based on the patented crystallization technology. These insights are derived directly from the experimental data and process descriptions found in the intellectual property documentation. Understanding these details helps stakeholders evaluate the feasibility and benefits of integrating this material into their supply chains. Comprehensive answers are provided below to clarify the technical advantages and operational implications.
Q: How does this purification method differ from traditional salt-formation techniques?
A: Traditional methods often require converting Losartan to its potassium salt and then back to the free acid to improve purity, a process that is complex and results in significant material loss. This patented method achieves high purity directly through controlled recrystallization of the free acid form, eliminating the need for salt conversion steps.
Q: What level of impurity reduction can be expected with this process?
A: The process is highly effective at removing polymeric impurities and side-products generated during synthesis. It consistently reduces single impurity levels to below 0.2%, and in optimized conditions, even below 0.1%, achieving a total purity of up to 99.6%.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method relies on standard unit operations such as dissolution, heating, cooling crystallization, and filtration. These are inherently scalable processes that do not require exotic catalysts or extreme pressure conditions, making them ideal for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Losartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process efficiency in the production of cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. We are committed to delivering high-purity Losartan that adheres to stringent purity specifications, leveraging advanced crystallization technologies to minimize impurities and maximize yield. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch, guaranteeing that the material supplied meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for Losartan and related intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization. Let us be your partner in delivering high-quality healthcare solutions to the world.
