Optimizing Dienogest Production: A Technical Analysis of High-Yield Steroid Intermediates
Optimizing Dienogest Production: A Technical Analysis of High-Yield Steroid Intermediates
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex hormonal active pharmaceutical ingredients (APIs), and the synthesis of dienogest represents a critical area of innovation for contraceptive therapeutics. Patent CN101993468A introduces a significantly refined synthetic methodology that addresses historical inefficiencies in producing this potent progestogen. By utilizing 3-methoxy-17-hydroxy-estra-2,5(10)-diene as a strategic starting material, the disclosed process streamlines the transformation through oxidation, cyanation, hydrolysis, and final functionalization steps. This technical insight report analyzes the mechanistic advantages of this route, highlighting how precise temperature control and reagent selection drive superior yield and purity profiles essential for reliable dienogest intermediate supplier partnerships. The elimination of harsh reduction conditions found in legacy routes marks a pivotal shift towards greener, more scalable cost reduction in hormonal API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing dienogest, such as those disclosed in U.S. Patent 4248790A, often relied on cumbersome multi-step sequences beginning with estrone-3-methyl ether. These conventional routes typically necessitated a Birch reduction to establish the requisite diene system, followed by complex epoxidation and ring-opening strategies to install the C17 substituents. Such methodologies frequently suffered from low atom economy and required rigorous purification between steps to remove stereoisomeric impurities that are difficult to separate. Furthermore, the use of strong reducing agents and multiple protection-deprotection cycles increased the operational complexity and safety risks associated with large-scale production. The cumulative yield losses across these extended sequences often rendered the final API cost-prohibitive for generic manufacturers seeking commercial scale-up of complex steroid intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN101993468A leverages a direct oxidation-cyanation strategy that bypasses the need for initial ring reduction. As illustrated in the reaction scheme below, the process initiates with an Oppenauer-type oxidation to generate the 17-ketone, followed immediately by a highly selective nucleophilic addition.
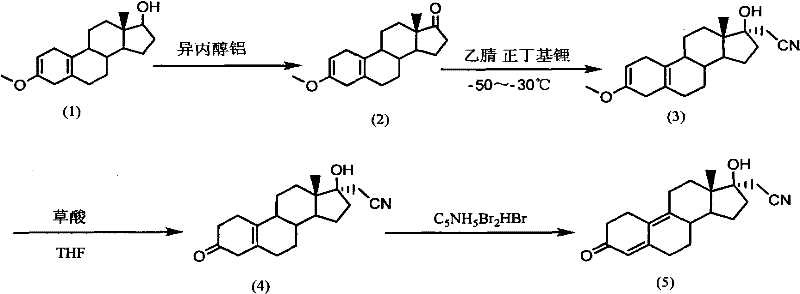
This novel approach capitalizes on the inherent reactivity of the 2,5(10)-diene system, allowing for the direct installation of the cyanomethyl group with exceptional stereocontrol. By avoiding the lengthy reconstruction of the steroid backbone required in older methods, this route significantly shortens the critical path to the final API. The integration of mild hydrolysis conditions using oxalic acid further distinguishes this method, offering a cleaner profile that reduces the burden on downstream purification units. For procurement teams, this translates to a more resilient supply chain capable of delivering high-purity dienogest precursors with reduced lead times.
Mechanistic Insights into Temperature-Controlled Cyanation and Hydrolysis
The cornerstone of this improved synthesis lies in the precise thermal management of the cyanation and hydrolysis steps, which dictates the overall impurity profile. During the cyanation phase, the generation of lithioacetonitrile from n-butyllithium and acetonitrile is conducted at cryogenic temperatures ranging from -50°C to -30°C. This specific thermal window is critical for suppressing competitive deprotonation events and ensuring that the nucleophilic attack on the 17-ketone occurs exclusively from the alpha-face, yielding the desired 17-beta-hydroxy-17-alpha-cyanomethyl configuration. Experimental data within the patent indicates that maintaining this range boosts yields to over 90%, a substantial improvement over the 78% observed in broader temperature ranges. Such kinetic control is vital for R&D directors focused on maximizing throughput without compromising stereochemical integrity.
Subsequent hydrolysis of the methoxy group and the nitrile functionality requires equally stringent parameters to prevent the formation of degradation byproducts. The use of oxalic acid in a tetrahydrofuran-water system at 17°C to 20°C creates an ideal environment for demethylation while preserving the sensitive diene structure. The patent data explicitly notes that allowing the temperature to drift to 25°C results in the emergence of approximately 10% impurities, likely due to acid-catalyzed isomerization or hydration of the double bonds. By adhering to the narrower 17-20°C window, the process consistently achieves purity levels exceeding 98% (HPLC) prior to the final bromination step. This level of control minimizes the need for extensive chromatographic purification, thereby enhancing the economic viability of the process for industrial applications.
How to Synthesize Dienogest Intermediates Efficiently
The execution of this synthetic pathway requires careful attention to reagent stoichiometry and phase separation techniques to ensure optimal recovery of intermediates. The initial oxidation step utilizes aluminum isopropoxide and pimelinketone in toluene, where the post-reaction workup involves a strategic hot filtration at 70-80°C to remove aluminum salts before crystallization. Following this, the cyanation step demands anhydrous conditions and inert atmosphere handling to maintain the activity of the organolithium species. The subsequent hydrolysis and final bromination-debromination sequence utilize pyridinium tribromide to establish the final 4,9-diene-3-one system characteristic of dienogest. For detailed operational parameters and safety protocols regarding these transformations, please refer to the standardized guide below.
- Perform Oppenauer oxidation on 3-methoxy-17-hydroxy-estra-2,5(10)-diene using aluminum isopropoxide and pimelinketone at 110°C, followed by controlled cooling and hot filtration to isolate the 17-ketone intermediate.
- Execute nucleophilic cyanation by generating lithioacetonitrile from n-butyllithium and acetonitrile at -50 to -30°C, reacting with the ketone intermediate to form the 17-alpha-cyanomethyl-17-beta-hydroxy derivative.
- Conduct acidic hydrolysis and demethylation using oxalic acid in THF/water at a strictly controlled temperature of 17-20°C to ensure >98% purity before final bromination and debromination.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this improved synthetic route offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line for API manufacturers. The simplification of the synthetic sequence reduces the number of unit operations required, which in turn lowers utility consumption and labor costs associated with batch processing. By eliminating the need for hazardous Birch reduction conditions and complex epoxide handling, the process inherently reduces safety liabilities and waste disposal costs, aligning with modern environmental compliance standards. These factors collectively contribute to a more stable pricing structure for the final API, making it an attractive option for generic drug developers aiming to penetrate competitive markets.
- Cost Reduction in Manufacturing: The most significant economic driver in this process is the dramatic improvement in yield during the cyanation step, where yields exceed 90% compared to the sub-80% benchmarks of prior art. This increase in efficiency means that less raw material is wasted per kilogram of output, directly lowering the cost of goods sold (COGS). Furthermore, the ability to isolate intermediates with high purity (>98%) via simple crystallization rather than column chromatography significantly reduces solvent usage and processing time. The elimination of expensive transition metal catalysts or specialized reagents further streamlines the bill of materials, ensuring that cost reduction in hormonal API manufacturing is achieved through fundamental process intensification rather than superficial cuts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aluminum isopropoxide, oxalic acid, and n-butyllithium ensures that the supply chain remains robust against fluctuations in specialty reagent availability. The process tolerances, particularly the defined temperature windows, are achievable with standard industrial reactor equipment, removing the need for exotic cryogenic infrastructure that can bottleneck production capacity. This accessibility allows for flexible manufacturing scheduling and rapid scale-up capabilities, ensuring that partners can meet fluctuating market demands for contraceptive APIs without significant lead time delays. The consistent quality of the intermediates also reduces the risk of batch failures, securing a steady flow of materials for downstream formulation.
- Scalability and Environmental Compliance: The aqueous workups and organic extractions described in the patent are amenable to continuous processing or large-batch operations, facilitating seamless technology transfer from pilot plant to commercial scale. The use of oxalic acid for hydrolysis generates benign byproducts compared to harsh mineral acids, simplifying wastewater treatment protocols and reducing the environmental footprint of the facility. Additionally, the high selectivity of the bromination-debromination sequence minimizes the formation of halogenated waste streams, aligning with increasingly stringent global regulations on chemical manufacturing emissions. This sustainability profile enhances the long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is essential for process chemists evaluating the feasibility of adopting this methodology for their own production lines. The answers reflect the specific improvements in yield and purity that distinguish this approach from legacy methods.
Q: How does the improved cyanation step enhance yield compared to prior art?
A: By strictly controlling the reaction temperature between -50°C and -30°C during the addition of the ketone to lithioacetonitrile, the process minimizes side reactions, achieving yields exceeding 90% compared to the 78% reported in previous methods like WO2007066158A.
Q: What is the critical control point for ensuring intermediate purity during hydrolysis?
A: The hydrolysis step using oxalic acid must be maintained specifically between 17°C and 20°C. Deviating from this narrow window, particularly rising to 25°C, can generate approximately 10% impurities, whereas strict adherence ensures purity levels above 98% (HPLC).
Q: Why is hot filtration utilized in the initial oxidation step?
A: Cooling the reaction mixture to 70-80°C before adding water facilitates the decomposition of excess aluminum isopropoxide into aluminum hydroxide. Hot filtration at this stage effectively removes these aluminum salts and other insoluble impurities before concentration, significantly improving the quality of the crude ketone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dienogest Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex hormones like dienogest depends on a partner who understands both the chemistry and the supply chain dynamics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101993468A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze steroid intermediates at every stage, guaranteeing that the material you receive meets the highest international pharmacopeial standards. Our commitment to technical excellence ensures that your API development timeline remains on track.
We invite you to engage with our technical procurement team to discuss how this optimized route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this high-yield methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value for your organization and secure your position in the global hormonal therapy market.
